An infiltration problem occurs if the irrigation water does not enter the soil rapidly enough during a normal irrigation cycle to replenish the soil with water needed by the crop before the next irrigation. The reduced infiltration rate, if due to quality of applied water, is generally a problem within the upper few centimetres of soil but occasionally may occur at greater depths. The end result is a decrease in water supply to the crop, similar to the reduction due to salinity, but for a different reason. A water infiltration problem reduces the quantity of water put into the soil for later use by the crop while salinity reduces the availability of the water in storage.
Infiltration refers to the entry of water into the soil. The rate at which water enters is referred to as the rate of infiltration. Permeability, the term used in the previous edition of Irrigation and Drainage Paper 29 (1976), more correctly refers to the percolation of infiltrated water through the soil. Since the water quality problem is primarily one related to the ease with which water enters and moves through the upper few centimetres of soil, we have chosen the term 'infiltration problem' rather than the previously used term 'permeability problem'. An infiltration rate as low as 3 mm/hour is considered low while a rate above 12 mm/hour is relatively high. This can be affected, however, by many factors other than water quality, including physical characteristics of the soil, such as soil texture and type of clay minerals, and chemical characteristics including exchangeable cations. The guidelines of Table 1 refer to infiltration problems as they relate directly to the unfavourable changes in soil chemistry caused by the quality of irrigation water applied. These problems concern both salinity and relative sodium content in the applied water. Figure 21 shows in graphic form that both salinity (ECw) and the sodium adsorption ratio (SAR) of the applied water affect the rate of infiltration of water into surface soil. Figure 21 can be used in place of the numerical evaluations in Table 1 given for infiltration problems.
The infiltration rate generally increases with increasing salinity and decreases with either decreasing salinity or increasing sodium content relative to calcium and magnesium - the sodium adsorption ratio. Therefore, the two factors, salinity and SAR, must be considered together for a proper evaluation of the ultimate effect on water infiltration rate.
Low salinity water (less than 0.5 dS/m and especially below 0.2 dS/m) is corrosive and tends to leach surface soils free of soluble minerals and salts, especially calcium, reducing their strong stabilizing influence on soil aggregates and soil structure. Without salts and without calcium, the soil disperses and the dispersed finer soil particles fill many of the smaller pore spaces, sealing the surface and greatly reducing the rate at which water infiltrates the soil surface. Soil crusting and crop emergence problems often result, in addition to a reduction in the amount of water that will enter the soil in a given amount of time and which may ultimately cause water stress between irrigations.
Very low salinity water (less than ECw = 0.2 ds/m) almost invariably results in water infiltration problems, regardless of the relative sodium ratio (or SAR). Rainfall is a very low salinity water and irrigated areas frequently experience exceptionally low rates of infiltration of rainfall resulting in excessive runoff.
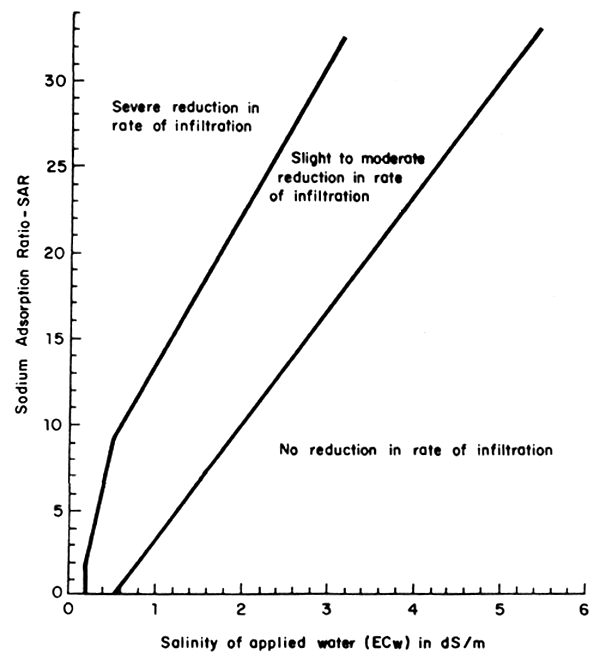
Fig. 21 Relative rate of water infiltration as affected by salinity and sodium adsorption ratio (Adapted from Rhoades 1977; and Oster and Schroer 1979)
Excessive sodium in irrigation water also promotes soil dispersion and structural breakdown but only if sodium exceeds calcium by more than a ratio of about 3:1. Such a relatively high sodium content (>3:1) often results in a severe water infiltration problem due to soil dispersion and plugging and sealing of the surface pores, in much the same way as does the very low salinity water. This is due to lack of sufficient calcium to counter the dispersing effects of the sodium. Excessive sodium may also make it extremely difficult to supply enough water to meet the crop water demand. Other related problems such as soil crusting, poor seedling emergence, lack of aeration, plant and root diseases, weed and mosquito control problems caused by the low rate of infiltration may further complicate crop management.
In the past, several procedures have been used to predict a potential infiltration problem. The Residual Sodium Carbonate (RSC) method (Eaton 1950; Richards 1954) was widely used at one time. The most commonly used recent method to evaluate the infiltration problem potential has been and probably still is the Sodium Adsorption Ratio (SAR) (Richards 1954). The SAR equation (1) as given in Figure 1 is:

| where: | Na | = | sodium in me/l |
| Ca | = | calcium in me/l | |
| Mg | = | magnesium in me/l |
In recent reports and journal articles, SAR is more and more frequently being reported as RNa and not SAR. The terms are synonymous. The SAR procedure encompasses the infiltration problems due to an excess of sodium in relation to calcium and magnesium. It does not take into account changes in calcium in the soil water that take place because of changes in solubility of calcium resulting from precipitation or dissolution during or following an irrigation. Sodium, an important part of salinity, remains soluble and in equilibrium with exchangeable soil sodium at all times. Whether concentrated from with-drawal of water by the crop between long irrigation intervals, diluted with applied water, or leached away in drainage, outside influences have little effect on sodium solubility or precipitation. Calcium, however, does not remain completely soluble or in constant supply but is constantly changing until an equilibrium is established. Calcium changes occur due to dissolution of soil minerals into the soil-water thus raising its calcium content, or to precipitation from soil-water, usually as calcium carbonate, thus reducing the calcium. Dissolution is encouraged by dilution and by carbon dioxide dissolved in the soil-water; precipitation may take place because of the presence of sufficient calcium along with enough carbonate, bicarbonate or sulphates to exceed the solubility of calcium carbonate (limestone) or calcium sulphate (gypsum). Soon after an irrigation, dissolution or precipitation may occur, changing the supply of calcium and establishing an equilibrium at a new calcium concentration, different to that in the applied water. The SAR equation, since it does not account for these changes, is therefore somewhat in error. However, the SAR equation and procedure is still considered an acceptable evaluation procedure for most of the irrigation water encountered in irrigated agriculture.
NOTE
The adjusted SAR procedure presented in the previous edition of this paper (Ayers and Westcot 1976) is no longer recommended. Oster and Rhoades (1977), Oster and Schroer (1979) and Suarez (1981) carefully evaluated that procedure and concluded that it overpredicts the sodium hazard. They suggest that, if used, the value obtained by that method should be further adjusted by an 0.5 factor to evaluate more correctly the effects of HCO3 on calcium precipitation (adj SAR × 0.5).
In this present edition the newer adj RNa procedure of Suarez (1981) is recommended but both the older SAR procedure and the new adj RNa are acceptable, with a preference expressed towards the adj RNa because it and the Cax of Table II offer a better insight into the change in calcium in the soil-water due to addition by dissolution of calcium from soil carbonates and silicates, or loss of calcium from soil-water by precipitation as carbonates.
| Salinity of applied water (ECw) (dS/m) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.1 | 0.2 | 0.3 | 0.5 | 0.7 | 1.0 | 1.5 | 2.0 | 3.0 | 4.0 | 6.0 | 8.0 | ||
| Ratio of HCO3/Ca | .05 | 13.20 | 13.61 | 13.92 | 14.40 | 14.79 | 15.26 | 15.91 | 16.43 | 17.28 | 17.97 | 19.07 | 19.94 |
| .10 | 8.31 | 8.57 | 8.77 | 9.07 | 9.31 | 9.62 | 10.02 | 10.35 | 10.89 | 11.32 | 12.01 | 12.56 | |
| .15 | 6.34 | 6.54 | 6.69 | 6.92 | 7.11 | 7.34 | 7.65 | 7.90 | 8.31 | 8.64 | 9.17 | 9.58 | |
| .20 | 5.24 | 5.40 | 5.52 | 5.71 | 5.87 | 6.06 | 6.31 | 6.52 | 6.86 | 7.13 | 7.57 | 7.91 | |
| .25 | 4.51 | 4.65 | 4.76 | 4.92 | 5.06 | 5.22 | 5.44 | 5.62 | 5.91 | 6.15 | 6.52 | 6.82 | |
| .30 | 4.00 | 4.12 | 4.21 | 4.36 | 4.48 | 4.62 | 4.82 | 4.98 | 5.24 | 5.44 | 5.77 | 6.04 | |
| .35 | 3.61 | 3.72 | 3.80 | 3.94 | 4.04 | 4.17 | 4.35 | 4.49 | 4.72 | 4.91 | 5.21 | 5.45 | |
| .40 | 3.30 | 3.40 | 3.48 | 3.60 | 3.70 | 3.82 | 3.98 | 4.11 | 4.32 | 4.49 | 4.77 | 4.98 | |
| .45 | 3.05 | 3.14 | 3.22 | 3.33 | 3.42 | 3.53 | 3.68 | 3.80 | 4.00 | 4.15 | 4.41 | 4.61 | |
| .50 | 2.84 | 2.93 | 3.00 | 3.10 | 3.19 | 3.29 | 3.43 | 3.54 | 3.72 | 3.87 | 4.11 | 4.30 | |
| .75 | 2.17 | 2.24 | 2.29 | 2.37 | 2.43 | 2.51 | 2.62 | 2.70 | 2.84 | 2.95 | 3.14 | 3.28 | |
| 1.00 | 1.79 | 1.85 | 1.89 | 1.96 | 2.01 | 2.09 | 2.16 | 2.23 | 2.35 | 2.44 | 2.59 | 2.71 | |
| 1.25 | 1.54 | 1.59 | 1.63 | 1.68 | 1.73 | 1.78 | 1.86 | 1.92 | 2.02 | 2.10 | 2.23 | 2.33 | |
| 1.50 | 1.37 | 1.41 | 1.44 | 1.49 | 1.53 | 1.58 | 1.65 | 1.70 | 1.79 | 1.86 | 1.97 | 2.07 | |
| 1.75 | 1.23 | 1.27 | 1.30 | 1.35 | 1.38 | 1.43 | 1.49 | 1.54 | 1.62 | 1.68 | 1.78 | 1.86 | |
| 2.00 | 1.13 | 1.16 | 1.19 | 1.23 | 1.26 | 1.31 | 1.36 | 1.40 | 1.48 | 1.54 | 1.63 | 1.70 | |
| 2.25 | 1.04 | 1.08 | 1.10 | 1.14 | 1.17 | 1.21 | 1.26 | 1.30 | 1.37 | 1.42 | 1.51 | 1.58 | |
| 2.50 | 0.97 | 1.00 | 1.02 | 1.06 | 1.09 | 1.12 | 1.17 | 1.21 | 1.27 | 1.32 | 1.40 | 1.47 | |
| 3.00 | 0.85 | 0.89 | 0.91 | 0.94 | 0.96 | 1.00 | 1.04 | 1.07 | 1.13 | 1.17 | 1.24 | 1.30 | |
| 3.50 | 0.78 | 0.80 | 0.82 | 0.85 | 0.87 | 0.90 | 0.94 | 0.97 | 1.02 | 1.06 | 1.12 | 1.17 | |
| 4.00 | 0.71 | 0.73 | 0.75 | 0.78 | 0.80 | 0.82 | 0.86 | 0.88 | 0.93 | 0.97 | 1.03 | 1.07 | |
| 4.50 | 0.66 | 0.68 | 0.69 | 0.72 | 0.74 | 0.76 | 0.79 | 0.82 | 0.86 | 0.90 | 0.95 | 0.99 | |
| 5.00 | 0.61 | 0.63 | 0.65 | 0.67 | 0.69 | 0.71 | 0.74 | 0.76 | 0.80 | 0.83 | 0.88 | 0.93 | |
| 7.00 | 0.49 | 0.50 | 0.52 | 0.53 | 0.55 | 0.57 | 0.59 | 0.61 | 0.64 | 0.67 | 0.71 | 0.74 | |
| 10.00 | 0.39 | 0.40 | 0.41 | 0.42 | 0.43 | 0.45 | 0.47 | 0.48 | 0.51 | 0.53 | 0.56 | 0.58 | |
| 20.00 | 0.24 | 0.25 | 0.26 | 0.26 | 0.27 | 0.28 | 0.29 | 0.30 | 0.32 | 0.33 | 0.35 | 0.37 | |
| 30.00 | 0.18 | 0.19 | 0.20 | 0.20 | 0.21 | 0.21 | 0.22 | 0.23 | 0.24 | 0.25 | 0.27 | 0.28 | |
3 Cax, HCO3, Ca are reported in me/l; ECw is in dS/m.
An alternative procedure, discussed in the following paragraphs, takes a new look at the older SAR equation and adjusts the calcium concentration of the irrigation water to the expected equilibrium value following an irrigation, and includes the effects of carbon dioxide (CO2), of bicarbonate (HCO3) and of salinity (ECw) upon the calcium originally present in the applied water but now a part of the soil-water. The procedure assumes a soil source of calcium - from soil lime (CaCO3) or other soil minerals such as silicates - and no precipitation of magnesium.
The new term for this is adj RNa (adjusted Sodium Adsorption Ratio) and the calculation procedure is presented in the following example as an improvement on the older Sodium Adsorption Ratio (SAR). It can be used to predict more correctly potential infiltration problems due to relatively high sodium (or low calcium) in irrigation water supplies (Suarez 1981; Rhoades 1982) and can be substituted for SAR in Table 1. The equation for calculation of adj RNa of the surface soil is very similar to the older SAR equation and is:
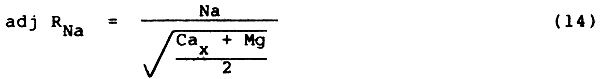
| where: | Na | = | sodium in the irrigation water reported in me/l |
| Cax | = | a modified calcium value taken from Table 11, reported in me/l. Cax represents Ca in the applied irrigation water but modified due to salinity of the applied water (ECw), its HCO3/Ca ratio (HCO3 and Ca in me/l) and the estimated partial pressure of CO2 in the surface few millimetres of soil (PCO2 = 0.0007 atmospheres) | |
| Mg | = | magnesium in the irrigation water reported in me/l |
To use the Cax table (Table 11), first determine the HCO3 to Ca ration (HCO3/Ca) and ECw from the water analysis, using HCO3 and Ca in me/l and the water salinity (ECw) in deciSiemens per metre. An appropriate range of calculated HCO3/Ca ratios appears on the left side of the table and the range of ECw across the top. Find the HCO3/Ca ratio that falls nearest to the calculated HCO3/Ca value for the subject water and read across to the ECw column that most closely approximates the ECw for the water being evaluated. The Cax value shown represents the me/l of Ca that is expected to remain in solution in the soil water at equilibrium and is to be used in equation (14). In Example 6, the three calculation procedures are compared 1) SAR, 2) adj SAR from FAO-29 1976, and 3) adj RNa.
The adj RNa obtained is used in place of the SAR in Table I to evaluate better the potential of the water to cause an infiltration problem if used for irrigation. Comparison of SAR and adj RNa for various types of water from around the world are presented in Annex I. The data in Annex I show that for most water, the SAR calculation is within ±10 percent of the value obtained after adjustment of the calcium concentration using equation (14) and Table 11.
If computer facilities are available, a simulation model can be relied upon to give valid evaluations of these adjusted sodium adsorption ratios (adj RNa). The adj RNa outlined in the foregoing is adapted from the procedure of Suarez (1981). A computer simulation model is also available (Rhoades 1982). Both give closely comparable results.
EXAMPLE 6 - COMPARISON OF METHODS TO CALCULATE THE SODIUM HAZARD OF A WATER
| Given: | The water analysis is: | ||
| Ca | = | 2.32 me/l | |
| Mg | = | 1.44 me/l | |
| Na | = | 7.73 me/l | |
| Sum | = | 11.49 me/l | |
| CO3 | = | 0.42 me/l | |
| HCO3 | = | 3.66 me/l | |
| Sum | = | 4.08 me/l | |
| ECw | = | 1.15 dS/m | |
Explanation: 1. The Sodium Adsorption Ratio (SAR) can be calculated from equation (1):
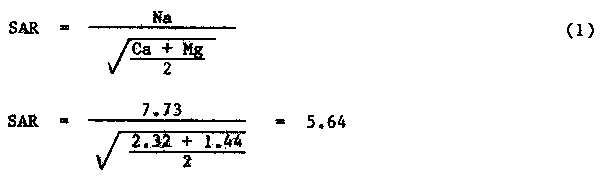
2. The adjusted Sodium Adsorption Ratio (adj SAR) can be calculated from the procedure given in Ayers and Westcot (1976): adj SAR = SAR [1 + (8.4 - pHc)] (15)
| where pHc | = | (pk2 - pkc) | + | p (Ca + Mg) + p (Alk) |
| (pk2 - pkc | = | 2.3 | ||
| p (Ca + Mg) | = | 2.3 | ||
| p (Alk) | = | 2.4 | ||
| pHc | = | 7.4 |
adj SAR = 5.64 [1 + (8.4 - 7.40] = 11.3
3. The adjusted Sodium Adsorption Ratio (adj RNa) can be calculated from equation (14) and Table 11):

| ECw | = | 1.15 dS/m |
| HCO3/Ca | = | 1.76 |
From Table 11, Cax = 1.43 me/l
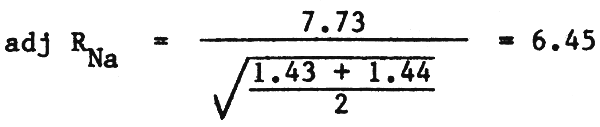
Irrigating to fill the soil-water reservoir for later withdrawal by the crop is difficult when infiltration rates are low, but there is little need to take action to increase infiltration unless the crop water demand or the leaching requirement cannot be met. Water ponding for too long a time frequently gives rise to secondary problems which are as important in causing yield loss as is an actual water shortage and often determine remedial measures to correct the infiltration problem. Some of the more troublesome secondary problems are crusting of seed beds, excessive weed growth and surface saturation which can cause root rot, diseases, nutritional disorders, poor aeration and poor germination. In some cases, water ponding for an excessive period of time has caused mosquito problems.
The management steps available to help maintain yields can be either chemical or physical. Chemical practices involve changing the soil or water chemistry that influences soil infiltration rates. This is normally accomplished by adding a chemical amendment, such as gypsum, to either the soil or the water or, in a few cases, by blending two or more sources of water to reduce the potential hazard. Physical methods include cultural practices that can be expected to improve or maintain infiltration rates during periods of irrigation or rainfall. Whether the physical or chemical approach is used, local conditions play an important role. A reduced infiltration caused by water quality is a different problem to a low infiltration rate caused by a clayey or compacted surface soil. Infiltration problems due to water quality are related to the impurities (Ca, Mg, Na, HCO3 and ECw) present in the water supply. Several possible options to solve a water quality-related infiltration problem are discussed in the following paragraphs. Each must be adapted to the local conditions and thoroughly field tested before any large-scale implementation.
The following management steps are directed at evaluating and overcoming infiltration problems caused by the chemical quality of the irrigation supply. Of equal importance, and also a water quality problem, is the reduction in infiltration that can take place due to a high sediment content in the supply water. It is beyond the scope of this publication to include this factor but it should be considered. See Section 8.17 for an example of the impacts from sediment.
Certain chemical amendments added to soil or water should improve a low infiltration rate caused by low salinity or by excessive sodium (high SAR) in the irrigation water. Improvement can be expected if the amendment increases the soluble calcium content or causes a significant increase in the salinity (ECw) of the applied water. Amendments are used to help increase the infiltration or counter the effects of sodium, since, at present, there is no economical process available for removing salts or sodium from irrigation water which is low enough in cost for general agricultural use. An amendment, such as gypsum, when added to soil or water will increase the calcium concentration in the water, thus reducing the sodium to calcium ratio and the SAR. Adding gypsum is also beneficial because it increases the salinity of low salt waters, thus improving infiltration (Figure 21). Gypsum of other similar additives will not cause any important improvement if poor infiltration is due to adverse soil texture, soil compaction, restrictive claypan or hardpan layers, or a high water table.
Most soil and water amendments in common use supply calcium directly (gypsum) or indirectly through an acid or acid-forming substance (sulphuric acid or sulphur) which reacts with soil lime (CaCO3) to release calcium to the soil solution. Acid or acid-forming amendments are not effective if lime is absent from the soil. Chemical amendments are expensive and add to the cost of crop production. They are justified only if their use results in a substantial improvement that can be evaluated in relation to cost. Field trials should be conducted to determine whether water or soil amendments improve water penetration or yield to an extent that justifies the cost. A crop receiving adequate water and producing near maximum yield would not be expected to show a further yield increase from the use of amendments, but, in some instances, such amendments may make irrigation management easier, though at an increased cost for the amendments, their handling and application.
Water amendments are most effective if the water infiltration problem is caused by a low salinity water (ECw < 0.2 dS/m) or by high SAR in a water of low to moderate salinity (ECw < 1.0 dS/m). If water salinity is moderate to high (ECw > 1.0 dS/m) in addition to a high SAR, soil applied amendments such as low-grade gypsum or sulphur may be preferred and often are more effective.
i. Gypsum
Gypsum can be either a soil or water amendment and is the most commonly used and widely available amendment for both. For reclamation of sodic soils, gypsum, in granular form, is applied broadcast at rates ranging from 5 to 40 t/ha and is worked into the soil. The 40 t/ha rate is used as a one-time application for extremely sodic soils and if rapid reclamation is needed. Annual rates of application in excess of 10 t/ha are usually uneconomical. High rates over 10 t/ha have normally been for immediate soil reclamation to allow roots to establish a proper rooting depth.
A water infiltration problem caused by low ECw or high SAR occurs primarily in the upper few centimetres of soil; therefore, the low application rates of gypsum to correct the surface problem are more effective if left on the soil surface or mixed with soil to a shallow depth rather than incorporated deeper into the soil as for reclamation. However, surface applied gypsum may be rapidly leached and the soil will again show the infiltration problem even though the gypsum may still be present a few centimetres below the soil surface. Small but repeated soil applications may be more effective for water-related surface infiltration problems, whereas single, large applications are more effective for sodic soil reclamation.
The application of gypsum to irrigation water to solve a water-related infiltration problem usually requires less gypsum per hectare than does a soil application. Gypsum is particularly effective when added to water if the water salinity is low (EC less than 0.5 dS/m). It is much less effective for higher salinity water because of the difficulty in applying and getting sufficient calcium into solution to counter the sodium present effectively. In practice it is unusual to get more than 1 to 4 me/l dissolved Ca in the usual fast-moving irrigation stream. These relatively small amounts of calcium in a low salinity water may increase infiltration by as much as 100-300 percent - a significant increase. However, if water salinity is relatively high, these small amounts of calcium are much less effective and change the infiltration rate to a much lesser degree.
The rate at which gypsum goes into solution will depend to a great extent upon the surface area or fineness of the grind. Finely-ground gypsum (less than 0.25 mm in diameter) dissolves much more rapidly. Therefore, the finely-ground, usually purer grades of gypsum are generally more satisfactory for water applications; the biggest drawback is the higher cost which often prevents small farmers from maintaining a continuous supply. The coarse grinds and lower grades are more satisfactory for soil application, but with care and ingenuity farmers have successfully used low grades for water amendments. Even though the finely-ground gypsum is much more costly per unit than is the coarse and lower grade, for water application the ease of handling and speed of dissolution often make it worth the added cost. Example 7 illustrates how gypsum can be used as a water amendment to improve infiltration.
EXAMPLE 7 - USE OF GYPSUM AS AN AMENDMENT
A low salinity water (ECw = 0.15 dS/m) is being used for irrigation of citrus. Infiltration problems have been experienced in the past causing oxygen stress in the citrus trees. The cause has been attributed to water ponding on the soil surface for extended periods of time. Since the critical time of fruit set is taking place, it was decided to add gypsum to the irrigation water to increase infiltration and reduce waterlogging and oxygen stress. A 5 hectare area needs an irrigation depth of 100 mm. The gypsum available is 70 percent pure and an increase of 2 me/l of calcium is desired in the water. How much gypsum should be used?
| Given: | ECw | = | 0.15 dS/m |
| Area | = | 5 ha | |
| Gypsum | = | 70 percent pure |
Total water requirement = 500 hectare mm = 5000 m3
1 milliequivalent per litre of calcium = 86 kg of 100% gypsum
per 1000 m3 of water
| Explanation: | The amount of 100 percent gypsum needed to supply 2 me/l of Ca in 5000 m3 of water can be found by: | |
| 1. | 1 me/l (Ca) = 86 kg (100% gypsum)/1000 m3 | |
| 2. | For 1 me/l (Ca) in 5000 m3 | |
| 1 me/l (Ca) = 5 × 86 = 430 kg of 100% gypsum | ||
| 3. | For 2 me/l (Ca) in 5000 m3 | |
| 2 me/l (Ca) = 430 kg × 2 = 860 kg of 100% gypsum | ||
| 4. | Since the gypsum is only 70% pure, the amount of gypsum needed is found by (860 × 100) ÷ 70 = 1230 kg of 70% pure gypsum | |
| A finely ground gypsum is best for water applications. Therefore the total quality of gyspum needed to supply 2 me/l of calcium in the 5000 m3 of water is 1230 kg of 70% pure gypsum. | ||
In a few instances, large pieces of rock gypsum have been placed in the irrigation ditch to supply calcium to the irrigation stream. The amount of calcium dissolving from the rock is low, so effectiveness depends upon the stream velocity and volume. The amount being dissolved can be determined by comparing the calcium concentration of upstream water with the concentration downstream. Its probable effectiveness can then be estimated by the changes in ECw and SAR brought about by the additional calcium and the potential change in infiltration as predicted by the guidelines of Table 1. Rock gypsum placed in the irrigation channel may increase maintenance costs as weed control and watercourse maintenance become more difficult because the gypsum will have to be removed during mechanical cleaning or dredging.
The ultimate goal of either water or soil amendment with gypsum is an increase in yield or a substantial increase in ease of irrigation management. An effective treatment should improve the water infiltration rate but the improvement must be weighed against the costs to determine whether the treatment is worth-while.
Gypsum occurs naturally in many soils in arid climates and some soils will contain gypsum in sufficient quantity to affect interpretations of both soil salinity (ECe) and sodicity (exchangeable sodium), and require a correction both to the measured soil salinity (ECe) and to the reported SAR which is frequently used to estimate the soil exchangeable sodium percent (ESP) (see Figure 1). The ECe procedure involves a saturated soil paste and, if gypsum is present, the ECe will include salinity attributable to the dissolution of gypsum - about 2 dS/m. Since gypsum is generally beneficial to most soils and detrimental to very few crops (citrus), the additional soil salinity due to gypsum may be subtracted from the measured ECe to give a more correct assessment of the soil salinity hazard. For example, a gypsiferous soil has a measured ECe of 6 dS/m, a soil salinity which is expected to reduce yields of many salt sensitive crops. Since 2 dS/m of the reported ECe can be attributed to the gypsum, the ECe safely can be discounted by 2 dS/m and the corrected ECe now becomes ECe = 4 dS/m, an amount much less hazardous to sensitive crops.
Naturally occurring soil gypsum also has a bearing on interpretation of many laboratory analyses of soils. In soil analysis, the laboratory sometimes reports the SAR of the saturation extract (ESP), as shown in Figure 1. This is not a correct evaluation if gypsum is present because all the sodium salts are completely soluble whereas the gypsum is only slightly soluble and can contribute a maximum of about 20 to 30 me/l calcium to the saturation extract. As an example of the problem in interpretation, a strongly gypsiferous soil, but with high salinity, may have an ECe of 12 dS/m, of which 2 dS/m can be attributed to the gypsum. If all the other salts are sodium, there should be, in the saturation extract, Na = 100 me/l and Ca not more than 30 me/l, yielding a calculated SAR of 26. Such a soil, having ECe = 12 dS/m and SAR of the saturation extract equal to 26, is normally classified as a saline-alkali soil which requires extensive reclamation by a massive gypsum application plus extensive leaching before cropping. This is an incorrect interpretation. The soil is moderately saline (ECe = 12 dS/m) but it is not sodic because the gypsum provides a steady supply of calcium. Even without leaching, it should be capable of growing excellent barley (tolerance of barley = 10 dS/m at 90 percent yield potential) and with 50 percent reduction in salinity (to ECe = 7 dS/m including 2 dS/m per cm attributed to the naturally occuring gypsum in the soil), it could be planted to field crops such as barley, cotton, sugarbeets, grain sorghum, wheat and soybeans without a loss in yield caused by salinity. The soil is not sodic and does not require soil amendments, but it does need leaching to widen the range of crop adaptability. Such soils are sometimes called “self reclaiming”, meaning that leaching alone will reclaim them and soil amendments are not needed.
A good rule of thumb to prevent such all-too-frequent interpretive errors has been adopted by the University of California Cooperative Extension Laboratories and is as follows: if the SAR of the saturation extract exceeds SAR = 10, confirmation of the indicated sodium problem is required by the laboratory. Confirmation is by the Schoonover Gypsum Requirement test given as method 22d in the USDA Handbook 60 (Richards 1954) or by the Exchangeable Cation Method given as methods 18 and 20a in the same handbook. These methods correct for the soluble cations attributable to salinity and estimate SAR and ESP more correctly. Where appropriate, the Schoonover method is simple and reliable, but it is not appropriate if appreciable exchangeable potassium is present.
Gypsum is sometimes present in irrigation water. If the soluble salts in the irrigation water include appreciable calcium, many sodic soils can be reclaimed over a period of one to five or more years simply by planting tolerant crops and adopting cultural practices to promote deep percolation of applied irrigation water. To reclaim a severely sodic soil in one year may require up to 40 t/ha of gypsum and extensive leaching to remove sodium (salts) released during reclamation. To reclaim the same soil relying upon calcium present in the irrigation water (Ca = 2–3 me/l or more) plus cultural practices (disking, ploughing, deep cultivation) and planting sodium-tolerant crops (pasture grasses and forage or similar), may take several years. Success or failure will depend to a great extent upon an adequate rate of infiltration and the depth of water that enters the soil, the calcium content of the irrigation water and the severity of the sodic problem. Deep cultivation will greatly enhance infiltration and speed reclamation whether amendments are used or not.
ii. Acid-forming amendments
Acids or acid-forming amendments also supply calcium to soils, but lime (CaCO3) must be present in the soil for them to be effective. Sulphur and sulphuric acid are both used extensively, but relatively few others have been used to any great extent. Table 12 gives comparative data for several common calcium supplying materials used for reclamation of sodic soil, but gypsum remains the most widely used because it is usually readily available and costs less for the me/l of calcium supplied. Several fertilizers are acid residual and contribute calcium through their acidic reaction.
Sulphur furnishes calcium if lime is present in the soil and is an excellent amendment for reclamation of sodic soils. It is not a satisfactory amendment for water application and is not very effective to improve a water infiltration problem. It is slow to react. The sulphur must first be acted upon by soil bacteria and be oxidized to form sulphurous and sulphuric acid which then reacts with lime to release calcium. The oxidation process is rather slow and requires a warm, well-aerated moist soil for about 30 days or longer. If sufficient time is available, it has proved to be a good amendment for reclamation of sodic-calcare-ous soils, but is not expected to provide a satisfactory solution for a water infiltration problem because the oxidation process is too slow and calcium released near the surface is soon leached during irrigations.
Sulphuric acid is a strong, corrosive acid, used for direct application to the soil surface at full strength or added to irrigation water where it reduces the water concentration of bicarbonate and contributes acidity to the soil surface to release calcium. It is very effective for reclaiming sodic soils and to improve water infiltration of limey soils because the sulphuric acid does not have to go through an oxidation process. It reacts rapidly with soil lime. Soil applications are made before cropping and are usually followed by extensive leaching to remove any excessive soluble salts present or formed because of the sulphuric acid reaction with lime and the soil. Applications in water must be carefully controlled and monitored to ensure that they are safe for the conditions of use - safe for pipelines, sprinklers, irrigation water distribution systems, and personnel. The ultimate effect on infiltration is about the same as that for a chemically equivalent amount of gypsum (Table 12). Sulphuric acid is highly corrosive and dangerous to handle. It may damage concrete pipelines, steel culverts, checkgates and aluminium pipes. It should only be applied by experienced operators.
| Amendment | Tons equivalent to 1 ton of 100 percent gypsum2 |
|---|---|
| Gypsum (CaSO4 . 2H2O)* | 1.00 |
| Sulphur (S)** | 0.19 |
| Sulphuric acid (H2SO4)* | 0.61 |
| Ferric sulphate (Fe2(SO4)3 . 9H2O)** | 1.09 |
| Lime sulphur (9 percent Ca + 24 percent S)* | 0.78 |
| Calcium chloride (CaCl2 . 2H2O)* | 0.86 |
| Calcium nitrate (Ca(NO3)2 . 2H2O)* | 1.06 |
| Calcium carbonate3 (CaCO3)** | 0.58 |
* Suitable for use as a water or soil amendment
** Suitable only for soil application
1 Adapted from Fireman and Branson (1965).
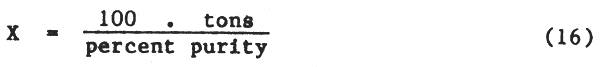
Example: If gypsum is 50 percent pure, X = 2.00 tons. This says that2.00 tons of 50 percent pure gypsum is equivalent to 1 ton of 100 percent pure gypsum.
Other amendments are sometimes used in local areas but their use depends greatly on the cost of supply and application. As shown in Table 13, several common fertilizers can also act as direct or indirect sources of calcium. Most acid fertilizers must go through an oxidation process similar to that for sulphur, and a source of calcium must be present in the soil (CaCO3). Therefore, they are of limited value for a water infiltration problem, but may be useful to prevent or delay formation of a sodic soil that may gradually develop as a result of using a slightly marginal sodic water.
| Fertilizer materials | Chemical Formula | Total Nitrogen (N) | Available Phosphoric Acid (P2O5) | Water Soluble Potash (K2O) | Combined Calcium (Ca) | Combined Sulphur (S) | Equivalent2 Acid or Base | |
|---|---|---|---|---|---|---|---|---|
| in kg Acid | Caco3 Base | |||||||
| Percent | ||||||||
| Nitrogen materials | ||||||||
| Ammonium nitrate | NH4NO3 | 33.5–34 | 62 | |||||
| Ammonium nitrate-sulphate | NH4NO3.(NH4)2SO4 | 30 | 6.5 | 68 | ||||
| Monoammonium phosphate | NH4H2PO4 | 11 | 48 | 58 | ||||
| Ammonium phosphate-sulphate | NH4H2PO4.(NH4)2SO4 | 13 | 39 | 7 | 69 | |||
| Ammonium phosphate-sulphate | NH4H2PO4.(NH4)2SO4 | 16 | 20 | 15 | 88 | |||
| Ammonium phosphate-nitrate | NH4H2PO4.NH4NO3 | 27 | 12 | 4.5 | 75 | |||
| Diammonium phosphate | (NH4)2HPO4 | 16–18 | 46–48 | 70 | ||||
| Ammonium sulphate | (NH4)2SO4 | 21 | 24 | 110 | ||||
| Anhydrous ammonia | NH3 | 82 | 147 | |||||
| Aqua ammonia | NH4OH | 20 | 36 | |||||
| Calcium ammonium nitrate solution | Ca(NO3)2.NH4NO3 | 17 | 8.8 | 9 | ||||
| Calcium nitrate | Ca(NO3)2 | 15.5 | 21 | 20 | ||||
| Calcium cyanamide | CaCN2 | 20–22 | 37 | 63 | ||||
| Sodium nitrate | NaNO3 | 16 | 29 | |||||
| Urea | CO(NH2)2 | 45–46 | 71 | |||||
| Urea formaldehyde3 | 38 | 60 | ||||||
| Urea ammonium nitrate solution | NH4NO3.CO(NH2)2 | 32 | 57 | |||||
| Phosphate materials | ||||||||
| Single superphosphate | Ca(H2PO4)2 | 18–20 | 18–21 | 12 | neutral | |||
| Triple superphosphate | Ca(H2PO4)2 | 45–46 | 12–14 | 1 | neutral | |||
| Phosphoric acid | H3PO4 | 52–54 | 110 | |||||
| Superphosphoric acid4 | 76–83 | 160 | ||||||
| Potash materials | ||||||||
| Potassium chloride | KCl | 60–62 | neutral | |||||
| Potassium nitrate | KNO3 | 13 | 44 | 23 | ||||
| Potassium sulphate | K2SO4 | 50–53 | 18 | neutral | ||||
| Sulphate of potashmagnesia | K2SO4.2MgSO4 | 26 | 1 | 15 | neutral | |||
1 From Soil Improvement Committee (1975).
2 Equivalent per 100 kg of each material.
3 Also known as ureaform, reaction product of urea and formaldehyde.
4 H3PO4, H4P2O7, H6P4O13 and other higher forms.
As shown in Table 1, an SAR of 12 or greater may appreciably reduce the rate of infiltration of water with a salinity less than ECw of 2.9 dS/m, and an SAR as low as 6 may appreciably reduce the infiltration rate of water with a salinity less than ECw of 1.2 ds/m. The infiltration rate can be increased either by increasing the water salinity or reducing the SAR.
Dilution reduces the SAR. This is due to the nature of the SAR equation (1). The numerator (Na) is reduced in proportion to the dilution and at a greater rate than is the denominator (Ca + Mg) because the denominator is reduced by the square root of the dilution. Example 8 shows how the SAR of a blended water is reduced when a tubewell water in Pakistan is blended into the normal canal supplies. Without blending the tubewell water would have very limited use, but as a result of blending the total amount of usable water has increased by the amount supplied by the tubewell.
EXAMPLE 8 - BLENDING IRRIGATION WATER TO REDUCE THE SAR OF A POOR QUALITY SUPPLY
A canal water supply is available but will not meet the total crop water demand. The canal supply could be blended with a poorer quality well water to the extent of 75% canal water and 25% well water. What is the SAR of the blended water?
| Given: | The water analysis is: | ||||||
| ECw | Ca | Mg | Na | HCO3 | SAR | ||
| (ds/m) | (me/l) | (me/l) | (me/l) | (me/l) | |||
| Canal water | 0.23 | 1.41 | 0.54 | 0.48 | 1.8 | 0.5 | |
| Well water | 3.60 | 2.52 | 4.00 | 32.0 | 4.5 | 18.0 | |
| Explanation: | The resulting blend quality can be found by using equation (13): | ||||||
| (me/l of (a) x proportion of (a) used) +(me/l of (b) x proportion of (b) used) =resulting blend in me/l | |||||||
| Ca = (1.41×0.75) + (2.52×0.25) = 1.69 me/l (blend) | |||||||
| Mg = (0.54×0.75) + (4.00×0.25) = 1.41 me/l (blend) | |||||||
| Na = (0.48×0.75) + (32.0×0.25) = 8.36 me/l (blend) | |||||||
| HCO3 = (1.8×0.75) + (4.5×0.25) = 2.48 me/l (blend) | |||||||
| EC = (0.23×0.75) + (3.6×0.25) = 1.07 dS/m (blend) | |||||||
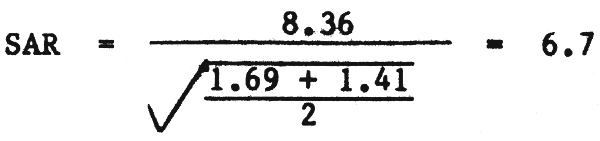 | |||||||
Many high SAR waters are usually abandoned if an alternate better quality source is made available. If the better supply is adequate for the area to be irrigated, there is nothing to be gained from blending the two supplies. However, if the better quality supply is not adequate for the land available for planting, blending a less desirable water with a better supply may allow more land to be planted, resulting in greater overall crop production and more income for the farmer.
Blending water supplies is not a common practice even when two supplies are available, one of which is much poorer in quality. Normally a better quality surface supply is used whenever available and the poorer quality groundwater is used whenever the surface supply is insufficient. Alternating supplies does not, however, offset an infiltration problem caused by the high SAR of a poorer quality supply. In fact, the problem can be greatly aggravated if low salinity-low SAR supplies are used after a highly saline-high SAR water. The high SAR water causes a correspondingly high ESP in the surface soil and, if low salinity water is then used, it can soon cause an appreciably reduced infiltration rate. An even more severe problem occurs when rain falls after using a saline or high SAR irrigation water. A light application of surface applied gypsum (1 to 2 t/ha) prior to irrigation with the better quality supply or before the rainy season is sometimes used in an attempt to overcome this problem. Blending would also prevent many of the secondary problems caused by short-term usage of high SAR water, such as surface crusting and sealing. Wherever possible high SAR water should be diluted to reduce infiltration problems, but in those instances where its use is alternated, the use of supplemental amendments should be considered.
Soil and water amendments and blending change the chemical nature of the water while physical methods keep the soil open by mechanical means. The most common physical method is either cultivation or deep tillage. Both are effective but normally short-lived and are thus only temporary solutions to a water infiltration problem.
Cultivation is usually done for weed control or soil aeration rather than to improve water penetration. Where infiltration problems are severe, cultivation or tillage are helpful as they roughen the surface and slow the flow of water, increasing the time during which infiltration can take place. A rough, cloddy furrow or field improves infiltration during the first one or two irrigations, after which another cultivation may be needed. Cultivation equipment can often be modified to leave a rougher surface. Cultivation breaks up the crust in the upper few centimetres of soil to improve infiltration. A common practice in areas where a water infiltration problem has been caused by low salinity water is to cultivate before each irrigation or before every second irrigation. This roughens the soil and opens cracks and air spaces that greatly increase the surface area exposed for infiltration.
Deeper tillage (chiselling, subsoiling) can be expected to improve deep water penetration for only one or two irrigations since the soil surface soon reverts to its original condition but, although improvement is not permanent, this practice may temporarily allow sufficient water to enter to make an appreciable difference in stored water and in the crop yield. Deep tillage physically tears, shatters and rips the soil, and is done prior to planting or during periods of dormancy when root pruning or root disturbances of permanent crops are less disruptive. Deep tillage should only be performed when soils are dry enough to shatter and crack. If done wet, increased compaction, aeration and permeability problems can be expected.
Crop residues or other organic matter left in the field will improve water penetration and is becoming a more widely accepted practice. It is one of the easiest methods to improve water infiltration, especially for small farmers who do not have the resources to implement more costly corrective measures. Unfortunately, in many instances, the small farmers use crop residues for other purposes and little, if any, is returned to the soil.
Crop residues left on the soil or worked into a rough cloddy soil surface will improve water penetration on sodic soils and will also improve water penetration into soils being irrigated by high SAR or low salinity water. Both crop residue left on the soil surface as well as the root system of the crop help in keeping the soil open. The benefits decline with time until replenished at the next cropping season.
The more fibrous and less easily decomposed crop residues, as from barley, rice, wheat, maize and sorghum, have improved water penetration, whereas residues from legumes and vegetable crops generally have not. The best residues are those which do not decompose or break down rapidly. These keep the soil porous by maintaining open channels and voids which improve water penetration. To be effective, relatively large quantities of residues are needed; for instance, manure has been used at rates of 40 to 400 metric tons per hectare to improve water infiltration. An organic application in the range of 10– 30 percent by soil volume in the upper 15 cm of soil may be needed to be effective.
Where water quality is affecting water infiltration, and organics are being tried to improve infiltration, it is important to incorporate the residues into the surface few centimetres of soil. Deeper incorporation is beneficial for soil structure and deeper percolation of applied water, but for infiltration problems caused by water quality, it is the surface soil that usually controls the depth of water entering the soil in a given period of time.
Rice hulls, sawdust, shredded bark, and many other waste products have been used in large volumes but with varying degrees of success. Tests with rice hulls in India increased the yield of rice in the first cropping season but yields reverted to their original level when the treatment was discontinued. From a long-term standpoint, the return of organic matter to the soil helps maintain soil structure and returns needed nutrients, but using a high rate of organic matter also causes problems. These include nutritional upsets, salinity effects caused by salty manure, nitrogen shortages or excesses owing to the use of certain types of materials (manures vs sawdust) and toxicities (chloride and potassium toxicities from rice hulls).
Physical and chemical methods in combination have proved to be the most effective approaches to solving water infiltration problems. However, these require extensive and continuing annual investment in both time and money to be effective. Many users try to complement these methods with irrigation practices to make the water infiltration problem easier to solve or manage. Several practices are discussed here.
Irrigating more frequently is a simple and effective approach especially for soils having an initially high infiltration rate but for which the rate drops rather quickly due to low salinity or high SAR. The objective is to supply the crop with adequate water at all times without secondary problems developing (waterlogging, poor aeration).
Irrigating more frequently maintains a higher average soil water content and reduces the possibility of a water stress that might result if irrigations were spaced further apart. If the crop is not stressed for water between irrigations, increasing the irrigation frequency does little good.
Pre-plant irrigation can be relied upon to fill the rooting depth to field capacity at a time when there is little chance of causing crop damage. In some difficult soils a pre-plant irrigation is the only opportunity to wet the deeper part of the crop root zone. It is also an effective method for wetting soils with a very slow infiltration rate.
Extending the duration of an irrigation applies more water and is beneficial provided that soil aeration, waterlogging, runoff and surface drainage problems do not result. Many irrigators try to extend the irrigation by reducing the volume of flow to a field and holding the water on the field for a greater period of time. Careful management and monitoring is needed to maintain water use efficiency and to keep runoff to a minimum. Excessive runoff is frequently collected in a pond at the low side of the irrigated field and is pumped back up slope through a pipeline to be re-circulated into the irrigation stream. These recirculation (return-flow) systems are becoming common in surface irrigated areas and can aid greatly in efficiently irrigating a soil with a low infiltration rate. In a few instances this system is installed following a comprehensive land levelling or grading programme to improve water use efficiency. By collecting and re-circulating water, both the total water use efficiency and depth of penetration can be more easily controlled.
Changing irrigation systems may be necessary on more difficult soils. For instance, changing from a surface irrigation system to one which applies water more precisely (sprinklers for sandy soils and localized (drip) irrigation for heavier clayey soils) may allow the user to approach the soil intake rate more closely. These changes require large capital expenditures and additional power to operate, but the system can be designed to apply water at the rate desired. If runoff occurs with sprinklers or localized (drip) irrigation, the application rate is too high. Changing the rate of application after installation may be difficult and complete redesigning of the system may be needed. In some cases an existing sprinkler or localized irrigation system can be intermittently operated to match the infiltration rate more closely, stopping irrigation at the time runoff begins and re-irrigating every few hours until the desired depth of applied water is reached. This technique does allow the use of an existing sprinkler or localized irrigation system, but will probably use a little more water, thus increasing production cost, and it may also need more investment in equipment to offset idle time.
Sprinklers apply water in droplets, some quite large. On impact, these large droplets can disperse the soil surface particles and aggravate or cause an infiltration problem accompanied by excessive runoff. Application rates normally vary from 3 mm to 6 mm per hour over the irrigated area. Sprinklers are well adapted to sandy and loamy soils but less so to heavy or clayey type soils. Localized drip or trickle irrigation systems are better adapted to loamy or clayey soils and apply water through many small outlets (emitters) at a rate of 2 to 4 litres per hour. At these low rates they do not disperse the soil particles as do sprinklers. They are less well adapted to sandy soils.