"Alginate" is the term usually used for the salts of alginic acid, but it can also refer to all the derivatives of alginic acid and alginic acid itself; in some publications the term "algin" is used instead of alginate. Alginate is present in the cell walls of brown algae as the calcium, magnesium and sodium salts of alginic acid. The goal of the extraction process is to obtain dry, powdered, sodium alginate. The calcium and magnesium salts do not dissolve in water; the sodium salt does. The rationale behind the extraction of alginate from the seaweed is to convert all the alginate salts to the sodium salt, dissolve this in water, and remove the seaweed residue by filtration. The alginate must then be recovered from the aqueous solution. The solution is very dilute and evaporation of the water is not economic. There are two different ways of recovering the alginate.
The first is to add acid, which causes alginic acid to form; this does not dissolve in water and the solid alginic acid is separated from the water. The alginic acid separates as a soft gel and some of the water must be removed from this. After this has been done, alcohol is added to the alginic acid, followed by sodium carbonate which converts the alginic acid into sodium alginate. The sodium alginate does not dissolve in the mixture of alcohol and water, so it can be separated from the mixture, dried and milled to an appropriate particle size that depends on its particular application.
The second way of recovering the sodium alginate from the initial extraction solution is to add a calcium salt. This causes calcium alginate to form with a fibrous texture; it does not dissolve in water and can be separated from it. The separated calcium alginate is suspended in water and acid is added to convert it into alginic acid. This fibrous alginic acid is easily separated, placed in a planetary type mixer with alcohol, and sodium carbonate is gradually added to the paste until all the alginic acid is converted to sodium alginate. The paste of sodium alginate is sometimes extruded into pellets that are then dried and milled.
These essentials of the process are illustrated in the flow diagram in Figure 32.
The process appears to be straightforward, certainly the chemistry is simple: convert the insoluble alginate salts in the seaweed into soluble sodium alginate; precipitate either alginic acid or calcium alginate from the extract solution of sodium alginate; convert either of these back to sodium alginate, this time in a mixture of alcohol and water, in which the sodium salt does not dissolve.
The difficulties lie in handling the materials encountered in the process, and to understand these problems a little more detail of the process is required.
To extract the alginate, the seaweed is broken into pieces and stirred with a hot solution of an alkali, usually sodium carbonate. Over a period of about two hours, the alginate dissolves as sodium alginate to give a very thick slurry. This slurry also contains the part of the seaweed that does not dissolve, mainly cellulose. This insoluble residue must be removed from the solution. The solution is too thick (viscous) to be filtered and must be diluted with a very large quantity of water. After dilution, the solution is forced through a filter cloth in a filter press. However, the pieces of undissolved residue are very fine and can quickly clog the filter cloth. Therefore, before filtration is started, a filter aid, such as diatomaceous earth, must be added; this holds most of the fine particles away from the surface of the filter cloth and facilitates filtration. However, filter aid is expensive and can make a significant contribution to costs. To reduce the quantity of filter aid needed, some processors force air into the extract as it is being diluted with water (the extract and diluting water are mixed in an in-line mixer into which air is forced). Fine air bubbles attach themselves to the particles of residue. The diluted extract is left standing for several hours while the air rises to the top, taking the residue particles with it. This frothy mix of air and residue is removed from the top and the solution is withdrawn from the bottom and pumped to the filter.
The next step is precipitation of the alginate from the filtered solution, either as alginic acid or calcium alginate.
FIGURE 32
Flow chart for the production of sodium alginate (after McHugh, 1987).
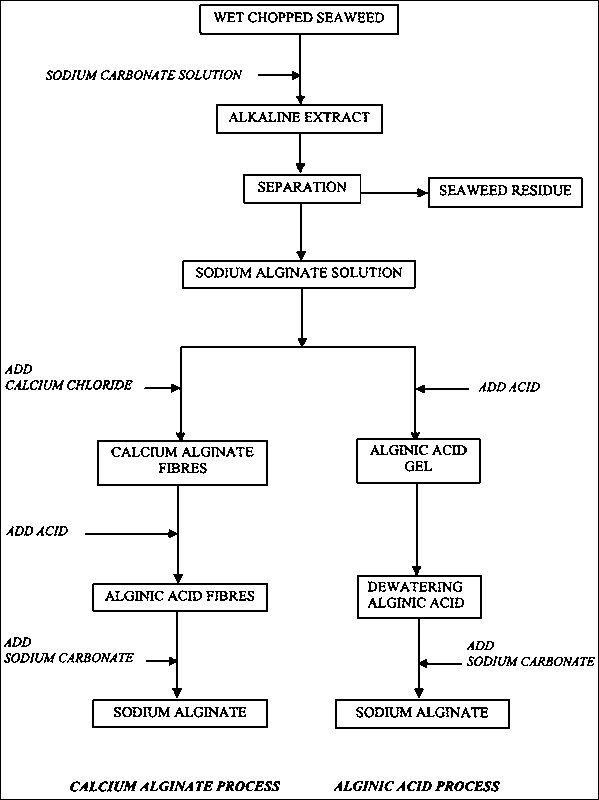
Alginic acid method
When acid is added to the filtered extract, alginic acid forms in soft, gelatinous pieces that must be separated from the water. Again flotation is often used; filtration is not possible because of the soft jelly-like nature of the solid. If an excess of sodium carbonate is used in the original extraction, this will still be present in the filtered extract so that when acid is added, carbon dioxide will form. Fine bubbles of this gas attach themselves to the pieces of alginic acid and lift them to the surface where they can be continuously scrapped away. The processor now has a jelly-like mass of alginic acid that actually contains only 1-2 percent alginic acid, with 98-99 percent water. Somehow, this water content must be reduced. It is too soft to allow the use of a screw press. Some processors place the gel in basket-type centrifuges lined with filter cloth. Centrifuging can increase the solids to 7-8 percent and this is sufficient if alcohol is to be used in the next step of converting it to sodium alginate. It is also now sufficiently firm to be squeezed in a screw press. The 7-8 percent alginic acid is place in a mixer and, allowing for the water contained in the alginic acid, enough alcohol (usually ethanol or isopropanol) is added to give a 50:50 mixture of alcohol and water. Then solid sodium carbonate is added gradually until the resulting paste reaches the desired pH. The paste of sodium alginate can be extruded as pellets, oven dried and milled.
Calcium alginate method
When a soluble calcium salt, such as calcium chloride, is added to the filtered extract, solid calcium alginate is formed. If the calcium solution and filtered extract are mixed carefully, the calcium alginate can be formed as fibres - bad mixing gives a gelatinous solid. This fibrous material can be readily separated on a metal screen (sieve) and washed with water to remove excess calcium. It is then stirred in dilute acid and converted to alginic acid, which retains the fibrous characteristics of the calcium alginate. This form of alginic acid can be easily squeezed in a screw press. A screw press with a graduated-pitch screw is usually used; the squeezing action must be applied very gradually, otherwise the material will just move backwards and out of the press. The product from the screw press looks relatively solid but still contains only 20-25 percent alginic acid. However, it is dry enough to form a paste when sodium carbonate is mixed with it to convert it to sodium alginate. Sodium carbonate is added to the alginic acid in a suitable type of mixer until the required pH is reached, then the paste is extruded as pellets, dried and milled.
The disadvantage of this second method, compared to the alginic acid method, is that an extra step is added to the process. The advantage is that the handling of the fibrous calcium alginate and alginic acid is much simpler and alcohol is not needed. Alcohol is expensive and while it is usually recovered and recycled, recovery is never 100 percent, so its use adds to the costs.
Other important factors in alginate production are colour control of the product, water supply and waste disposal.
If the original seaweed is highly coloured, e.g. Ascophyllum, the alkaline extract will also be highly coloured and the process will eventually yield a dark product that commands only a low price as it is limited to use in technical applications. Lighter coloured seaweeds, such as Macrocystis, yield a lighter coloured alginate suitable for food and other applications. Colour can be controlled by the use of bleach - sodium hypochlorite - that is added to the filtered alkaline extract or even to the paste at the final conversion stage. Care must be taken, since excessive bleach can lower the viscosity of the alginate, reducing its value. Sometimes the seaweed is soaked in a formalin solution before it is extracted with alkali. The formalin helps to bind the coloured compounds to the cellulose in the cell walls, so much of the colour is left behind in the seaweed residue when the alkaline extract is filtered.
Large quantities of water are used in the process, especially when diluting the thick (viscous) initial alkaline extract to a viscosity suitable for filtration. A plentiful and reliable water supply is a necessity for an alginate factory to survive.
Waste waters from filtration are alkaline, they contain calcium from the calcium precipitation (excess calcium gives a more fibrous calcium alginate) and acid from the acid conversion step. In some countries the waste is pumped out to sea. Where environmental concerns are greater, or when water supplies are limited, recycling is not too difficult and its costs may be partly offset by the lowering of the quantity and cost of water used by the factory. A means of disposing of solid wastes - the seaweed residue and used filter aid - must be found. There have been several positive studies reported on the use of this waste to adsorb heavy metals, such as cadmium, zinc and copper, from industrial liquid wastes (e.g. Romero-Gonzalez, Williams and Gardiner, 2001). Attempts to ferment this waste to produce ethanol from the cellulose content appear to be less promising, in economic terms (Horn, Aasen and Oestgaard, 2000).
For further details
In the above descriptions of the production process, suggestions about the detail of reagent concentrations, time, temperatures, etc., have been deliberately omitted to simplify the reading of it. Commercial processors regard these details as confidential and do not publish them. However, some detail can be found in McHugh (1987) and in the results of a series of trials, run on a pilot-plant scale using Macrocystis as raw material, published by Hernández-Carmona et al. (1998), Hernández-Carmona, McHugh and López-Gutiérrez (1999), McHugh et al. (2001) and Hernández-Carmona et al. (2002).
Sodium alginate, produced as described above, is the main form of alginate in use. Smaller quantities of alginic acid and the calcium, ammonium and potassium salts, and an ester, propylene glycol alginate, are also produced. Calcium alginate and alginic acid are made during the calcium alginate process for making sodium alginate; each can be removed at the appropriate stage and, after thorough washing, can be dried and milled to a required particle size. The other salts are made by neutralization of moist alginic acid with the appropriate alkali, usually ammonium hydroxide or potassium carbonate; sufficient water or alcohol can be added to keep the material at a workable consistency; they are processed as described for the paste-conversion method in the calcium alginate process for sodium alginate production.
Propylene glycol alginate, an ester of alginic acid, has different properties and uses from the salts such as sodium alginate. It was first patented in 1947 and has been the subject of further patents as methods for its production were improved. It is made by taking moist alginic acid (20 percent or greater solids) that has been partially reacted with sodium carbonate, and treating it with liquid propylene oxide in a pressure vessel for 2 hours at about 80°C. The product is dried and milled. Further details and references to the patents can be found in McHugh (1987).
The capacities of alginate producers are summarized according to their broad geographical location and given in Table 5, and the principal alginate producers are listed below.
TABLE 5
Alginate processors. Capacity in tonnes (2001)
| |
Ca & |
PGA |
Total |
% |
|
Europe |
16 000 |
- |
16 000 |
44 |
|
Africa |
- |
- |
- |
- |
|
Americas |
3 000 |
1 500 |
4 500 |
13 |
|
Asia-Pacific |
14 000 |
1 600 |
15 600 |
43 |
|
Total |
33 000 |
3 100 |
36 100 |
|
______________________________________Source: H. Porse, CP Kelco ApS, 2002, pers. comm.
ISP Alginates (UK) Ltd
Waterfield
Tadworth
Surrey KT20 5HQ
United Kingdom
Tel: [INT+44] + (1737) 377 000
Fax: [INT+44] + (1737) 377148
Website: www.ispcorp.com/products/alginates
______________________________________
FMC Biopolymer
1735 Market Street
Philadelphia PA 19103
United States of America
Tel: [INT+1] + (215) 299 6000
Fax: [INT+1] + (215) 299 5809
Websites: www.fmc.com;
www.fmcbiopolymer.com
______________________________________
Degussa Texturant Systems
Lise-Meitner-St.34
85354 Freising
Germany
Tel: [INT+49] + (8161) 548 266
Fax: [INT+49] + (8161) 548 582
Website: www.texturantsystems.com
______________________________________
Danisco Cultor
Edwin Rahrs Vej 38
8220 Brabrand
Denmark
Tel: [INT+45] + 89 43 50 00
Fax: [INT+45] + 86 25 06 81
Website: www.daniscocultor.com
______________________________________
Kimica Corporation
15-4 Uchikanda 2 - chome
Chiyoda-ku
Tokyo
Japan
Tel: [INT+81] + (3) 3252 8708
Fax: [INT+81] + (3) 3252 8704
Website: www.kimica.com
______________________________________
Fuji Chemical Industry Co Ltd
1570, Nakanoshima
Wakayama City
Japan
Tel: [INT+81] + (73) 423 1247
Fax: [INT+81] + (73) 431 3005
Website: www.waila.or.jp/kasei
______________________________________
Algisa, Compania Industrial de Alginatos S.A.
PO Box 51645
Santiago 1
Chile
Tel: [INT+56] + 2 738 7904
Fax: [INT+56] + 2 738 7038
______________________________________
China Seaweed Industrial Association
Room 203 - Building 18 Maizidian Street -
Chaoyang Dist. Beijing 100026
China
Tel: [INT+86] + 10 6419 5140
Fax: [INT+86] + 10 6419 5141
This seems to be sponsored by the Ministry of Fisheries and represents the alginate industry.
The uses of alginates are based on three main properties. The first is their ability, when dissolved in water, to thicken the resulting solution (more technically described as their ability to increase the viscosity of aqueous solutions). The second is their ability to form gels; gels form when a calcium salt is added to a solution of sodium alginate in water. The gel forms by chemical reaction, the calcium displaces the sodium from the alginate, holds the long alginate molecules together and a gel is the result. No heat is required and the gels do not melt when heated. This is in contrast to the agar gels where the water must be heated to about 80°C to dissolve the agar and the gel forms when cooled below about 40°C. The third property of alginates is the ability to form films of sodium or calcium alginate and fibres of calcium alginates.
Alginate molecules are long chains that contain two different acidic components, abbreviated here for simplicity to M and G. The way in which these M and G units are arranged in the chain and the overall ratio, M/G, of the two units in a chain can vary from one species of seaweed to another. In other words all "alginates" are not necessarily the same. So some seaweeds may produce an alginate that gives a high viscosity when dissolved in water, others may yield a low viscosity alginate. The conditions of the extraction procedure can also affect viscosity, lowering it if conditions are too severe. All of this results in sellers normally offering a range of alginates with differing viscosities.
Similarly, the strength of the gel formed by the addition of calcium salts can vary from one alginate to another. Generally alginates with a higher content of G will give a stronger gel; such alginates are said to have a low M/G ratio.
Some examples: Macrocystis can gives a medium-viscosity alginate, or a high viscosity with a careful extraction procedure (lower temperature for the extraction). Sargassum usually gives a low viscosity product. Laminaria digitata gives a soft to medium strength gel, while Laminaria hyperborea and Durvillaea give strong gels. These are some of the reasons why alginate producers like to have a variety of seaweed sources, to match the alginate to the needs of particular applications.
In textile printing, alginates are used as thickeners for the paste containing the dye. These pastes may be applied to the fabric by either screen or roller printing equipment. Alginates became important thickeners with the advent of reactive dyes. These combine chemically with cellulose in the fabric. Many of the usual thickeners, such as starch, react with the reactive dyes, and this leads to lower colour yields and sometimes by-products that are not easily washed out. Alginates do not react with the dyes, they easily wash out of the finished textile and are the best thickeners for reactive dyes. Alginates are more expensive than starch and recently starch manufacturers have made efforts to produce modified starches that do not react with the reactive dyes, so it is becoming a more competitive market. This use of alginate represents a large market, but it is affected by economic recessions when there is often a fall in demand for clothing and textiles. The types of alginate required vary from medium-to-high viscosity with older screen printing equipment, to low viscosity if modern, high speed, roller printing is used. Textile printing accounts for about 50 percent of the global alginate market.
The thickening property of alginate is useful in sauces and in syrups and toppings for ice cream. By thickening pie fillings with alginate, softening of the pastry by liquid from the filling is reduced. Addition of alginate can make icings non-sticky and allow the baked goods to be covered with plastic wrap. Water-in-oil emulsions such as mayonnaise and salad dressings are less likely to separate into their original oil and water phases if thickened with alginate. Sodium alginate is not useful when the emulsion is acidic, because insoluble alginic acid forms; for these applications propylene glycol alginate (PGA) is used since this is stable in mild acid conditions. Alginate improves the texture, body and sheen of yoghurt, but PGA is also used in the stabilization of milk proteins under acidic conditions, as found in some yoghurts. Some fruit drinks have fruit pulp added and it is preferable to keep this in suspension; addition of sodium alginate, or PGA in acidic conditions, can prevent sedimentation of the pulp. In chocolate milk, the cocoa can be kept in suspension by an alginate/phosphate mixture, although in this application it faces strong competition from carrageenan. Small amounts of alginate can thicken and stabilize whipped cream.
For more information about factors that affect the viscosity of alginate solutions, see King (1983: 132-141). This discusses the effects of concentration of alginate, its molecular weight, the presence of any calcium remaining in the alginate from the extraction process, pH, temperature and other salts. For a briefer discussion, see McHugh (1987) or Clare (1993).
Alginates have some applications that are not related to either their viscosity or gel properties. They act as stabilizers in ice cream; addition of alginate reduces the formation of ice crystals during freezing, giving a smooth product. This is especially important when ice cream softens between the supermarket and the home freezer; without alginate or similar stabilizer the refrozen ice cream develops large ice crystals, giving it an undesirable crunchy mouth feel. Alginate also reduces the rate at which the ice cream will melt. Beer drinkers prefer some foam on the top of a newly-poured glass, and a poor foam leads to a subjective judgement that the beer is poor quality. Addition of a very low concentration of propylene glycol alginate will provide a stable, longer lasting beer foam. A variety of agents are used in the clarification of wine and removal of unwanted colouring - wine fining - but in more difficult cases it has been found that the addition of sodium alginate can be effective.
The gelling properties of alginate were used in the first production of artificial cherries in 1946. A flavoured, coloured solution of sodium alginate was allowed to fall, in large drops, into a solution of a calcium salt. Calcium alginate immediately formed as a skin on the outside of the drop and when the drop was allowed to sit in the solution, the calcium gradually penetrated the drop converting it all into a gel that hardened with further standing. Because the cherry-flavoured gels did not melt, they became very popular in bakery products. Fruit substitutes can now be made by automated and continuous processes that are based on similar principles. Either the calcium can be applied externally, as above, or internally. In the latter case a calcium salt that does not dissolve is added to the fruit puree, together with a weak acid; the weak acid slowly attacks the calcium salt and releases water-soluble calcium that then reacts with the alginate and forms the gel.
Edible dessert jellies can be formed from alginate-calcium mixtures, often promoted as instant jellies or desserts because they are formed simply by mixing the powders with water or milk, no heat being required. Because they do not melt, alginate jellies have a different, firmer mouth feel when compared to gelatin jellies, which can be made to soften and melt at body temperature. Mixtures of calcium salts and sodium alginate can be made to set to a gel at different rates, depending on the rate at which the calcium salt dissolves. Gel formation can also be delayed even after everything is mixed together; this is done using a gel-retarder that reacts with the calcium before the alginate does, so no calcium is available to the alginate until all the retarder is used. In this way gel formation can be delayed for several minutes if desired, such as when other ingredients need to be added and mixed before the gel starts to set.
Alginate gels are used in re-structured or re-formed food products. For example, re-structured meats can be made by taking meat pieces, binding them together and shaping them to resemble usual cuts of meat, such as nuggets, roasts, meat loaves, even steaks. The binder can be a powder of sodium alginate, calcium carbonate, lactic acid and calcium lactate. When mixed with the raw meat they form a calcium alginate gel that binds the meat pieces together. This is used for meats for human consumption, such as chicken nuggets; it has become especially useful in making loaves of meat for fresh pet food; some abattoir wastes are suitable and cheap ingredients. Up to 1 percent alginate is used. Similar principles are applied to making shrimp substitutes using alginate, proteins such as soy protein concentrate, and flavours. The mixture is extruded into a calcium chloride bath to form edible fibres which are chopped, coated with sodium alginate and shaped in a mould. Restructured fish fillets have been made using minced fish and a calcium alginate gel. Onion rings are made from dried onion powder; pimento olive fillings are made using pimento pulp. In 2001, a new line of olives launched in Spain were stuffed with flavoured pastes, such as garlic, herbs, hot pepper, lemon and cheese. Each of these is made with green manzanilla olives and an alginate-based paste containing the appropriate ingredient to provide the flavour.
Calcium alginate films and coatings have been used to help preserve frozen fish. The oils in oily fish such as herring and mackerel can become rancid through oxidation even when quick frozen and stored at low temperatures. If the fish is frozen in a calcium alginate jelly, the fish is protected from the air and rancidity from oxidation is very limited. The jelly thaws with the fish so they are easily separated. If beef cuts are coated with calcium alginate films before freezing, the meat juices released during thaw are re-absorbed into the meat and the coating also helps to protect the meat from bacterial contamination. If desired, the calcium alginate coating can be removed by re-dissolving it with sodium polyphosphate.
For further details
For more information about alginate gels, how they form, calcium salts to use, retardants, acid-release agents, and effect of alginates from different seaweeds, the most thorough discussion is to be found in King (1983: 141-154). While this may appear to be an old reference, these basics of gel formation are still the same. Other, briefer, treatments of gel formation are available in Clare (1993), Nussinovitch (1997) or Onsoyen (1997).
Re-structured foods are discussed in Chapter 20 in Nussinovitch (1997).
A brief tabular summary of alginate applications in food can be found in Onsoyen (1997).
Many commercial chemical syntheses and conversions are best carried out using biocatalysts such as enzymes or active whole cells. Examples include the use of enzymes for the conversion of glucose to fructose, the production of L-amino acids for use in foods, the synthesis of new penicillins after hydrolysis of penicillin G, the use of whole cells for the conversion of starch to ethanol (for beer brewing), and the continuous production of yoghurt. To carry out these processes on a moderate to large scale, the biocatalysts must be in a concentrated form and be recoverable from the process for re-use.
This can be achieved by "immobilizing" the enzymes or cells by entrapping them in a material that will still allow penetration by the substance to be converted or changed. Originally, single enzymes were isolated and used for a specific conversion, but now similar or better results can be obtained using whole cells, and this is more economical. An added advantage of immobilization is that the cells last longer. Ordinary suspended cells may have good activity for only 1-2 days, while immobilized cells can last for 30 days. Beads made with calcium alginate were one of the first materials to be used for immobilization. The whole cells are suspended in a solution of sodium alginate and this is added dropwise to a calcium chloride solution. The beads form in much the same way as described for artificial cherries. In use, they are packed into a column and a solution of the substance to be converted is fed into the top of the column and allowed to flow through the bed of beads containing the immobilized biocatalyst in the cells. The conversion takes place and the product comes out at the bottom. A simple example is to immobilize yeast cells, flow a solution of sugar through the beads, and the sugar is converted to alcohol.
For further details
Details of methods for making immobilized materials using alginates are available from alginate manufacturers (such as FMC Biopolymers, ISP Alginates, etc.). Today, many other materials besides alginate are used for immobilization. Nussinovitch (1997), in Chapter 15, discusses immobilization by a variety of polymers, including alginate, and gives many examples of its applications.
If a fine jet of sodium alginate solution is forced into a bath of a calcium chloride solution, calcium alginate is formed as fibres. If low viscosity alginates are used, a strong solution can be used without any viscosity problems and the calcium bath is not diluted as rapidly. The fibres have very good strength when both wet and dry. As with most polymer fibres formed by extrusion, stretching while forming increases the linearity of the polymer chains and the strength of the fibre. Good quality stable fibres have been produced from mixed salts of sodium and calcium alginate, and processed into non-woven fabric that is used in wound dressings. They have very good wound healing and haemostatic properties and can be absorbed by body fluids because the calcium in the fibre is exchanged for sodium from the body fluid to give a soluble sodium alginate. This also makes it easy to remove these dressings from large open wounds or burns since they do not adhere to the wound. Removal can be assisted by applying saline solutions to the dressing to ensure its conversion to soluble sodium alginate. Recently, the consumer division of a multinational pharmaceutical company launched a new line of adhesive bandages and gauze pads based on calcium alginate fibres. They are being promoted as helping blood to clot faster - twice as fast as their older, well established product.
Alginic acid powder swells when wetted with water. This has led to its use as a tablet disintegrant for some specialized applications. Alginic acid has also been used in some dietary foods, such as biscuits; it swells in the stomach and, if sufficient is taken, it gives a "full" feeling so the person is dissuaded from further eating. The same property of swelling has been used in products such as Gavisconä tablets, which are taken to relieve heartburn and acid indigestion. The swollen alginic acid helps to keep the gastric contents in place and reduce the likelihood of reflux irritating the lining of the oesophagus.
Alginate is used in the controlled release of medicinal drugs and other chemicals. In some applications, the active ingredient is placed in a calcium alginate bead and slowly released as the bead is exposed in the appropriate environment. More recently, oral controlled-release systems involving alginate microspheres, sometimes coated with chitosan to improve the mechanical strength, have been tested as a way of delivering various drugs. Pronova Biomedical AS, a leading supplier of ultra-pure alginates and chitosans for controlled release and other medical materials applications, was acquired by FMC Bioploymer in early 2002; FMC had previously acquired Pronova Biopolymer, producer of food and technical grade alginates.
Paper
The main use for alginate in the paper industry is in surface sizing. Alginate added to the normal starch sizing gives a smooth continuous film and a surface with less fluffing. The oil resistance of alginate films give a size with better oil resistance and enhances greaseproof properties. An improved gloss is obtained with high gloss inks. If papers or boards are to be waxed, alginate in the size will keep the wax mainly at the surface. They give better coating runability than other thickeners, especially in hot, on-machine coating applications. Alginates are also excellent film formers and improve ink holdout and printability. The quantity of alginate used is usually 5-10 percent of the weight of starch in the size.
Alginate is also used in starch adhesives for making corrugated boards because it stabilizes the viscosity of the adhesive and allows control of its rate of penetration. One percent sodium alginate, based on the weight of starch used, is usually sufficient.
Paper coating methods and equipment have developed significantly since the late 1950s with the demand for a moderately priced coated paper for high quality printing. Trailing blade coating equipment runs at 1 000 m/minute or more, so the coating material, usually clay plus a synthetic latex binder, must have consistent rheological properties under the conditions of coating. Up to 1 percent alginate will prevent change in viscosity of the coating suspension under the high shear conditions where it contacts the roller. The alginate also helps to control water loss from the coating suspension into the paper, between the point where the coating is applied and the point where the excess is removed by the trailing blade. The viscosity of the coating suspension must not be allowed to increase by loss of water into the paper because this leads to uneven removal by the trailing blade and streaking of the coating. Medium to high viscosity alginates are used, at a rate of 0.4-0.8 percent of the clay solids. Because of the solvent resistance of alginate films, the print quality of the finished paper is improved.
Welding rods
Coatings are applied to welding rods or electrodes to act as a flux and to control the conditions in the immediate vicinity of the weld, such as temperature or oxygen and hydrogen availability. The dry ingredients of the coating are mixed with sodium silicate (water-glass) which gives some of the plasticity necessary for extrusion of the coating onto the rod; it also acts as the binder for the dried coating on the rod. However, the wet silicate has no binding action and does not provide sufficient lubrication to allow effective and smooth extrusion. An additional lubricant is needed, and a binder that will hold the damp mass together before extrusion and maintain the shape of the coating on the rod during drying and baking. Alginates are used to meet these requirements. The quantities of alginates used are very dependent on the type of welding rod being coated and the extrusion equipment being used. Alginate manufacturers are the best source of information for using alginates in welding rod applications.
Binders for fish feed
The worldwide growth in aquaculture has led to the use of crude alginate as a binder in salmon and other fish feeds, especially moist feed made from fresh waste fish mixed with various dry components. Alginate binding can lower consumption by up to 40 percent and pollution of culture ponds is sharply reduced.
Release agents
The poor adhesion of films of alginate to many surfaces, together with their insolubility in nonaqueous solvents, have led to their use as mould release agents, originally for plaster moulds and later in the forming of fibreglass plastics. Sodium alginate also makes a good coating for anti-tack paper, which is used as a release agent in the manufacture of synthetic resin decorative boards. Films of calcium alginate, formed in situ on a paper, have been used to separate decorative laminates after they have been formed in a hot-pressing system.
A summary of alginate markets is shown in Table 6. The total market has a value of about US$ 195 million.
There are difficulties and costs in the marketing of seaweed hydrocolloids, such as alginate, agar and carrageenan, which are not always apparent to those outside the industry. In some markets, one may compete with another; in others, one might be the only real choice. They must all compete, in at least some of their uses, with plant gums (such as guar and locust bean) and cellulose derivatives (such as carboxy methyl cellulose (CMC) and methyl cellulose) that are often cheaper. It is important to realize that price may not be the determining factor in a buyer's choice of a seaweed hydrocolloid; quality and its reproducibility from one batch to another may be more important. Frequently a buyer uses less than 1 percent of the hydrocolloid in his product, so a 20 percent price difference may be inconsequential in the total cost of a product. Many a buyer of seaweed hydrocolloids, satisfied with one particular brand or grade, will, despite a higher price, stay with it because the risks of changing may not seem to be worth the saving. So, in seaweed hydrocolloids, those brands already established in the market often hold a very strong, entrenched position. To dislodge them, a marketing group should include a strong technical team that can run tests and trials to convince the buyer of the equivalence of the new product; sometimes this requires a detailed knowledge of the buyer's industry. In promoting new sales, the hydrocolloid producer may have to provide complete formulations and technical know-how to potential buyers. Therefore selling costs of the seaweed hydrocolloids can be high and account must be taken of this by the potential producer.
The buyers of alginates fall into two groups. The first is a number of large buyers who know exactly what they want and who require little servicing because they have their own resources. This group includes those speciality gum companies who service smaller users by preparing their own blends of seaweed hydrocolloids and other hydrocolloids, according to the requirements of a particular customer.
The second and larger group are the smaller users who need some technical service support. Frequently this group yields more profitable sales in the long term because they may be sold specifically formulated products at a premium price and they are generally more reluctant to change to a competitor's product. At the same time, it takes more time and expense to establish such sales. Many of the major producers have such speciality products, shown by the large range of products listed by them.
Alginate manufacturers usually sell direct to the major markets, but in minor markets it is more economical to sell through an agent, leaving the task of market penetration to them, but providing technical support where necessary. Agents need to have an appreciation of the application of hydrocolloids and a knowledge of the client's industry. This ideal might be achieved by an agent selling a variety of chemicals to just one industry, like the food industry, but an agent who deals principally in hydrocolloids over a range of industries, which is a not uncommon situation, usually needs more backup from the producer. Large wholesalers or agents may buy from the producer and re-sell; otherwise they operate on a commission of 5-15 percent, depending partly on the degree of assistance required from the producer.
TABLE 6
Alginate markets (2001)
|
Application |
tonnes |
% |
|
Food and pharmaceutical |
10 000 |
33 |
|
All technical grades |
20 000 |
67 |
|
Total |
30 000 |
|
Source: H. Porse, CP Kelco ApS, 2002, pers. comm.
The above paragraphs are based on McHugh (1987), but the statements made are just as valid at the time of writing.
The overall annual growth rate for alginates is 2-3 percent, with textile printing applications accounting for about half of the global market. However, the textile industry is flat at present, as it rides a trough in the cycle of peaks and troughs, and it is 90 percent based in Asia and the Near East (Turkey). Pharmaceutical and medical uses are about 20 percent by value of the market and have stayed buoyant, with 2-4 percent annual growth rates, driven by ongoing developments in controlled release technologies and the use of alginates in wound care applications. Food applications are worth about 20 percent of the market. That sector has been growing only slowly, and recently has grown at only 1-2 percent annually. The paper industry takes about 5 percent and the sector is very competitive, not increasing but just holding its own. The alginate industry faces strong competition from Chinese producers, whose prices do not reflect the real expense of cultivating Laminaria japonica, even in China, yet they do not appear to import sufficient wild seaweeds to offset those costs. The result is low profitability for most of the industry, with the best opportunities lying in the high end of the market, such as pharmaceutical and medical applications.