
PESTE DES PETITS
RUMINANTS
A field manual

 Food and Agriculture
Food and Agriculture
Organization
of the United Nations
 |
RECOGNIZING PESTE DES PETITS RUMINANTS A field manual |

|
Table of contents |
The designations employed and the presentation of material in this publication do not imply the expression of any opinion whatsoever on the part of the Food and Agriculture Organization of the United Nations concerning the legal status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. All rights reserved. No part of this publication may
be reproduced, stored in a retrieval system, or transmitted in any form or by any
means, electronic, mechanical, photocopying or otherwise, without the prior
permission of the copyright owner. Applications for such permission, with a statement
of the purpose and extent of the reproduction, should be addressed to the Director,
Information Division, Food and Agriculture Organization of the United Nations, Viale
delle Terme di Caracalla, 00100 Rome, Italy. |
This manual was prepared by Dr P.L. Roeder and Prof. T.U. Obi of the FAO
Emergency Prevention System for Transboundary Animal and Plant Pests and
Diseases (EMPRES) (Livestock) Unit. Drs W. Taylor (consultant) and A. Diallo
of the International Cooperation Centre on Agrarian Research and Development,
Department of Breeding and Tropical Veterninary Medicine (CIRAD-EMVT) kindly
commented on the text during drafting; their contributions are gratefully
acknowledged.
FAO wishes to acknowledge the financial assistance to the production of this
first edition provided by the International Fund for Agricultural Development
(IFAD) through FAO's Regional Animal Disease Surveillance and Control Network
(RADISCON) project.
The colour plates appear by kind permission of the
following: Front cover and Plate 5 Prof. T.U. Obi Plates 1, 4, 7, 8 and 11
Dr P.L. Roeder Plates 2, 3, 6 and 10 Dr W.P. Taylor Plate 9 Dr P.C. Lefèvre
Plate 12 Commonwealth Scientific and Industrial Resarch Organization (CSIRO),
Australia.
This booklet is one of a series prepared by FAO's Emergency System for
Transboundary Animal Plants and Pests (EMPRES) (Livestock) Unit as an aid
to emergency preparedness for the major transboundary diseases of livestock.
Peste des petits ruminants (PPR), which is also known as goat plague, is
a disease of increasing importance in Africa and Asia wherever small ruminants
form an important component of agricultural food production. It can affect
a broad range of species, including some antelopes, as has already been seen
in zoological garden collections but, fortunately, not in the wild. The disease
was once thought to be a comparatively restricted problem in West Africa,
but it is now known to extend throughout most of West, Central and East Africa,
reaching eastwards through western and South Asia. Undoubtedly much of this
increased recognition is owing to greater awareness and the availability
of new laboratory diagnostic tests. However, it is possible that the disease
has actually spread, rather than just being more frequently recognized. Dramatic
PPR events in the last decade in Asia and East Africa suggest that the disease
may be growing in severity in newly invaded areas. Many of the other regions
where small ruminant production is a significant contributor to food security
are close to the PPR-affected area and there is a serious risk that the disease
will spread to them, especially in southern Africa and the Central Asian
republics.
Early warning is the key to early reaction for containment, control and rapid elimination. |
PPR may have passed unrecognized for years in some countries because it is frequently confused with other diseases that cause respiratory problems and mortality of small ruminants. Many veterinarians, animal health workers and livestock owners in areas where PPR is absent or recently introduced are not familiar with its clinical and pathological features. The manual has been prepared to help them recognize this transboundary disease as it emerges and evolves. PPR is an important disease in its own right, but it is also very important that it be differentiated from rinderpest, particularly as the Global Rinderpest Eradication Programme (GREP) proceeds to the anticipated eradication of rinderpest by the year 2010. The aim of this manual is to make all concerned with the health of small ruminants "think PPR" and recognize it rapidly when it occurs. Comments and suggestions for improvement are welcomed for use in preparing subsequent editions.
For details on this and other publications, and to obtain additional copies
contact:
EMPRES (Livestock) Animal Health Service FAO
Animal Production and Health Division
Viale delle Terme di Caracalla
00100 Rome, Italy
Tel. +39 06 57054798/6772 Fax +39 06 57053023
E-mail [email protected]
EMPRES home page:
www.fao.org/empres
Members of the Regional Animal Disease Surveillance and Control Network
(RADISCON) may also obtain copies through the project by sending a message
to the RADISCON Coordinating Unit in FAO, Rome: Fax +39 06 57053500
E-mail:
[email protected]
Peste des petits ruminants (PPR) is a highly contagious and infectious viral
disease of domestic and wild small ruminants. It was first described in
Côte d'Ivoire in West Africa in 1942. Gradually it was realized that
several clinically similar diseases occurring in other parts of West Africa
shared the same cause - the virus now called peste des petits ruminants.
Investigators soon confirmed the existence of the disease in Nigeria, Senegal
and Ghana. For many years it was thought that it was restricted to that part
of the African continent until a disease of goats in the Sudan, which was
originally diagnosed as rinderpest in 1972, was confirmed to be PPR. The
true extent of the disease has only become apparent in recent years and is
still being clarified. The realization that many of the cases diagnosed as
rinderpest among small ruminants in India may, instead, have involved the
PPR virus, together with the emergence of the disease in other parts of western
and South Asia, point to its ever-increasing importance.
PPR is an important disease in its own right, but it has also created problems
because of its apparent similarity to rinderpest - the clinical signs of
PPR closely resemble those of rinderpest, making differential diagnosis
difficult. It should, however, be borne in mind that clinical disease caused
by rinderpest in small ruminants is a relatively rare event, even in Asia.
PPR is a severe, fast-spreading disease of mainly domestic small ruminants. It is characterized by the sudden onset of depression, fever, discharges from the eyes and nose, sores in the mouth, disturbed breathing and cough, foul-smelling diarrhoea and death.
The virus which causes PPR, the peste des petits ruminants virus (PPRV), belongs to the morbillivirus group of the paramyxovirus family of viruses. It is closely related to the rinderpest virus of cattle and buffaloes, the measles virus of humans, the distemper virus of dogs and some wild carnivores, and the morbilliviruses of aquatic mammals. To date, genetic characterization of PPR virus strains has allowed them to be organized into four groups; three from Africa and one from Asia. One of the African groups of PPRV is also found in Asia. The epidemiological significance of these groupings is less clear at present than that of rinderpest virus groupings.
Clinical disease is seen in sheep and goats and has been described in zoological garden collections of wild small ruminants including Laristan sheep, Dorcas-type gazelles, gemsbok and the Nubian ibex. Cattle, buffaloes, camels and pigs can become infected but there is little or no evidence of disease associated with their infection. Probable global distribution of PPR virus infection Note: Clinical disease has not been reported by every country; for some only serological evidence exists. Source: Based on official reports to the International Office of Epizootics (OIE); published reports; other official country reports; and reports of FAO Reference and Collaborating Centres (1999).
PPR infection has been recognized in many of the African countries that lie
between the Atlantic Ocean and the Red Sea. The affected area extends north
to Egypt and south to Kenya, in the east, and Gabon, in the west. PPR has
not been recognized in most of North and southern Africa. In some of the
countries where the disease has not been confirmed there are serological
and/or clinical indications that the infection is, nevertheless, present.
A recent (1998) serological survey in the United Republic of Tanzania did
not detect any antibodies to PPR suggesting that infection has not extended
that far south.
In recent years the disease has been seen in the Near East and the Arabian
Peninsula, in countries including the Islamic Republic of Iran, Iraq, Israel,
Jordan, Kuwait, Lebanon, Oman, Saudi Arabia, the United Arab Emirates and
Yemen, and there is serological evidence from the Syrian Arab Republic and
Turkey. Outbreaks of PPR are now known to be common in India, Nepal, Bangladesh,
Pakistan and Afghanistan.
Countries that have imported small ruminants from these areas are advised
to investigate thoroughly any disease syndrome characterized by disturbed
breathing, discharges from the eyes, nose and mouth, sores in the mouth and
diarrhoea in order to rule out PPR.
It is still not clear whether the apparent geographical spread of the disease
in the last 50 years is real or whether it reflects increased awareness,
wider availability of diagnostic tools or even a change in the nature of
the virus. It seems most likely that a combination of factors is responsible
for the present knowledge of its range and it is known that confusion of
PPR with pneumonic pasteurellosis and other pneumonic diseases of small ruminants
has delayed its recognition in some countries.
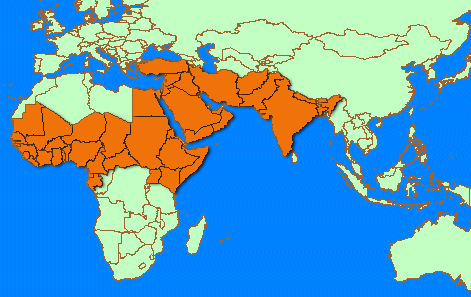
Probable global distribution Based on official reports to the OIE; published reports, other official coutry reports and reports of FAO Reference and Collaborating Centres |
The discharges from eyes, nose and mouth, as well as the loose faeces, contain
large amounts of the virus. Fine infective droplets are released into the
air from these secretions and excretions, particularly when affected animals
cough and sneeze. Other animals inhale the droplets and are likely to become
infected. Although close contact is the most important way of transmitting
the disease, it is suspected that infectious materials can also contaminate
water and feed troughs and bedding, turning them into additional sources
of infection. These particular hazards are, however, probably fairly short-term
since the PPR virus, like its close relative rinderpest, would not be expected
to survive for long outside the host.
In endemic areas, most of the sick and dying animals are over four months
and up to 18 to 24 months of age.
Clinical signs appear an average of two to six days after natural infection
with the virus (the incubation period). This is followed by the sudden onset
of fever with rectal temperature of at least 40° to 41oC. Affected animals
are markedly depressed and appear sleepy. Their hair stands erect giving
them a bloated appearance, especially the short-haired breeds. Soon after
this stage, a clear watery discharge starts to issue from the eyes, nose
and mouth, later becoming thick and yellow as a result of secondary bacterial
infection (Figure 1). The discharges wet the chin
and the hair below the eye; they tend to dry, causing matting together of
the eyelids, obstruction of the nose and difficulty in breathing.
The carcass of an affected animal is usually emaciated, the hindquarters
soiled with soft/watery faeces and the eyeballs sunken. The eyes and nose
contain dried-up discharges. The following changes may be seen:
Dirty-white, false membranes; erosions on the gums, soft and hard palates,
tongue and cheeks and into the oesophagus.
Swollen; erosions and possibly scabs or nodules in late cases.
Congested (reddened) lining; clear or creamy yellow exudates; erosions.
Dark red or purple areas; firm to the touch, mainly in the anterior and cardiac
lobes (evidence of pneumonia) (Figures 8 and 9).
Soft and swollen. Abomasum Congested (reddened) lining; haemorrhages.
Congested (reddened) lining; haemorrhages; some erosions.
Small red haemorrhages along the folds of the lining, joining together as
time passes and becoming darker, even green/black in stale carcasses (Figure
10).
PPR is frequently confused with other diseases that present fever and grossly
similar clinical signs, especially when it is newly introduced. When carrying
out an investigation, examination of the way the disease behaves in the herd
or flock is as important as the findings on a single goat or sheep. The most
frequent sources of confusion are:
Could be a symptom of: rinderpest, foot-and-mouth disease, bluetongue or
contagious ecthyma (orf or "sore mouth").
Could be a symptom of: pneumonic pasteurellosis or contagious caprine
pleuropneumonia (CCPP).
Could be a symptom of: coccidiosis or gastro-intestinal helminth infestations.
Pneumonia is usually a very obvious presenting sign in PPR so, without doubt,
pneumonic pasteurellosis and CCPP have caused the most difficulty in differential
diagnosis.
is a purely respiratory disease of sheep and goats caused by the bacterium
Pasteurella haemolytica. Dark red/purple areas, firm to the touch, are evident
mainly in the anterior and cardiac lobes of the lung
(Figure 9). There are no oral lesions or diarrhoea.
The numbers of affected and dead animals are usually lower than for PPR except
under exceptional conditions of stress and crowding such as can occur when
large numbers of sheep are assembled for trade. The main problem of
differentiation arises when oral lesions and diarrhoea are either absent
or not very obvious in PPR, as is sometimes the case. Using appropriate culture
media, Pasteurella haemolytica bacteria are easily isolated in pure and profuse
culture from pneumonic lungs of sheep, even from the lungs of PPR-affected
animals. Isolation of Pasteurella haemolytica bacteria from the lungs of
sheep, therefore, neither confirms a diagnosis of primary pneumonic
pasteurellosis nor rules out the presence of PPR. Diagnostic tests for detecting
PPRV should be carried out in all suspected cases of pneumonic pasteurellosis
where there is a risk of PPR.
is a disease of goats (sheep are not affected) caused by a Mycoplasma sp.
Like PPR, it is characterized by fever, difficult/abnormal breathing and
coughing, but there mouth lesions or diarrhoea are not present in CCPP. At
post mortem examination, the lung lesions in CCPP are more diffuse and a
fibrinous fluid is found in the chest cavity. Fibrin deposits cover the lungs
and are frequently connected to the chest wall by fibrinous strands
(Figure 11). In PPR high-risk areas it is advisable
to rule out PPR by laboratory testing of, at least, serum samples from
convalescent flocks, even if CCPP is suspected.
in small ruminants has been described primarily in Asia. Generally, this
disease occurs in small ruminants only when they are in contact with affected
cattle or buffaloes, so it is important during investigations to examine
all species. Confirmation requires the resources of a specialist laboratory
(see Sources of assistance). The samples required
for laboratory confirmation of both rinderpest and PPR are identical. As
the Global Rinderpest Eradication Programme (GREP) progresses, it becomes
increasingly important that PPR and rinderpest be differentiated because,
at this stage of the programme, any outbreak of rinderpest anywhere represents
an international emergency.
is more commonly seen in sheep than goats. The most important distinguishing
features of FMD, other than the appearance of the lesions, are the absence
of breathing problems and diarrhoea, and the presence of lameness (often
marked). Sudden death of very young lambs without other signs often occurs.
The oral lesions when present are often very small and difficult to see;
the mouth does not exude such a foul odour as in PPR. Bluetongue, like PPR,
is characterized by fever, discharges and oral lesions
(Figure 12). However, it differs from PPR in: the
presence of oedema of the head region; bluish discoloration of the oral cavity,
the coronary band of the hooves and the less hairy parts of the body; and
lameness.
virus infection is endemic throughout the regions of the world affected by
PPR. Clinical disease is, however, not generally experienced in indigenous
breeds in these countries, being mainly restricted to exotic introduced animals.
The presence of antibody to bluetongue viruses in single samples does not
confirm a provisional diagnosis of bluetongue.
is often confused with PPR because of the nodules and thick scabs sometimes
seen on the lips in the late stages of PPR. Confusion is especially likely
to arise in severe cases of orf where lesions extend into the mouth and nose.
In uncomplicated orf, there is usually no oral necrosis, diarrhoea or pneumonia.
The International Office of Epizootics (OIE) Manual of Standards for Diagnostic
Tests and Vaccines contains guidelines on the collection of samples and the
diagnostic techniques for diagnosis of PPRV infection. A provisional diagnosis
of PPR can be made from epidemiological and clinical features. A disease
characterized by discharges, diarrhoea, and deaths with breathing problems
in sheep and/or goats, but not in-contact cattle, with mainly adolescents
being affected and dying must arouse a suspicion of PPR. The observation
of characteristic post mortem changes would further strengthen the provisional
diagnosis.
Because of the necessity to detect PPR amid a number of other acute diseases
with grossly similar presenting signs, and to differentiate it from rinderpest,
some laboratory tests need to be carried out. These tests may detect the
virus itself, evidence of the presence of the virus (virus antigen or genetic
material) or antibodies against the virus found in blood serum.
The chances of a successful laboratory confirmation of the clinical diagnosis
increase as the numbers of samples examined and animals sampled increase.
There are several important points to observe when using the services of
a laboratory:
The samples required are:
Cotton buds or swabs of absorbent cotton wool are inserted into the conjunctival
sac and swirled around to collect tears. The bud/swab is broken off into
a container and about 150 microlitres of sterile phosphate-buffered saline
(PBS pH 7.2 to 7.6) are added (if available).
This material can be collected by a spatula or finger rubbed across the gum
and inside the upper and lower lips. The material collected is then scraped
into a container and 150 microlitres of PBS are added (if available).
It is recommended that the following tissues be collected during post mortem
examination: lymph nodes found around the lungs (mediastinal) and alimentary
tract (mesenteric); portions of the spleen and the lungs.
This is needed for virus isolation and should be collected in bottles containing
anticoagulants (heparin or ethylenediamine tetracetic acid [EDTA]).
These are needed for antibody detection.
Control of PPR outbreaks relies on movement control (quarantine) combined
with the use of focused ("ring") vaccination and prophylactic immunization
in high-risk populations. Until recently, the most practical vaccination
against PPR made use of tissue culture rinderpest vaccine. Recently, a homologous
PPR vaccine has been developed and the vaccine seed is available through
the Pan African Veterinary Vaccine Centre (PANVAC) at Debre Zeit, Ethiopia,
for Africa, or CIRAD-EMVT at Montpellier, France, for other areas. This vaccine
of choice is becoming increasingly available. The vaccines can protect small
ruminants against PPR for at least three years. The use of rinderpest vaccine to protect
small ruminants against PPR is now contraindicated because its use produces
antibodies to rinderpest which compromise serosurveillance for rinderpest,
and thereby the Global Rinderpest Eradication Programme.
Differentiating between rinderpest and PPR to obtain a definitive identification
of PPR can be difficult, especially when the disease is encountered for the
first time and national laboratories lack adequate facilities. Samples for
diagnostic confirmation can be submitted to either the FAO World Reference
Laboratory for Rinderpest at the Institute for Animal Health, Pirbight
Laboratory, United Kingdom or the FAO Collaborating Centre at the International
Cooperation Centre on Agrarian Research and Development, Department of Breeding
and Tropical Veterninary Medicine (CIRAD-EMVT) Laboratory, Montpellier, France,
which can assist with the diagnosis of PPR. Addresses are given below.
FIGURE 1:
FIGURE 2:
FIGURE 3:
FIGURE 4:
FIGURE 5:
FIGURE 6:
FIGURE 7:
FIGURE 8:
FIGURE 9:
FIGURE 10:
FIGURE 11:
FIGURE 12:
Trade in small ruminants, at markets where animals from different sources
are brought into close contact with one another, affords increased opportunities
for PPR transmission, as does the development of intensive fattening units.
Appearance of disease in a herd or flock
When PPR occurs in an area for the first time, it is possible that acute
high fever with extreme depression and death occur before any other typical
signs have been seen. A more typical picture, however, is that of a
fast-spreading syndrome in sheep and/or goats characterized by the sudden
onset of depression, discharges from eyes, nose and mouth, abnormal breathing
with coughing, diarrhoea and deaths. The outbreak will not involve cattle,
whether rinderpest vaccinated or not, even if they are in contact with affected
sheep and/or goats. Although both goats and sheep are susceptible to infection
and may show disease, they are not always affected simultaneously. For example,
in Africa PPR is seen most commonly in goats, while in western and South
Asia sheep are usually the most noticeable victims. The disease can, however,
strike both species with equally devastating consequences.
The appearance of clinical PPR may be associated with any of the following:
One to two days after fever has set in, the mucous membranes of the mouth
and eyes become very reddened (Figure 2). Then epithelial
necrosis causes small pin-point greyish areas to appear on the gums, dental
pad, palate, lips, inner aspects of the cheeks and upper surface of the tongue.
These areas increase in number and size and join together. The lining of
the mouth is changed in appearance. It becomes pale and coated with dying
cells (Figure 3) and, in some cases, the normal membrane
may be completely obscured by a thick cheesy material
(Figure 4). Underneath the dead surface cells there
are shallow erosions. In mild cases these changes may not be severe and will
require careful examination to be seen. Gentle rubbing across the gum and
palate with a finger may yield a foul-smelling material containing shreds
of epithelial tissue. Similar changes may also be seen in the mucous membranes
of the nose, the vulva and the vagina. The lips tend to swell and crack and
become covered with scabs (Figure 5).
As the disease progresses, a characteristic foul smell exudes from the mouth.
Affected animals resist attempts to open their mouths because of the pain.
Diarrhoea commonly appears about two to three days after the onset of fever
(Figure 6) although, in early or mild cases, it may
not be obvious. The faeces are initially soft and then watery, foul-smelling
and may contain blood streaks and pieces of dead gut tissue. Where diarrhoea
is not an obvious presenting sign, the insertion of a cotton wool swab into
the rectum may reveal evidence of soft faeces which may be stained with blood.
Affected animals breathe fast, sometimes so fast that they exhibit rocking
movements with both the chest and abdominal walls moving as the animal breathes.
Severely affected cases show difficult and noisy breathing marked by extension
of the head and neck, dilation of the nostrils, protrusion of the tongue
and soft painful coughs - they have obvious signs of pneumonia.
Such victims may eventually become dehydrated with sunken eyeballs, and death
often follows within seven to ten days from onset of the clinical reaction.
Other animals will recover after a protracted convalescence.
A common feature in later stages of the disease is the formation of small
nodular lesions in the skin on the outside of the lips around the muzzle
(Figure 7). The exact cause of these is not known
(possibly Dermatophilus infection or reactivation of a latent contagious
ecthyma infection - orf or "sore mouth") but they cause confusion because
of their similarity to the symptoms of primary contagious ecthyma or even
sheep/goat pox.
Up to 100 percent of the animals in a flock may be affected in a PPR outbreak
with between 20 and 90 percent dying. These proportions are usually lower
in endemic areas where older animals have survived earlier infection. Pregnant
animals may abort.
In summary, suspect PPR if you see any combination of:
Any appearance of one or more of these signs in combination must be considered
suspicious.
Mouth
Lips
Nasal cavity
Lungs
Lymph nodes (associated with the lungs and the intestines)
Small intestines
Large intestines (caecum, colon and rectum)
Mouth lesions
Difficult breathing
Diarrhoea
Pneumonic pasteurellosis
Contagious caprine pleuropneumonia (CCPP)
Rinderpest disease
Foot-and-mouth disease (FMD)
Bluetongue
Contagious ecthyma (orf, "sore mouth", contagious pustular dermatitis)
Laboratory confirmation
Detection of virus antigens by the agar gel immunodiffusion test (AGIDT)
is a relatively simple, fast and cheap process. It is extremely useful as
an initial test, but it does not discriminate between PPR and rinderpest
viruses and further tests are needed to do this. Histopathology combined
with immunohistochemical staining (e.g. immunoperoxidase) is a useful procedure
because it is performed on formalin-fixed material and can discriminate between
PPR and rinderpest when performed with specific monoclonal antibodies. Virus
antigens can also be detected by immunocapture ELISA (ICE) which is rapid
and sensitive, and differentiates between PPR and rinderpest. Standardized
reagent kits are commercially available for AGIDT and ICE.
Detection of virus genetic material is performed by the reverse transcriptase
polymerase chain reaction (RT PCR) which requires specialist facilities and
expertise. Despite its high cost, it is now one of the tests used most frequently
in reference centres, together with enzyme linked immunosorbent assay(ELISA),
because it is rapid, accurate, highly sensitive and can discriminate between
PPR and rinderpest. Combining this test with nucleotide sequencing provides
virus characterization information that is useful in epidemiological studies.
Detection of the virus is done by isolation of the PPR virus in cultured
cells. This method of diagnosis can be very valuable as it provides live
virus for biological characterization studies. If facilities are available,
it should always be attempted and isolated viruses stored for later studies.
Detection of antibodies for diagnosis requires the collection of two blood
samples, three weeks apart, from the same animals, which is not always feasible
in the field. Exceptionally, in a country that can be certain that it was
free from PPR, testing single samples taken late in the course of the disease
(at least a week after the appearance of clinical signs) can be diagnostic.
Surveys for antibodies are very useful to determine the presence or absence
of infection and its extent in a population. Competitive ELISA has now largely
replaced the virus neutralization test.
Samples required for laboratory testing
Tears
Gum debris
Tissues
Two sets of each tissue are required; one set is chilled but not frozen,
and the other is put in 10 percent formalin solution to preserve the samples.
Where cold storage is a problem, as is often the case, formalin can be used
to preserve the samples when they are sent to the laboratory.
Unclotted blood
Clotted blood or serum
National laboratories will provide guidance about exactly which samples are
required, but it is advisable to collect as many of the samples listed above
as possible when dealing with an outbreak.
It should be noted that submission of samples to any laboratory outside the
country of origin is always subject to prior agreement with the recipient
and transportation in containers meeting International Air Transport Association
(IATA) regulation standards. Detailed instructions for the collection and
dispatch of rinderpest samples (which are also applicable to PPR samples)
are contained in the publication Collection and submission of diagnostic
specimens to the FAO World Reference Laboratory for Rinderpest, which
can be obtained from FAO EMPRES; it can also be supplied electronically as
an attachment to e-mail or by fax on request.
FAO World Reference Laboratory for Rinderpest, Reference Laboratory for
PPR
Institute for Animal Health Pirbright Laboratory Ash Road Pirbright ,Woking,
Surrey GU24 0NF, United Kingdom,
Tel. +44 1483 232441 Fax + 44 1483 232448 E-mail
[email protected]
FAO Reference Laboratory for PPR
CIRAD-EMVT Campus international de Baillarguet Montferrier-sur-Lez BP 5034
34032 Montpellier Cedex 1 France
Tel. +33 4 67593705 Fax +33 4 67593798 E-mail
[email protected]
PPR in a goat: purulent eye and nose discharges
Discharges from the nose and eyes in advanced PPR infection; the hair below
the eyes is wet and there is matting together of the eyelids as well as partial
blockage of the nostrils by dried-up purulent discharges.
PPR in a goat: inflamed (reddened) eye membranes
Reddening of the mucous membranes of the eye (the conjunctiva) in the early
stages of infection. Note the purulent eye discharges.
PPR in a goat: early mouth lesions showing areas of dead cells
Early pale, grey areas of dead cells on the gums.
PPR in a goat: later mouth lesions
The membrane lining the mouth is completely obscured by a thick cheesy material;
shallow erosions are found underneath the dead surface cells.
PPR in a goat: swollen, eroded lips
The lips are swollen, oedematous and show areas of erosion.
PPR in a goat: signs of diarrhoea
The hindquarters are soiled with liquid faeces.
PPR in a goat: nodular lesions around the mouth
Such nodules are a common finding in the later stages of PPR infection.
PPR in a goat: the early lesions of pneumonia
Note the small, red, solid areas of lung tissue caused directly by PPR virus
infection.
PPR in a sheep: advanced pneumonia
Note the extensive, dark red/purple areas, firm to the touch, in the anterior
and cardiac lobes of the lungs. Although such pneumonia is commonly seen
in PPR, it is caused by secondary bacterial infection, most commonly Pasteurella
haemolytica. These lesions are typical of pneumonic pasteurellosis.
PPR in a goat: "zebra striping" in the large intestine
Note the lines of haemorrhage along the tips of the folds of the lining of
the caecum and colon. Later, the individual haemorrhages join up and, after
death, turn black.
Typical lesions of contagious caprine pleuropneumonia (CCPP) in a goat
Note the yellowish, fibrinous deposit on the surface of the lungs and adhesions
to the inside of the rib cage.
Bluetongue disease in a sheep
Note the bluish discoloration of the coronary bands of the hoof. The lips
will usually be found to be swollen and discoloured blue at the same time.