Freezing is one of the oldest and most widely used methods of food preservation, which allows preservation of taste, texture, and nutritional value in foods better than any other method. The freezing process is a combination of the beneficial effects of low temperatures at which microorganisms cannot grow, chemical reactions are reduced, and cellular metabolic reactions are delayed (Delgado and Sun, 2000).
Freezing preservation retains the quality of agricultural products over long storage periods. As a method of long-term preservation for fruits and vegetables, freezing is generally regarded as superior to canning and dehydration, with respect to retention in sensory attributes and nutritive properties (Fennema, 1977). The safety and nutrition quality of frozen products are emphasized when high quality raw materials are used, good manufacturing practices are employed in the preservation process, and the products are kept in accordance with specified temperatures.
The need for freezing and frozen storage
Freezing has been successfully employed for the long-term preservation of many foods, providing a significantly extended shelf life. The process involves lowering the product temperature generally to -18 °C or below (Fennema et al., 1973). The physical state of food material is changed when energy is removed by cooling below freezing temperature. The extreme cold simply retards the growth of microorganisms and slows down the chemical changes that affect quality or cause food to spoil (George, 1993).
Competing with new technologies of minimal processing of foods, industrial freezing is the most satisfactory method for preserving quality during long storage periods (Arthey, 1993). When compared in terms of energy use, cost, and product quality, freezing requires the shortest processing time. Any other conventional method of preservation focused on fruits and vegetables, including dehydration and canning, requires less energy when compared with energy consumption in the freezing process and storage. However, when the overall cost is estimated, freezing costs can be kept as low (or lower) as any other method of food preservation (Harris and Kramer, 1975).
Current status of frozen food industry in U.S. and other countries
The frozen food market is one of the largest and most dynamic sectors of the food industry. In spite of considerable competition between the frozen food industry and other sectors, extensive quantities of frozen foods are being consumed all over the world. The industry has recently grown to a value of over US$ 75 billion in the U.S. and Europe combined. This number has reached US$ 27.3 billion in 2001 for total retail sales of frozen foods in the U.S. alone (AFFI, 2003). In Europe, based on U.S. currency, frozen food consumption also reached 11.1 million tons in 13 countries in the year 2000 (Quick Frozen Foods International, 2000). Table 1 represents the division of frozen food industry in terms of annual sales in 2001.
Advantages of freezing technology in developing countries
Developed countries, mostly the U.S., dominate the international trade of fruits and vegetables. The U.S. is ranked number one as both importer and exporter, accounting for the highest percent of fresh produce in world trade. However, many developing countries still lead in the export of fresh exotic fruits and vegetables to developed countries (Mallett, 1993).
For developing countries, the application of freezing preservation is favorable with several main considerations. From a technical point of view, the freezing process is one of the most convenient and easiest of food preservation methods, compared with other commercial preservation techniques. The availability of different types of equipment for several different food products results in a flexible process in which degradation of initial food quality is minimal with proper application procedures. As mentioned earlier, the high capital investment of the freezing industry usually plays an important role in terms of economic feasibility of the process in developing countries. As for cost distribution, the freezing process and storage in terms of energy consumption constitute approximately 10 percent of the total cost (Person and Lohndal, 1993). Depending on the government regulations, especially in developing countries, energy cost for producers can be subsidized by means of lowering the unit price or reducing the tax percentage in order to enhance production. Therefore, in determining the economical convenience of the process, the cost related to energy consumption (according to energy tariffs) should be considered. Electricity prices for some countries are given in Table 2.
Table 1. Frozen food industry in terms of annual sales in
2001
(Source: Information Resources)
|
Food items |
Sales US$ |
% Change |
|
Total Frozen Food Sales |
26 600 |
6.1 |
|
Baked Goods |
1 400 |
9.0 |
|
Breakfast Foods |
1 050 |
4.1 |
|
Novelties |
1 900 |
10.5 |
|
Ice Cream |
4 500 |
5.7 |
|
Frozen Dessert/Fruit/Toppings |
786 |
5.4 |
|
Juices/Drinks |
827 |
-9.7 |
|
Vegetables |
2 900 |
4.3 |
Increasing consumer demand in developing countries due to modernization
The proportion of fresh food preserved by freezing is highly related to the degree of economic development in a society. As countries become wealthier, their demand for high-valued commodities increases, primarily due to the effect of income on the consumption of high-valued commodities in developing countries. The commodities preserved by freezing are usually the most perishable ones, which also have the highest price. Therefore, the demand for these commodities is less in developing areas. Besides, the need for adequate technology for freezing process is the major drawback of developing countries in competing with industrialized countries. The frozen food industry requires accompanying developments and facilities for transporting, storing, and marketing their products from the processing plant to the consumer (Mallett, 1993). Thus, a large amount of capital investment is needed for these types of facilities. For developing countries, especially in rural or semi-rural areas, the frozen food industry has therefore not been developed significantly compared to other countries.
In recent years, due to the changing consumer profile, the frozen food industry has changed significantly. The major trend in consumer behavior documented over the last half century has been the increase in the number of working women and the decline in the family size. These two factors resulted in a reduction in time spent preparing food. The entry of more women into the workforce also led to improvements in kitchen appliances and increased the variability of ready-to-eat or frozen foods available in the market. Besides, the increased usage of microwave ovens, affecting food habits in general and the frozen food market in particular, as well as allowing rapid preparation of meals and greater flexibility in meal preparation. The frozen food industry is now only limited by imagination, an output of which increases continuously to supply the increasing demand for frozen products and variability.
Table 2. Unit electricity prices for industry1 (U.S. Dollars
per Kilowatt-hour)
Source: United States - Energy Information Administration, Monthly Energy Review,
July 2003.
|
Country |
1999 |
2000 |
2001 |
2002 |
|
Argentina |
n.a. |
0.075 |
0.069 |
n.a. |
|
Belgium |
0.056 |
0.048 |
n.a. |
n.a. |
|
Bolivia |
n.a. |
0.062 |
0.069 |
n.a. |
|
Chile |
n.a. |
0.052 |
0.056 |
n.a. |
|
Chinese Taipei (Taiwan) |
0.058 |
0.061 |
0.056 |
n.a. |
|
Colombia |
n.a. |
0.052 |
0.042 |
n.a. |
|
Costa Rica |
n.a. |
0.068 |
0.076 |
n.a. |
|
Cuba |
n.a. |
0.080 |
0.078 |
n.a. |
|
Ecuador |
n.a. |
0.036 |
0.061 |
n.a. |
|
El Salvador |
n.a. |
0.111 |
0.110 |
n.a. |
|
Finland |
0.046 |
0.039 |
0.038 |
0.043 |
|
Germany |
0.057 |
0.041 |
0.044 |
n.a. |
|
Greece |
0.050 |
0.042 |
0.043 |
0.046 |
|
Guyana |
n.a. |
0.082 |
0.080 |
n.a. |
|
Hungary |
0.055 |
0.049 |
0.051 |
0.060 |
|
India |
0.081 |
0.080 |
n.a. |
n.a. |
|
Ireland |
0.057 |
0.049 |
0.060 |
0.075 |
|
Italy |
0.086 |
0.089 |
n.a. |
n.a. |
|
Korea (Korea, South) |
0.056 |
0.062 |
0.057 |
n.a. |
|
Mexico |
0.042 |
0.051 |
0.053 |
n.a. |
|
Netherlands |
0.061 |
0.057 |
0.059 |
n.a. |
|
New Zealand |
0.030 |
0.030 |
0.028 |
0.033 |
|
Nicaragua |
n.a. |
0.117 |
0.115 |
n.a. |
|
Paraguay |
n.a. |
0.032 |
0.036 |
n.a. |
|
Peru |
n.a. |
0.056 |
0.057 |
n.a. |
|
Poland |
0.037 |
0.037 |
0.045 |
0.049 |
|
Portugal |
0.078 |
0.067 |
0.066 |
0.068 |
|
Russia |
0.012 |
0.011 |
n.a. |
n.a. |
|
South Africa |
0.017 |
0.017 |
0.013 |
n.a. |
|
Spain |
0.049 |
0.043 |
0.041 |
n.a. |
|
Switzerland |
0.090 |
0.069 |
0.069 |
0.073 |
|
Turkey |
0.079 |
0.080 |
0.079 |
0.094 |
|
United Kingdom |
0.064 |
0.055 |
0.048 |
n.a. |
|
United States 2 |
0.044 |
0.046 |
0.050 |
0.048 |
|
Uruguay |
n.a. |
0.064 |
0.070 |
n.a. |
n.a. = Not Available.
1 Energy end-use prices including taxes converted using exchange rates.
2 Electricity prices in the United States, including income taxes, environmental charges, and other charges.
Market share of frozen fruits and vegetables
Today in modern society, frozen fruits and vegetables constitute a large and important food group among other frozen food products (Arthey, 1993). The historical development of commercial freezing systems designed for special food commodities helped shape the frozen food market. Technological innovations as early as 1869 led to the commercial development and marketing of some frozen foods. Early products saw limited distribution through retail establishments due to insufficient supply of mechanical refrigeration. Retail distribution of frozen foods gained importance with the development of commercially frozen vegetables in 1929.
The frozen vegetable industry mostly grew after the development of scientific methods for blanching and processing in the 1940s. Only after the achievement of success in stopping enzymatic degradation, did frozen vegetables gain a strong retail and institutional appeal. Today, market studies indicate that considering overall consumption of frozen foods, frozen vegetables constitute a very significant proportion of world frozen-food categories (excluding ice cream) in Austria, Denmark, Finland, France, Germany, Italy, Netherlands, Norway, Sweden, Switzerland, UK, and the USA. The division of frozen vegetables in terms of annual sales in 2001 is shown in Table 3.
Commercialization history of frozen fruits is older than frozen vegetables. The commercial freezing of small fruits and berries began in the eastern part of the U.S. in about 1905 (Desrosier and Tressler, 1977). The main advantage of freezing preservation of fruits is the extended usage of frozen fruits during off-season. Additionally, frozen fruits can be transported to remote markets that could not be accessed with fresh fruit. Also, freezing preservation makes year-round further processing of fruit products possible, such as jams, juice, and syrups from frozen whole fruit, slices, or pulps. In summary, the preservation of fruits by freezing has clearly become one the most important preservation methods.
Future trends in freezing technology
The frozen food industry is highly based in modern science and technology. Starting with the first historical development in freezing preservation of foods, today, a combination of several factors influences the commercialization and usage of freezing technology. The future growth of frozen foods will mostly be affected by economical and technological factors. Growth in population, personal incomes, relative cost of other forms of foods, changes in tastes and preferences, and technological advances in freezing methods are some of the factors concerned with the future of freezing technology (Enochian and Woolrich, 1977).
Population growth and increasing demand for food has generated the need for commercial production of food commodities in large-scale operations. Thus, availability of proper equipment suitable for continuous processing would be valuable for freezing preservation methods. In addition depending on personal incomes, relative cost of frozen products is one of the most important of economical factors. Producing the highest quality at the lowest cost possible is highly dependent on the technology used. As a result, developments in freezing technology in recent years have mostly been characterized by the improvements in mechanical handling and process control to increase freezing rate and reduce cost (George, 1993).
Today an increasing demand for frozen foods already exits and further expansion of the industry is primarily dependent on the ability of food processors to develop higher qualities in both process techniques and products. Improvements can only be achieved by focusing on new technologies and investigating poorly understood factors that influence the quality of frozen food products. Improvements in new and convenient forms of foods, as well as more information on relative cost and nutritive values of frozen foods, will contribute toward continued growth of the industry (Desrosier and Tressler, 1977).
Table 3. Frozen vegetables in terms of annual sales in
2001
(Source: Information Resources)
|
Vegetables |
Sales |
% Change |
|
Broccoli |
184 |
4.4 |
|
Com/Corn on the Cob |
312 |
3.5 |
|
Green Beans |
115 |
6.0 |
|
Mixed Vegetables |
450 |
7.2 |
|
Peas |
207 |
3.9 |
|
Potatoes |
1 070 |
4.4 |
Freezing is a widely used method of food preservation based on several advantages in terms of retention of food quality and ease of process. Beginning with the earliest history of freezing, the technology has been highly affected over the years by the developments and improvements in freezing techniques. In order to understand and handle the concepts associated with freezing of foods, it is necessary to examine the fundamental factors governing the freezing process.
Freezing has long been used as a method of preservation, and history reveals it was mostly shaped by the technological developments in the process. A small quantity of ice produced without using a "natural cold" in 1755 was regarded as the first milestone in the freezing process. Firstly, ice-salt systems were used to preserve fish and later on, by the late 1800’s, freezing was introduced into large-scale operations as a method of commercial preservation. Meat, fish, and butter, the main products preserved in this early example, were frozen in storage chambers and handled as bulk commodities (Persson and Lohndal, 1993).
In the following years, scientists and researchers continuously worked to achieve success with commercial freezing trials on several food commodities. Among these commodities, fruits were one of the most important since freezing during the peak growing season had the advantage of preserving fruit for later processing into jams, jellies, ice cream, pies, and other bakery foods. Although commercial freezing of small fruits and berries first began around 1905 in the eastern part of the United States, the commercial freezing of vegetables is much more recent. Starting from 1917, only private firms conducted trials on freezing vegetables, but achieving good quality in frozen vegetables was not possible without pre-treatments due to the enzymatic deterioration. In 1929, the necessity of blanching to inactivate enzymes before freezing was concluded by several researchers to avoid deterioration and off-flavours caused by enzymatic degradation.
The modern freezing industry began in 1928 with the development of double-belt contact freezers by a technologist named Clarence Birdseye. After the revolution in the quick freezing process and equipment, the industry became more flexible, especially with the usage of multi-plate freezers. The earlier methods achieved successful freezing of fish and poultry, however with the new quick freezing system, packaged foods could be frozen between two metal belts as they moved through a freezing tunnel. This improvement was a great advantage in the commercial large-scale freezing of fruits and vegetables. Furthermore, quick-freezing of consumer-size packages helped frozen vegetables to be accepted rapidly in late 1930s.
Today, freezing is the only large-scale method that bridges the seasons, as well as variations in supply and demand of raw materials such as meat, fish, butter, fruits, and vegetables. Besides, it makes possible movement of large quantities of food over geographical distances (Persson and Londahl, 1993). It is important to control the freezing process, including the pre-freezing preparation and post-freezing storage of the product, in order to achieve high-quality products (George, 1993). Therefore, the theory of the freezing process and the parameters involved should be understood clearly.
The freezing process mainly consists of thermodynamic and kinetic factors, which can dominate each other at a particular stage in the freezing process (Franks, 1985). Major thermal events are accompanied by reduction in heat content of the material during the freezing process as is shown in Figure 1. The material to be frozen first cools down to the temperature at which nucleation starts. Before ice can form, a nucleus, or a seed, is required upon which the crystal can grow; the process of producing this seed is defined as nucleation. Once the first crystal appears in the solution, a phase change occurs from liquid to solid with further crystal growth. Therefore, nucleation serves as the initial process of freezing, and can be considered as the critical step that results in a complete phase change (Sahagian and Goff, 1996).
Freezing point is defined as the temperature at which the first ice crystal appears and the liquid at that temperature is in equilibrium with the solid. If the freezing point of pure water is considered, this temperature will correspond to 0 °C (273°K). However, when food systems are frozen, the process becomes more complex due to the existence of both free and bound water. Bound water does not freeze even at very low temperatures. Unfreezable water contains soluble solids, which cause a decrease in the freezing point of water lower than 0 °C. During the freezing process, the concentration of soluble solids increases in the unfrozen water, resulting in a variation in freezing temperature. Therefore, the temperature at which the first ice crystal appears is commonly regarded as the initial freezing temperature. There are empirical equations in literature that can calculate the initial freezing temperature of certain foods as a function of their moisture content (Levy, 1979).
Figure 1. A schematic illustration of overall freezing process.

There are several methods of food freezing, and depending on the method used, the quality of the frozen food may vary. However, regardless of the method chosen, the main principle behind all freezing processes is the same in terms of process parameters. The International Institute of Refrigeration (IIR) has provided definitions to establish a basis for the freezing process. According to their definition, the freezing process is basically divided into three stages based on major temperature changes in a particular location in the product, as shown in Figures 2 and 3 for pure water and food respectively.
Beginning with the prefreezing stage, the food is subjected to the freezing process until the appearance of the first crystal. If the material frozen is pure water, the freezing temperature will be 0 °C and, up to this temperature, there will be a subcooling until the ice formation begins. In the case of foods during this stage, the temperature decreases to below freezing temperature and, with the formation of the first ice crystal, increases to freezing temperature. The second stage is the freezing period; a phase change occurs, transforming water into ice. For pure water, temperature at this stage is constant; however, it decreases slightly in foods, due to the increasing concentration of solutes in the unfrozen water portion. The last stage starts when the product temperature reaches the point where most freezable water has been converted to ice, and ends when the temperature is reduced to storage temperature (Persson and Lohndal, 1993).
The freezing time and freezing rate are the most important parameters in designing freezing systems. The quality of the frozen product is mostly affected by the rate of freezing, while time of freezing is calculated according to the rate of freezing. For industrial applications, they are the most essential parameters in the process when comparing different types of freezing systems and equipment (Persson and Lohndal, 1993).
Figure 2. Practical definition of the freezing process for pure water (Mallett, 1993).

Figure 3. Practical definition of the freezing process for foods (Mallett, 1993).

Again, freezing time is one of the most important parameters in the freezing process, defined as time required to lower product temperature from its initial temperature to a given temperature at its thermal center. Since the temperature distribution within the product varies during freezing process, the thermal center is generally taken as reference. Thus, when the geometrical center of the product reaches the given final temperature, this ensures the average product temperature has been reduced to a storage value. Freezing time depends on several factors, including the initial and final temperatures of the product and the quantity of heat removed, as well as dimensions (especially thickness) and shape of product, heat transfer process, and temperature. The International Institute of Refrigeration (1986) defines various factors of freezing time in relation to both the product frozen and freezing equipment (Persson and Lohndal, 1993). The most important are:
Dimensions and shape of product, particularly thickness
Initial and final temperatures
Temperature of refrigerating medium
Surface heat transfer coefficient of product
Change in enthalpy
Thermal conductivity of product
Calculation of freezing time in food systems is difficult in comparison to pure systems since the freezing temperature changes continuously during the process. Using a simplified approach, time elapsed between initial freezing until when the entire product is frozen can be regarded as the freezing time. Plank’s equation (Eq.1) is commonly used to estimate freezing time, however due to assumptions involved in the calculation it is only useful for obtaining an approximation of freezing time. The derivation of the equation starts with the assumption the product being frozen is initially at freezing temperature. Therefore, the calculated freezing time represents only the freezing period. The equation can be further modified for different geometries including slab, cylinder, and sphere, where for each geometry, the coefficients are arranged in relation to the dimensions (Plank, 1980).
Table 4. Coefficients P and R of Equation 1
|
Geometry |
P |
R |
Dimension |
|
Infinite slab |
1/2 |
1/8 |
thickness e |
|
Infinite cylinder |
1/4 |
1/16 |
radius r |
|
Sphere |
1/6 |
1/24 |
radius r |

(1)
where l1 is the latent heat of frozen fraction, k and r are the thermal conductivity and density of the frozen layer, while h is the coefficient of heat transfer by convection to the exterior. Tf denotes the body temperature of the product when introduced into a freezer in wich the external temperature is Te The coefficients R and P are given in Table 4 and arranged according to the geometry of the product frozen. where the letter e denotes the dimension (i.e. for infinite slab geometry, e is thickness of the slab and for infinite cylinder or sphere e is replaced by r which denotes the radius of the clylinder or sphere).
As mentioned earlier, the equation of Plank assumes the food is at a freezing temperature at the beginning of the freezing process. However, the food is usually at a temperature higher than freezing temperature. The real freezing time should therefore be the sum of time calculated from the equation of Plank and the time needed for the product’s surface to decrease from initial temperature to freezing temperature (Barbosa-Canovas and Ibarz, 2002).
Several works have attempted to calculate real freezing time, as in one presented by Nagaoka et al., (1955). Nagaoka’s equation (Eq. 2) calculates the amount of heat elimination required to decrease a product’s temperature from initial temperature to freezing temperature, as well as the amount of heat released during the phase change and the amount of heat eliminated to reach freezing temperature. Further empirical equations can be found in literature in detail (Chen, 1985; Levy, 1979; Succar and Hakayawa, 1983).
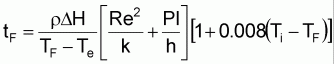
(2)
where Ti is the temperature of the food at the initiation of freezing, DH is the difference between the enthalpy of the food at initial temperature and end of freezing. Re and Pl are the dimensionless numbers, while k and h are the thermal conductivity and the coefficient of heat transfer, respectively.
For calculating freezing time of products with irregular shape, a common property of most food products - especially fruits and vegetables - a dimensionless factor has been employed in equations (Cleland et al., 1987a,b).
The freezing rate (°C/h) for a product or package is defined as the ratio of difference between initial and final temperature of product to freezing time. At a particular location within the product, a local freezing rate can be defined as the ratio of the difference between the initial temperature and desired temperature to the time elapsed in reaching the given final temperature (Persson and Lohndal, 1993). The quality of frozen products is largely dependent on the rate of freezing (Ramaswamy and Tung, 1984). Generally, rapid freezing results in better quality frozen products when compared with slow freezing. If freezing is instantaneous, there will be more locations within the food where crystallization begins. In contrast, if freezing is slow, the crystal growth will be slower with few nucleation sites resulting in larger ice crystals. Large ice crystals are known to cause mechanical damage to cell walls in addition to cell dehydration. Thus, the rate of freezing for plant tissues is extremely important due to the effect of freezing rate on the size of ice crystals, cell hydration, and damage to cell walls (Rahman, 1999). The figure 4 shows a general behavior of the dynamics curve of freezing preservation.
Rapid freezing is advantageous for freezing of many foods, however some products are susceptible to cracking when exposed to extremely low temperature for long periods. Several mechanisms, including volume expansion, contraction and expansion, and building of internal pressure, are proposed in literature explaining the mechanisms of product damage during freezing (Hung and Kim, 1996).
Figure 4. Freezing preservation dynamics curve.

For fruits and vegetables, the amount of energy required for freezing is calculated based on the enthalpy change and the amount of product to be frozen. The following equation is reported by Riedel (1949) for calculation of refrigeration requirements for fruits and vegetables.

(3)
XSNJ: Percentage of the product solids different
from juice (Dry matter fraction of the juice)
DHj: Enthalpy change during freezing of the juice
fraction
DT: Temperature difference between
initial and final temperature of the product
Refrigeration is defined as the elimination of heat from a material at a temperature higher than the temperature of its surroundings. The mechanism of refrigeration is a part of the freezing process and freezing storage involved in the thermodynamic aspects of freezing. According to the second law of thermodynamics, heat only flows from higher to lower temperatures. Therefore, in order to raise the heat from a lower to a higher temperature level, expenditure of work is needed. The aim of industrial refrigeration processes is to eliminate heat from low temperature points towards points with higher temperature. For this reason, either closed mechanical refrigeration cycles in which refrigeration fluids circulate, or open cryogenic systems with liquid nitrogen (LIN) or carbon dioxide (CO2), are commonly used by the food industry.
The main elements in a closed mechanical refrigeration system are the condenser, compressor, evaporator, and the expansion valve. The refrigerants hydrochlorofluorocarbon (HCFC) and ammonia are examples of the refrigerants circulated in these types of mechanical refrigeration systems. A simple scheme for the closed mechanical refrigeration system is shown in Figure 5.
Figure 5. A simple scheme for a one-stage closed mechanical
refrigeration system.
(Adapted from Stoecker, W.F. and Jones J.W.,
Refrigeration and Air Conditioning, McGraw-Hill, New York, 1982)

Starting at the suction point of the compressor, fluid in a vapor state is compressed into the compressor where an increase in temperature and pressure takes place. The fluid then flows through the condenser where it decreases in energy by giving off heat and converting to a liquid state. After the phase, a change occurs inside the condenser, the fluid flows through the expansion valve where the pressure decreases to convert liquid into a form of liquid-gas mixture. Finally, the liquid-gas mixture flows through the evaporator where it is converted into a saturated vapor state and removes heat from the environment in the process of cooling. With this last stage the loop restarts again.
The other refrigeration system employed by the food industry is the cryogenic system with carbon dioxide or liquid nitrogen. The refrigerant in this system is consumed differently from the circulating fluid in closed mechanical systems.
Refrigerants
There are several refrigerants available for refrigeration systems. The selection of a proper refrigerant is based on physical, thermodynamic, and chemical properties of the fluid. Environmental considerations are also important in refrigerant selection, since leaks within the system produce deleterious effects on the atmospheric ozone layer. Some refrigerants, including halocarbons, have been banned to avoid potential hazardous effects (Stoecker and Jones, 1982). For industrial applications, ammonia is commonly used, while chlorofluoromethane and tetrafluoroethane are also recommended as refrigerants (Persson and Lohndal, 1993).
Freezing equipment selection is based on the requirements for freezing a certain quantity of food per hour. For any type of freezer, freezing capacity (expressed in tonnes per hour) is defined as the ratio of the quantity of the product that can be loaded into the freezer to the holding time of the product in that particular freezer. The first parameter, the amount of food product loaded into the freezer, is affected by both the dimensions of the product and the mechanical constraints of the freezer. The denominator (holding time) has an important role in freezing systems and is based on the calculation of the amount of heat removed from the product per hour, which varies depending on the type of product frozen (Persson and Lohndal, 1993).
There is a variety of freezing systems available for freezing, and for most products, more than one type of freezer can be used. Therefore, in selecting a freezing system initially, a cost-benefit analysis should be conducted based on three important factors: economics, functionality, and feasibility. Financial considerations mainly involve capital investment and the production cost of selected equipment. Product losses during freezing operation should be included in cost estimation since generating higher cost freezers may have other benefits in terms of reducing product losses. Functional factors are mostly based on the suitability of the selected freezer for particular products. The mode of process, either in-line or batch, should be considered based on the fact that computerized systems are becoming more important for ease of handling and lowering production costs. Mechanical constraints for the freezer should also be considered since some types of freezers are not physically suitable for freezing certain products. Lastly, the feasibility of the process should be considered in terms of plant location or location of the processing area, as well as cleanability and hygienic design, and desired product quality (Johnston et al., 1994).
These factors and initial considerations can help eliminate several choices in freezer selection, but the relative importance of factors may change depending on the process. For developing countries where the freezing application is relatively new, the cost factor becomes more important than other factors due to the decreased production rates and need for lower capital investment costs.
The industrial equipment for freezing can be categorized in many ways, namely as equipment used for batch or in-line operation, heat transfer systems (air, contact, cryogenic), and product stability. The rate of heat transfer from the freezing medium to the product is important in defining the freezing time of the product. Therefore, the equipment selected for freezing process characterizes the rate of freezing.
The air blast freezer is one the oldest and commonly used freezing equipment due to its temperature stability and versatility for several product types. In general, air is used as the freezing medium in the freezing design, either as still air or forced air. Freezing is accomplished by placing the food in freezing rooms called sharp freezers. Still, air freezing is the cheapest way of freezing and has the added advantage of a constant temperature during frozen storage, which allows usage for unprocessed bulk products like beef quarters and fish. However, it is the slowest method of freezing due to the low surface heat transfer coefficient of circulating air inside the room. Freezing time in sharp freezers is largely dependent on the temperature of the freezing chamber and the type, initial temperature, and size of product (Desrosier and Desrosier 1977). An improved version of the still air freezer is the forced air freezer, which consists of air circulation by convection inside the freezing room. However, even modification of the sharp freezer with extra refrigeration capacity and fans for increased air circulation does not help control the air flow over the products during slow freezing. A typical design for air blast freezers is shown in Figure 6.
There are a considerable number of designs and arrangements for air blast freezers, primarily grouped in two categories depending on the mode of process, as either inline or batch. Continuous freezers are the most suitable systems for mass production of packaged products with similar freezing times, in which the product is carried through on trucks or on conveyors. The system works on a semi-batch principle when trucks are used, since they remain stationary during the process except when a new truck enters one end of the tunnel, thus moving the others along to release a finished one at the exit. The batch freezers are more flexible since a variety of products can be frozen at the same time on individual trolleys. Over-loading may be a problem for these types of freezers, thus the process requires closer supervision than continuous systems.
Figure 6. Air blast freezer.

Tunnel freezers
In tunnel freezers, the products on trays are placed in racks or trolleys and frozen with cold air circulation inside the tunnel. In order to allow air circulation, optimum space is provided between layers of trolley, which can be moved continuously in and out of the freezer manually or by forklift trucks. This freezing system is suitable for all types of products, although there are some mechanical constraints including the requirement of high manpower for handling, cleaning, and transportation of trays (Mallett, 1993). A trolley for a tunnel freezer is shown in Figure 7.
Figure 7. Trolley in a tunnel freezer.

Belt freezers
Belt freezers were first designed to provide continuous product flow with the help of a wire mesh conveyor inside the blast rooms. A poor heat transfer mechanism and the mechanical problems were solved in modern belt freezers by providing a vertical airflow to force air through the product layer. Airflow has good contact with the product only when the entire product is evenly distributed over the conveyor belt. In order to decrease required floor space, the belts can be arranged in a multi-tier belt freezer or a spiral belt freezer. Spiral belt freezers consist of a belt that can be bent laterally around a rotating drum to maximize belt surface area in a given floor space. This type of design has the advantage of eliminating product damage in transfer points, especially for products that require gentle handling (Mallett, 1993). Both packed and unpacked products with long freezing times (10 min to 3 hr) can be frozen in spiral belt freezers due to the flexibility of the equipment (ASHRAE, 1994). A typical spiral belt freezer is shown in Figure 8.
Fluidized bed freezers
The fluidized bed freezer, a fairly recent modified type of air-blast freezer for particular product types, consists of a bed with a perforated bottom through which cold air is blown vertically upwards (Rahman, 1999). The system relies on forced cold air from beneath the conveyor belt, causing the products to suspend or float in the cold air stream (George, 1993). The use of high air velocity is very effective for freezing unpacked foods, especially when they can be completely surrounded by flowing air, as in the case of fluidized bed freezers.
Figure 8. The cross-section view of a spiral belt
freezer.
(Courtesy of Frigoscandia Equipment Ltd., UK)

Figure 9a. Cross-sectional view of a fluidized bed
freezer.
(Courtesy of Frigoscandia Equipment Ltd., UK)

The use of fluidization has several advantages compared with other methods of freezing since the product is individually quick frozen (IQF), which is convenient for particles with a tendency to stick together (Persson and Lohndal, 1993). The idea of individually quick frozen foods (IQF) started with the first technological developments aimed at quick freezing. The need for an effective means of freezing small particles with the potential for lumping during the process is the objective of IQF freezing. Small vegetables, prawns, shrimp, french-fried potatoes, diced meat, and fruits are some of the products now frozen with this technology. A typical fluidized-bed freezer is shown in Figures 9a and 9b.
Figure 9b. Simple working principle of a fluidized bed
freezer.
(Saravacos, G.D., Kostaropoulos, A.E., 2002)

Contact freezing is the one of the most efficient ways of freezing in terms of heat transfer mechanism. In the process of freezing, the product can be in direct or indirect contact with the freezing medium. For direct contact freezers, the product being frozen is fully surrounded by the freezing medium, the refrigerant, maximizing the heat transfer efficiency. A schematic illustration is given in Figure10. For indirect contact freezers, the product is indirectly exposed to the freezing medium while in contact with the belt or plate, which is in contact with the freezing medium (Mallett, 1993).
Figure 10. Direct contact freezer.

Immersion freezers
The immersion freezer consists of a tank with a cooled freezing media, such as glycol, glycerol, sodium chloride, calcium chloride, and mixtures of salt and sugar. The product is immersed in this solution or sprayed while being conveyed through the freezer, resulting in fast temperature reduction through direct heat exchange (Hung and Kim, 1996). Direct immersion of a product into a liquid refrigerant is the most rapid way of freezing since liquids have better heat conducting properties than air. The solute used in the freezing system should be safe without taste, odour, colour, or flavour, and for successful freezing, products should be greater in density than the solution. Immersion freezing systems have been commonly used for shell freezing of large particles due to the reducing ability of product dehydration when the outer layer is frozen quickly. A commonly seen problem in these freezing systems is the dilution of solution with the product, which can change the concentration and process parameters. Thus, in order to avoid product contact with the liquid refrigerant, flexible membranes can be used (George, 1993). A simple illustration of the immersion freezer is shown in Figure 11.
Figure 11. Simple illustration of a typical immersion freezer (Fellows, 2000).

Figure 12. Indirect contact freezer.

Indirect contact freezers
In this type of freezer, materials being frozen are separated from the refrigerant by a conducting material, usually a steel plate. The mechanism of indirect contact freezer is shown in Figure 12. Indirect contact freezers generally provide an efficient medium for heat transfer, although the system has some limitations, especially when used for packaged foods due to resistance of package to heat transfer. Additionally, corrosive effects may occur due to interaction of metal packages with heat transfer surfaces.
Figure 13a. Pressure application in a plate freezer.

Plate freezers
The most common type of contact freezer is the plate freezer. In this case, the product is pressed between hallow metal plates, either horizontally or vertically, with a refrigerant circulating inside the plates. Pressure is applied for good contact as schematically shown in Figure13a.
Figure 13b. Plate freezer with a two-stage compressor and
sea water condenser
(Courtesy of DSI Samifi Freezers
S.r.I.)

This type of freezing system is only limited to regular-shaped materials or blocks like beef patties or block-shaped packaged products. A typical plate freezer is shown in Figure 13b.
Contact belt freezers
This type of freezer is designed with single-band or double-band for freezing of thin product layers as shown in Figure 14. The design can be either straight forward or drum. Typical products frozen in belt freezers are, fruit pulps, egg yolk, sauces and soups (Persson and Lohndall, 1993).
Cryogenic freezing is a relatively new method of freezing in which the food is exposed to an atmosphere below -60 °C through direct contact with liquefied gases such as nitrogen or carbon dioxide (Hung and Kim, 1996). This type of system differs from other freezing systems since it is not connected to a refrigeration plant; the refrigerants used are liquefied in large industrial installations and shipped to the food-freezing factory in pressure vessels. Thus, the small size and mobility of cryogenic freezers allow for flexibility in design and efficiency of the freezing application. Low initial investment and rather high operating costs are typical for cryogenic freezers (Persson and Lohndal, 1993).
Figure 14. Contact belt freezer.
(Courtesy of
Frigoscandia Equipment Ltd., UK)

Liquid Nitrogen freezers
Liquid nitrogen, with a boiling temperature of -196 °C at atmospheric pressure, is a by-product of oxygen manufacture. The refrigerant is sprayed into the freezer and evaporates both on leaving the spray nozzles and on contact with the products. The system is designed in a way that the refrigerant passes in counter current to the movement of the products on the belt giving high transfer efficiency. The refrigerant consumption is in the range of 1.2-kg refrigerant per kg of the product. Typical food products used in this system are, fish fillets, seafood, fruits, berries (Persson and Lohndal, 1993).
Liquid carbon dioxide freezers
Liquid carbon dioxide exists as either a solid or gas when stored at atmospheric pressure. When the gas is released to the atmosphere at -70 °C, half of the gas becomes dry-ice snow and the other half stays in the form of vapor. This unusual property of liquid carbon dioxide is used in a variety of freezing systems, one of which is a pre-freezing treatment before the product is exposed to nitrogen spray (George, 1993).
Proper packaging of frozen food is important to protect the product from contamination and damage while in transit from the manufacturer to the consumer, as well as to preserve food value, flavour, colour, and texture. There are several factors considered in designing a suitable package for a frozen food. The package should be attractive to the consumer, protected from external contamination, and effective in terms of processing, handling, and cost (Rahman, 1999). Proper selection is based on the type of package and material. There are typically three types of packaging used for frozen foods: primary, secondary, and tertiary. The primary package is in direct contact with the food and the food is kept inside the package up to the time of use. Secondary packaging is a form of multiple packaging used to handle packages together for sale. Tertiary packaging is used for bulk transportation of products (Harrison and Croucher, 1993).
Packaging materials should be moisture-vapor-proof to prevent evaporation, thus retaining the highest quality in frozen foods. Oxygen should also be completely evacuated from the package using a vacuum or gas-flush system to prevent migration of moisture and oxygen (ASHRAE, 1994; Sebranek, 1996). Glass and rigid plastic are examples of moisture-vapor-proof packaging materials. Many packaging materials, however, are not moisture-vapor-proof, but are sufficiently moisture-vapor-resistant to retain satisfactory quality in foods. Most bags, wrapping materials, and waxed cartons used in freezing packaging are moisture-vapor-resistant. In general, the containers should be leakage free while easy to seal. Durability of the material is another important factor to consider, since the packaging material must not become brittle at low temperatures and crack (MSU, 1999).
A range of different packaging materials, mainly grouped as rigid and non-rigid containers, can be used for primary packaging. Glass, plastic, tin, and heavily waxed cardboard materials are in the rigid container group and usually used for packaging of liquid food products. Glass containers are mostly used for fruits and vegetables if they are not water-packed. Plastics are the derivatives of the oil-cracking industry (Brydson, 1982). Non-rigid containers include bags and sheets made of moisture-vapor-resistant heavy aluminum foil, polyethylene or laminated papers. Bags are the most commonly used packaging materials for frozen fruits and vegetables due to their flexibility during processing and handling (Harrison and Croucher, 1993). They can be used with or without outer cardboard cartons to protect against tearing.
Shape and size of the container are also important factors in freezing products. Serving size may vary depending on the type of product and selection should be based on the amount of food determined for one meal. For shape of the container, freezer space must be considered since rigid containers with flat tops and bottoms stack well in the freezer, while round containers waste freezer space.
The quality of the final product depends on the history of the raw material. Using the lowest possible temperature is essential for frozen storage, transport, and distribution in achieving a high-quality product, since deteriorative processes are mainly temperature dependent. The lower the product temperature is, the slower the speed of reaction is leading to loss of quality. The temperatures of supply chains in freezing applications from the factory to the retail cabinet should be carefully monitored. The temperature regime covering the freezing process, the cold-store temperatures (£ -18 °C), distribution temperatures (£ -15 °C), and retail display (£ -12 °C) are given as legal standards (Harrison and Croucher, 1993).
The preservation of fruits and vegetables by freezing is one of the most important methods for retaining high quality in agricultural products over long-term storage. In particular, the freshness qualities of raw fruits and vegetables can be retained for long periods, extending well beyond the normal season of most horticultural crops (Arthey, 1993). The potential application of freezing preservation of fruits and vegetables, including tropical products, has been increasing recently in parallel with developments in developing countries. Freezing of fruits and vegetables in small and medium scale operations is detailed in the following sections and a general flowchart is shown in Figure 15.
Figure 15. A general flow chart of frozen fruits and vegetables (Mallett, 1993).

The effect of freezing, frozen storage, and thawing on fruit quality has been investigated over several decades. Today frozen fruits constitute a large and important food group (Skrede, 1996). The quality demanded in frozen fruit products is mostly based on the intended use of the product. If the fruit is to be eaten without any further processing after thawing, texture characteristics are more important when compared to use as a raw material in other industries. In general, conventional methods of freezing tend to destroy the turgidity of living cells in fruit tissue. Different from vegetables, fruits do not have a fibrous structure that can resist this destructive effect. Additionally, fruits to be frozen are harvested in a fully ripe state and are soft in texture. On the contrary, a great number of vegetables are frozen in an immature state (Boyle et al., 1977). Fruits have delicate flavours that are easily damaged or changed by heat, indicating they are best eaten when raw and decrease in quality with processing. In the same way, attractive colour is important for frozen fruits. Chemical treatments or additives are often used to inactivate the deteriorative enzymes in fruits. Therefore, proper processing is essential for all steps involved, from harvesting to packaging and distribution. A freezing guide for freezing fruits is shown in table 5.
The characteristics of raw materials are of primary importance in determining the quality of the frozen product. These characteristics include several factors such as genetic makeup, climate of the growing area, type of fertilization, and maturity of harvest (Boyle et al., 1977).
The ability to withstand rough handling, resistance to virus diseases, molds, uniformity in ripening, and yield are some of the important characteristics of fruits in terms of economical aspects considered in production. The use of mechanical harvesting generally causes bruising of fruits and results in a wide range of maturity levels for fruits. In contrast, hand-picking provides gentler handling and maturity sorting of fruits. However in most cases, it is non-economical compared to mechanical harvesting due to high labor cost (Boyle et al., 1977).
As a rule, harvesting of fruits at an optimum level for commercial use is difficult. Simple tests like pressure tests are applied to determine when a fruit has reached optimum maturity for harvest. Colour is also one of the characteristics used in determining maturity since increased maturation causes a darker colour in fruits. A combination of colour and pressure tests is a better way to assess maturity level for harvesting (Skrede, 1996).
Controlled atmosphere storage is a common method of storage for some fruits prior to freezing. In principle, a controlled atmosphere high in carbon dioxide and low in oxygen content slows down the rate of respiration, which may extend shelf life of any respiring fruit during storage. Due to the fact that these fruits do not ripen appreciably after picking, most fruits are picked as near to eating-ripe maturity as possible.
Freezing preservation of fruits can only help retain the inherent quality present initially in a product since the process does not improve the quality characteristics of raw materials. Therefore, quality level of the raw materials prior to freezing is the major consideration for successful freezing. Washing and cutting generally results in losses when applied after thawing. Thus, fruits should be prepared prior to the freezing process in terms of peeling, slicing or cutting. Freezing preservation does not require specific unit operations for cleaning, rinsing, sorting, peeling, and cutting of fruits (Spiess, 1984).
Fruits that require peeling before consumption should be peeled prior to freezing. Peeling is done by scalding the fruit in hot water, steam or hot lye solutions (Boyle and Wolford, 1968). The effect of peeling on the quality of frozen products has been studied for several fruits, including kiwi (Robertson, 1985), banana (Cano et al., 1990), and mango (Cano and Marín, 1992). The rate of freezing can be increased by decreasing the size of products frozen, especially for large fruits. An increase in the freezing rate results in smaller ice crystals, which decreases cellular damage in fruit tissue. Banana, tomato, mango, and kiwi are some examples of large fruits commonly cut into smaller cubes or slices prior to freezing (Skrede, 1996).
The objective of blanching is to inactivate the enzymes causing detrimental changes in colour, odour, flavour, and nutritive value, but heat treatment causes loss of such characteristics in fruits (Gutschmidt, 1968). Therefore, only a few types of fruits are blanched for inactivation of enzymes prior to freezing. The loss of water-soluble minerals and vitamins during blanching should also be minimized by keeping blanching time and temperature at an optimum combination (Spiess, 1984).
Addition of sugars is an extremely important pretreatment for fruits prior to freezing since the treatment has the effect of excluding oxygen from the fruit, which helps to retain colour and appearance. Sugars when dissolved in solutions act by withdrawing water from cells by osmosis, resulting in very concentrated solutions inside the cells. The high concentration of solutes depresses the freezing point and therefore reduces the freezing within the cells, which inhibits excessive structural damage (Munoz-Delgado, 1978). Sugar syrups in the range of 30-60 percent sugar content are commonly used to cover the fruit completely, acting as a barrier to oxygen transmission and browning. Several experiments have shown the protective effect of sugar on flavour, odour, colour, and nutritive value during freezing, especially for frozen berries (Gutschmidt, 1968).
Fruits exposed to oxygen are susceptible to oxidative degradation, resulting in browning and reduced storage life of products (Munoz-Delgado, 1978; Tomassicchio et al., 1986). Therefore, packaging of frozen fruits is based on excluding air from the fruit tissue. Replacement of oxygen with sugar solution or inert gas, consuming the oxygen by glucose-oxidase and/or the use of vacuum and oxygen-impermeable films are some of the methods currently employed for packaging frozen fruits. Plastic bags, plastic pots, paper bags, and cans are some of the most commonly used packaging materials (with or without oxygen removal) selected, based on penetration properties and thickness (Gradziel, 1988).
There are several types of fruit packs suitable for freezing: syrup pack, sugar pack, unsweetened pack, and tray pack and sugar replacement pack. The type of pack is usually selected according to the intended use for the fruit. Syrup-packed fruits are generally used for cooking purposes, while dry-packed and tray-packed fruits are good for serving raw in salads and garnishes.
Syrup pack
The proportion of sugar to water used in a syrup pack depends on the sweetness of the fruit and the taste preference of the consumer. For most fruits, 40 percent sugar syrup is recommended. Lighter syrups are lower in calories and mostly desirable for mild-flavoured fruits to prevent masking the flavour, while heavier syrups may be used for very sour fruits (Kendall, 2002).
Syrup is prepared by dissolving the sugar in warm water and cooling the solution down before usage. Just enough cooled syrup is used to cover the prepared fruit after it has been settled by jarring the container. In order to keep the fruit under the syrup, a small piece of crumpled waxed paper or other water resistant wrapping material is placed on top; the fruit is pressed down into the syrup before closing, then sealed and frozen (Beck, 1996).
Pectin can be used to reduce sugar content in syrups when freezing berries, cherries, and peaches. Pectin syrups are prepared by dissolving 1 box of powdered pectin with 1 cup of water. The solution is stirred and boiled for 1 minute; 1/2 cup of sugar is added and dissolved; the solution is then cooled down with the addition of cold water. Previously prepared fruit is put into a 4 to 6 quart bowl and enough pectin syrup is added to cover the fruit with a thin film. The pack is sealed and promptly frozen (Brady, 2002).
Sugar packs
In preparing a sugar pack, sugar is first sprinkled over the fruit. Then the container is agitated gently until the juice is drawn out and the sugar is dissolved. This type of pack is generally used for soft sliced fruits such as peaches, strawberries, plums, and cherries, by using sufficient syrup to cover the fruit. Some whole fruits may also be coated with sugar prior to freezing (Beck, 1996).
Unsweetened packs
Unsweetened packs can be prepared in several ways, either dry-packed, covered with water containing ascorbic acid, or packed in unsweetened juice. When water or juice is used in syrup and sugar packs, fruit is submerged by using a small piece of crumpled water-resistant material. Generally, unsweetened packs yield a lower quality product when compared with sugar packs, with the exception, some fruits such as raspberries, blueberries, scalded apples, gooseberries, currants, and cranberries maintain good quality without sugar (Beck, 1996).
Tray packs
Unsweetened packs are generally prepared by using tray packs in which a single layer of prepared fruit is spread on shallow trays, frozen, and packaged in freezer bags promptly. The fruit sections remain loose without clumping together, which offers the advantage of using frozen fruit piece by piece.
Sugar replacement packs
Artificial sweeteners can be used instead of sugar in the form of sugar substitutes. The sweet taste of sugar can be replaced by using these kinds of sweeteners, however the beneficial effects of sugar like colour protection and thick syrup can not be replaced. Fruits frozen with sugar substitutes will freeze harder and thaw more slowly than fruits preserved with sugar (Beck, 1996).
Freezing is often considered the simplest and most natural way of preservation for vegetables (Cano, 1996). Frozen vegetables and potatoes form a significant proportion of the market in terms of frozen food consumption (Mallett, 1993). The quality of frozen vegetables depends on the quality of fresh products, since freezing does not improve product quality. Pre-process handling, from the time vegetables are picked until ready to eat, is one of the major concerns in quality retention.
Table 5. Fruit freezing guide
(Kendall, Colorado
State University, Cooperative Extension, 2002)
|
Fruit |
Preparation |
Type of Pack |
|
Apples |
Wash, peel, and slice into antidarkening solution - 3 tablespoons lemon juice per quart of water |
Pack in 30-40% syrup, adding 1/2 teaspoon crystalline ascorbic
acid per quart of syrup. |
|
Apricots |
Wash, halve, and pit. |
Pack in 40% syrup, adding 3/4 teaspoon crystalline ascorbic acid per quart of syrup. |
|
Avocados |
Peel soft, ripe avocados. |
Add 1/8 teaspoon crystalline ascorbic acid to each quart of puree. Package in recipe-size amounts. |
|
Berries |
Select firm, fully ripe berries. |
Use 30% syrup pack, dry unsweetened pack, dry sugar pack, (3/4 cup sugar per quart of berries), or tray pack. |
|
Cherries |
Select well-colored, tree-ripened cherries. |
Pack in 30-40% syrup. Add 1/2 teaspoon ascorbic acid per quart of syrup. For pies and other cooked products, pack in dry sugar using 3/4-cup sugar per quart of fruit. |
|
Citrus fruits, |
Select firm fruit, free of soft spots. Wash and peel. |
Pack in 40% syrup or in fruit juice. Add 1/2 teaspoon ascorbic acid per quart of syrup or juice. |
|
Grapes |
Select firm, ripe grapes. Wash and remove stems. Leave
seedless grapes whole. |
Pack in 20% syrup or pack without sugar. Use dry pack for halved grapes and tray pack for whole grapes. |
|
Melons |
Select firm-fleshed, well-colored, ripe melons. Wash rinds
well. |
Pack in 30% syrup or pack dry using no sugar. Pulp also may be crushed (except watermelon), adding 1 tablespoon sugar per quart. Freeze in recipe-size containers. |
The choice of the right cultivar and maturity before crop is harvested are the two most important factors affecting raw material quality. Raw material characteristics are usually related to the vegetable cultivar, crop production, crop maturity, harvesting practices, crop storage, transport, and factory reception.
The choice of crop cultivars is mostly based on their suitability for frozen preservation in terms of factory yield and product quality. Some of the characteristics used as selection criteria are as follows (Cano, 1996):
Suitability for mechanical harvesting
Uniform maturity
Exceptional flavour and uniform colour and desirable texture
Resistance to diseases
High yield
Although cultivar selection is a major factor affecting the quality of the final product, many practices in the field and factors during growth of crop can also have a significant effect on quality. Those practices include site selection for growth, nutrition of crop, and use of agricultural chemicals to control pests or diseases. The maturity assessment for harvesting is one of the most difficult parts of the production. In addition to conventional methods, new instruments and tests have been developed to predict the maturity of crops that help determining the optimum harvest time, although the maturity assessment differs according to crop variety (Hui et al., 2004).
At optimum maturity, physiological changes in several vegetables take place very rapidly. Thus, the determination of optimum harvesting time is critical (Arthey, 1993). Some vegetables such as green peas and sweet corn only have a short period during which they are of prime quality. If harvesting is delayed beyond this point, quality deteriorates and the crop may quickly become unacceptable (Lee, 1989). Most of the vegetables are subjected to bruising during harvesting.
Vegetables at peak flavour and texture are used for freezing. Postharvest delays in handling vegetables are known to produce deterioration in flavour, texture, colour, and nutrients (Lee, 1989). Therefore, the delays between harvest and processing should be reduced to retain fresh quality prior to freezing. Cooling vegetables by cold water, air blasting, or ice will often reduce the rate of post-harvest losses sufficiently, providing extra hours of high quality retention for transporting raw material to considerable distances from the field to the processing plant (Deitrich et al., 1977).
Blanching is the exposure of the vegetables to boiling water or steam for a brief period of time to inactivate enzymes. Practically every vegetable (except herbs and green peppers) needs to be blanched and promptly cooled prior to freezing, since heating slows or stops the enzyme action, which causes vegetables to grow and mature. After maturation, however, enzymes can cause loss in quality, flavour, colour, texture, and nutrients. If vegetables are not heated sufficiently, the enzymes will continue to be active during frozen storage and may cause the vegetables to toughen or develop off-flavours and colours. Blanching also causes wilting or softening of vegetables, making them easier to pack. It destroys some bacteria and helps remove any surface dirt (Desrosier and Tressler, 1977).
Blanching in hot water at 70 to 105 °C has been associated with the destruction of enzyme activity. Blanching is usually carried out between 75 and 95 °C for 1 to 10 minutes, depending on the size of individual vegetable pieces (Holdsworth, 1983). Blanched vegetables should be promptly cooled down to control and minimize the degradation of soluble and heat-labile nutrients (Deitrich et al., 1977).
The enzymes used as indicators of effectiveness of the blanching treatment are peroxidase, catalase, and more recently lipoxygenase. Peroxidase inactivation is commonly used in vegetable processing, since peroxidase is easily detected and is the most heat stable of these enzymes (Arthey, 1993).
Vegetables can be blanched in hot water, steam, and in the microwave. Hot water blanching is the most common way of processing vegetables. Blanching times recommended for various vegetables are given in Table 6, which indicates that the operation time can vary depending on the intended product use. For water blanching, vegetables are put in a basket and then placed in a kettle of boiling water covered with a lid. Timing begins immediately (Archuleta, 2003). Steam blanching takes longer than the water method, but helps retain water-soluble nutrients such as water-soluble vitamins. For steam blanching, a single layer of vegetables is placed on a rack or in a basket at 3-5 cm above water boiling in a kettle. A tightly fitted lid is placed on the kettle and timing is started. Microwave blanching is usually recommended for small quantities of vegetables prior to freezing. Due to the non-uniform heating disadvantage of microwaves, research is still being conducted to obtain better results with microwave blanching.
Table 6. Vegetable freezing guide (Archuleta, 2003)
|
Vegetable |
Preparation |
Blanch/Freeze |
|
|
Asparagus |
Wash and sort by size. |
Water blanch: |
2 min |
|
Steam blanch: |
3 min |
||
|
Beans |
Wash and trim the ends. |
Water blanch: |
Steam blanch: |
|
Whole: 3 min. |
Whole: 4 min. |
||
|
Cut: 2min. |
Cut: 3min. |
||
|
Beets |
Wash and remove the tops leaving 2.5 cm of stem and root. |
Cook until tender: 25-30 min |
|
|
Broccoli |
Wash and cut into pieces. |
Water blanch: |
3 min. |
|
Steam blanch: |
3 min. |
||
|
Cabbage |
Wash and cut into wedges. |
Water blanch: |
3 min. |
|
Steam blanch: |
4 min. |
||
|
Carrots |
Wash, peel and trim. |
Water blanch: 5 min. |
|
|
Cauliflower |
Discard leaves; steam and wash. |
Water blanch: |
Steam blanch: |
|
Whole: 5 min. |
Whole: 7 min |
||
|
Corn |
Remove husks and silks. |
Water blanch: |
Steam blanch: |
|
Whole: 5 min. |
Whole: 7 min |
||
|
Greens |
Select young tender greens. |
Water blanch: |
2 min. |
|
Steam blanch: |
3 min. |
||
|
Herbs |
Wash. |
No heat treatment is needed. |
|
|
Mushrooms |
Wipe and damp with paper towel. |
May be frozen without heat treatment. |
|
|
Peas |
Shell garden peas. |
Water blanch: |
Steam blanch: |
|
1-1/2 min. |
1-1/2 min. |
||
|
Peppers |
Wash, remove stems and seeds. |
Freeze whole or cut as desired. No heat treatment is needed. |
|
|
Potatoes |
Peel, cut or grate as desired. |
Water blanch: |
|
|
Whole: 5 min. |
|
||
|
Pieces: 2-3 min. |
|
||
There are several factors to consider in packaging frozen vegetables, which include protection from atmospheric oxygen, prevention of moisture loss, retention of flavour, and rate of heat transfer through the package (Arthey, 1996). There are two basic packing methods recommended for frozen vegetables: dry pack and tray pack.
In the dry pack method, the blanched and drained vegetables are put into meal-sized freezer bags and packed tightly to cut down on the amount of air in the package. Proper headspace (approximately 2 cm) is left at the top of rigid containers before closing. For freezer bags, the headspace is larger. Provision for headspace is not necessary for foods such as broccoli, asparagus, and brussels sprouts, as they do not pack tightly in containers (Kendall, 2002).
In the tray pack method, chilled, well-drained vegetables are placed in a single layer on shallow trays or pans. Trays are placed in a freezer until the vegetables become firm, then removed. Vegetables are filled into containers. Tray-packed foods do not freeze in a block but remain loosely distributed so that the amount needed can be poured from the container and the package reclosed (Kendall, 2002).
The technical approach of chapter 1 focuses mainly on the large scale freezing industry. However, it is important to highlight that all preliminary steps before freezing food products are quite similar whether on a large or small scale. Furthermore, chapter 3 focuses more on the more suitable approach for small food freezing industry.