
IDENTITY
Biological features
The giant tiger prawn inhabits the coasts of Australia, South East Asia, South Asia and East Africa.
Similar to all penaeid shrimp, the rostrum well developed and toothed dorsally and ventrally. Carapace without longitudinal or transverse sutures. Cervical and orbito-antennal sulci and antennal carinae always present. Hepatic and antennal spines pronounced. Pterygostomain angle round. Stylocerite at first antennular segment. Basial spines on first and second pereiopods and exopods on the first to fourth pereiopods usually present. No fixed subapical spines on telson. Adrostral sulcus and carina are short, not reaching posteriorly beyond midlength of carapace. Gastrofrontal carina absent. Females have closed-type thelycum. Petasma in male symmetrical with thin median lobes. The most distinct features for identification of this species are: fifth pereiopods without exopod; hepatic carina horizontally straight; and gastroorbital carina occupying the posterior half of the distance between hepatic spine and postorbital margin of carapace. Depending on substratum, feed and water turbidity, body colours vary from green, brown, red, grey, blue and transverse band colours on abdomen and carapace are alternated between blue or black and yellow. Adults may reach 33 cm in length and females are commonly larger than males.
Similar to all penaeid shrimp, the rostrum well developed and toothed dorsally and ventrally. Carapace without longitudinal or transverse sutures. Cervical and orbito-antennal sulci and antennal carinae always present. Hepatic and antennal spines pronounced. Pterygostomain angle round. Stylocerite at first antennular segment. Basial spines on first and second pereiopods and exopods on the first to fourth pereiopods usually present. No fixed subapical spines on telson. Adrostral sulcus and carina are short, not reaching posteriorly beyond midlength of carapace. Gastrofrontal carina absent. Females have closed-type thelycum. Petasma in male symmetrical with thin median lobes. The most distinct features for identification of this species are: fifth pereiopods without exopod; hepatic carina horizontally straight; and gastroorbital carina occupying the posterior half of the distance between hepatic spine and postorbital margin of carapace. Depending on substratum, feed and water turbidity, body colours vary from green, brown, red, grey, blue and transverse band colours on abdomen and carapace are alternated between blue or black and yellow. Adults may reach 33 cm in length and females are commonly larger than males.
PROFILE
Historical background
Shrimp farming has been practiced for more than a century for food and the livelihood of coastal people in some Asian countries, such as Indonesia, the Philippines, Taiwan Province of China, Thailand and Viet Nam. Penaeus monodon was originally harvested together with other shrimp species from traditional trapping-growing ponds or as a significant by-product of extensive milkfish ponds. From 1970-1975, research on breeding was conducted and monoculture techniques in small ponds were gradually developed at the Tungkang Marine Laboratory in Taiwan Province of China and partly at the IFREMER (Centre Océanologique du Pacifique) in Tahiti in the South Pacific. In Thailand, extensive and semi-intensive farms were commercially established in 1972 and 1974 respectively, after the first success in breeding P. monodon at Phuket Fisheries Station in 1972. Between 1980 and 1987 there was a boom of small-scale intensive farms in Taiwan Province of China due to commercial success in formulated feed development, mainly to produce shrimp for export to Japan. However, it is believed that a viral disease outbreak caused the collapse of the industry in Taiwan Province of China in 1987-1988. This led Thailand, encouraged by extremely high prices in the Japanese market due to supply shortages, to replace Taiwan Province of China as the world's leading producer of farm-raised P. monodon in 1988. Later, the culture of this species spread throughout southeast and south Asia, as it can grow-up to a large size (40-60 g) with high value and demand in the international market. The locally adapted culture technology has allowed Thai farmers to overcome serious disease, environmental and trade problems and maintain its status as a leading producer.
The introduction or importation of wild broodstock is commonly practiced among the major producing countries because local supplies are insufficient and domestication technology has not yet been commercially developed. However, disease-free broodstock are highly desirable and some countries require health certification of imported stock.
The introduction or importation of wild broodstock is commonly practiced among the major producing countries because local supplies are insufficient and domestication technology has not yet been commercially developed. However, disease-free broodstock are highly desirable and some countries require health certification of imported stock.
Main producer countries

Main producer countries of Penaeus monodon (FAO Fishery statistics, 2006)
Habitat and biology
Penaeus monodon mature and breed only in tropical marine habitats and spend their larval, juvenile, adolescent and sub-adult stages in coastal estuaries, lagoons or mangrove areas. In the wild, they show marked nocturnal activity, burrowing into bottom substratum during the day and emerging at night to search for food as benthic feeders. Under natural conditions, the giant tiger prawn is more of a predator than an omnivorous scavenger or detritus feeder than other penaeid shrimp. After moulting, the new shell is still soft which causes prawns to become vulnerable and they may subsequently be eaten by their predators or companions. Adults are often found over muddy sand or sandy bottoms at 20-50 m depth in offshore waters. Wild males posses spermatozoa from around 35 g body weight and females becomes gravid from 70 g. Mating occurs at night, shortly after moulting while the cuticle is still soft, and sperm are subsequently kept in a spermatophore (sac) inserted inside the closed thelycum of the female. There are five stages in ovarian maturation; undeveloped, developing; nearly ripe; ripe; and spent. P. monodon females are highly fecund with gravid females producing as many as 500 000 to 750 000 eggs. Spawning occurs at night and fertilization is external with females suddenly extruding sperm from the thelycum as eggs are laid in offshore waters. Hatching occurs 12-15 hours after fertilization. The larvae, termed nauplii, are free swimming and resemble tiny aquatic spiders. This first stage in larval development does not feed but lives on its yolk reserve and passes rapidly through six moults. The next larval stages [protozoea, mysis and early postlarvae (PL) respectively] remain planktonic for some time and are carried towards the shore by tidal currents. Protozoea, which have feathery appendages and elongated bodies, moult three times and then metamorphose into the mysis stage. Mysis, which have segmented bodies, eyestalk and tails characteristic of adult shrimp, also moult three times before metamorphosing into PL with similar characteristics to adult shrimp. The PL subsequently change their habit to feed on benthic detritus, polychaete worms and small crustaceans after PL 6. Juvenile and adolescent stages can tolerate salinity conditions as low as 1-2‰.
PRODUCTION

Due to their larger size and better survival, captured wild seeds were used commonly in south Asia for extensive ponds, which require a minimal amount of seed for stocking. However, the use of wild seeds has been reduced, due to overfishing and the outbreak of white spot disease in shrimp nursery grounds. Therefore most Penaeus monodon grow-out farms now rely solely on hatchery-produced seeds.
Broodstock
Healthy females (25-30 cm body length and 200-320 g weight) and males (20-25 cm; 100-170 g) captured from the wild are preferably used as broodstock in the induced ovarian maturation process. Broodstock from greater depths (60-80 m), or more than 20 miles offshore, are preferable due to the lower prevalence of shrimp diseases, which are higher in coastal shrimp farming areas. Once the shrimp have recovered from transport stress for a few days, they are stocked in a circular maturation tank that is normally covered and kept in a dark room. The same stocking density (2-3/m²) is used for both females and males. Shrimp are subsequently induced to moult by manipulating the salinity of the water. After mating has occurred, which is easily determined by the presence of a spermatophore in the thelycum and hardening of the shell, the eyestalk of females is unilaterally ablated for endocrine stimulation. Broodstock are fed with squid, mussel or cockle meat, supplemented by polychaete or Artemia biomass to enhance reproductive performance.
The early stage of ovarian development can be first observed within a week after ablation. Later, gravid females with ripe stage eggs, which can be observed by the opaque diamond-shaped ovary under torchlight, are collected and transferred into spawning tanks. After spawning, these females can be re-used in the maturation process a few times, while males can be further used for several months, depending on prawn health and tank conditions.
Whether the spawners are caught directly from the sea or from an induced maturation tank, they generally spawn on the first or second night in the hatchery. However, spawning can be delayed for long distance or overnight transportation if each spawner is placed tightly in a PVC pipe to straighten its body. Gravid females should be placed individually in a small spawning tank to avoid the spread of disease that may occur in mixed spawning.
After spawning, eggs are generally kept in the same tank for fertilization until hatching. Nauplii are then collected and cleaned (rinsed with flowing seawater to remove the fat and debris released by the spawner) for transfer to larval rearing tanks or for transportation to other remote hatcheries. In Thailand, thousands of specialized small-scale or backyard hatcheries in inland areas buy nauplii and grow them to PL 12-15 because they cannot conduct the costly broodstock maturation operation. Broodstock maturation facilities located at the coast require a large volume of clean, clear seawater, while closed system larval rearing generally needs much less seawater. Therefore the inland hatcheries, whose land costs are much lower, are able to operate economically by purchasing seawater or brine that has been transported by trucks from the sea or from salt pans.
Hatchery
Smaller indoor concrete tanks (4-5 tonnes) than those originally used have now proved more efficient and manageable for larval rearing, particularly during the application of closed systems for disease prevention. If an outdoor system is unavoidable, due to economic constraints, tanks should be covered by black cloth or roof tiles in order to avoid the diurnal fluctuation of water temperature, and also to reduce light intensity. Nauplii are usually stocked at 100 000/tonne and cultured up to late mysis or early PL with approximately 70-80 percent survival rate. They are then transferred to a new tank and cultured up to PL 12-15; a further 70-80 percent survival rate is achieved in this stage. Diatoms (either Chaetoceros, or Skeletonema or Tetraselmis) that have been reared in monoculture, are fed at an approximate density of 30 000-50 000 cells/ml, starting from the protozoea stage and continuing until early PL (4-5). Diatoms can be replaced by micro-encapsulated diets or dry formulated feeds if their production is interrupted by rain. Artemia nauplii, at an average of 50 g of cysts per 100 000 larvae are given from mysis up to early PL stage. Artemia flakes are also used to supplement Artemia nauplii for cost reduction. From PL 4 to PL 15, artificial diets are commonly used to reduce the deterioration in water quality that occurs when fresh feed is applied. From hatching, it takes about 26 days to reach PL 15.
Due to its benthic habit, the harvesting of nursed juveniles in separate earthen ponds is difficult; thus nursing of hatchery produced postlarvae is impractical. Nursing in concrete tanks also yields poor survival, due to the cannibalistic behaviour of PL at high stocking densities. Since intensive ponds are well treated to eliminate all fish predators, it is safe to stock PL 15 prawns directly into the grow-out ponds. If the pond has not been well-prepared in time, or some predators have been observed, or the PL seem weak, PL 15 may be acclimatized by impoundment in nets, pens or small enclosure within grow-out ponds for less than a week before release.
In semi-intensive ponds, where postlarvae are not fully fed by artificial diets and some fish predators still remain, postlarvae are commonly nursed for a few weeks in an earthen compartment (5-10 percent of pond area) within the grow-out ponds. This enables the feed to be concentrated in this small nursing area, resulting in juveniles that are larger and thus better able to escape from the remaining fish predators after release into the grow-out pond.
There are three on-growing culture practices: extensive, semi-intensive and intensive, which represent low, medium, and high stocking densities respectively. Due to its benthic feeding habit Penaeus monodon is commercially cultured only in earthen ponds, under widely varying salinities from 2 to 30‰.
Extensive
Commonly found in Bangladesh, India, Indonesia, Myanmar, the Philippines and Viet Nam, extensive grow-out of shrimp is conducted in tidal areas where water pumping is unnecessary. Ponds with an irregular shape according to land boundaries are generally larger than five hectares and easily constructed by manual labour for cost reduction. Wild seeds, which either enter the pond through the gate by the tide or are purchased from collectors, are usually stocked at a density not exceeding 2/m². Shrimp feed on natural foods that enter the pond regularly on the tide and are subsequently enhanced by organic or chemical fertilizers. If available, fresh fish or molluscs may be used as supplementary feed. Due to the low stocking densities, larger sized shrimp (>50 g) are commonly harvested within six months or more. The yield is lowest in these extensive systems, at 50-500 kg/ha/yr. Due to the increase in land costs and the shortage of wild seeds, almost no new extensive farms are being constructed today. After gaining experience in shrimp farming, many farmers have upgraded their ponds to semi-intensive systems to provide better incomes.
Semi-intensive
Semi-intensive ponds (1-5 ha) are commonly stocked with hatchery-produced seeds at the rate of 5 to 20 PL/m². Water exchange is regularly carried out by tide and supplemented by pumping. The shrimp feed on natural foods enhanced by pond fertilization, supplemented by artificial diets. Production yields range from 500 to 4 000 kg/ha/yr.
Intensive
Intensive farms are commonly located in non-tidal areas where ponds can be completely drained and dried before each stocking. This culture system is found in all Penaeus monodon producing countries and is commonly practiced in Thailand, the Philippines, Malaysia and Australia. Ponds are generally small (0.1 to 1.0 ha) with a square or rectangular shape. Stocking density ranges from 20 to 60 PL/m². Heavy aeration, either powered by diesel engines or electric motors, is necessary for internal water circulation and oxygen supply for both animals and phytoplankton. Feeding with artificial diets is carried out 4-5 times per day followed by feed tray checking. Final FCR is normally between 1.2:1 and 2.0:1. Since the outbreak of white spot disease, reduced water exchange and closed systems have become commonplace, due to their lower risk of introducing viral diseases through intake water. However, feed and phytoplankton blooms need to be carefully monitored and managed to avoid deterioration of the pond bottom and water quality due to wastes. P. monodon has a habit of slowly nibbling feed on the pond bottom; this causes substantial nutrient losses because pellet stability is generally not longer than two hours. Efficient feed management is the major criteria for a successful crop, since feed represents over 50 percent of the production costs in intensive systems. Water quality parameters such as pH, salinity, dissolved oxygen, alkalinity, Secchi disc, H2S and unionised ammonia are regularly measured. If closed system culture is applied, stocking should be minimized, otherwise the pond must be harvested earlier (within 3.5 months instead of 4-5 months) and smaller shrimp will be yielded (20 g, instead of 30-35 g as achieved in semi-intensive and intensive systems with water exchange). Production yields of 4 000 to 15 000 kg/ha/yr are commonplace.
As shrimp feed technology has been readily available, each major producing country has developed its own commercial feed factories instead of relying on expensive imported feed. Prolonged storage of imported feed caused by sea transport or because of the necessity to import economically large volumes in each order, tends to reduce feed quality due to the rancidity.
Bamboo traps are traditionally used for the partial harvest of selected large shrimp in extensive culture. Semi-intensive ponds are commonly harvested by draining the pond by tide through a bag net installed at the outlet sluice gate. Intensive ponds are normally harvested similarly to semi-intensive ponds. If the tide does not allow harvesting, the drainage canal can be blocked to allow the water to be pumped out to reduce the water level. It is still necessary to pick the remaining shrimp by hand after the pond has been drained.
In Thailand, artificial sluice gates are temporarily installed inside the pond for the harvesting of many closed system ponds where a sluice gate for water exchange is not necessary. Shrimp are then trapped in this artificial gate during the pumping out of the water. For the live shrimp market, ponds are partially harvested by cast net in the early morning. Due to its burrowing habit, a drag net is not practical unless it is installed with electric shock gear to stimulate the shrimp to jump.
If shrimp are sold directly to processing plants, specialized teams for harvesting and handling are commonly used to ensure the first grade quality of their raw materials. After rough sorting, shrimp are washed, weighed and immediately killed in iced water at 0 °C. The most difficult job is to clean the shrimp picked by hand from pond bottoms at the end of the harvest, because they contain a lot of mud, organic matter and debris. Shrimp are then kept in ice in insulated containers and transported by small pick-up trucks for short distances or by large insulated trucks over long distances, either to processing plants or shrimp markets. For transportation of live shrimp from farms directly to aquarium tanks in restaurants, the shrimp are kept in aerated plastic containers at a density of 0.2-0.3 kg/litre of water. The containers are typically placed on small pick up trucks with a roof. For the export of live shrimp from Thailand to Hong Kong and China, water temperature is gradually reduced to 16-17 °C until the shrimp become inactive. The dormant shrimp are then packed alternately in layers of chilled sawdust or polystyrofoam beads in insulated boxes for export by air. This dry packing can minimize freight costs and shrimp can survive for 12-15 hours. Domestic markets mainly require chilled product supplied directly from farms or from shrimp markets.
In processing plants, shrimp are properly cleaned and sorted according to export standard sizes. Depending on market requirement, shrimp are processed in several categories before quick freezing at -10 °C and stored below -20 °C for further export by ship or air cargo. Due to an increasing demand and higher profit margin, many processing plants increasingly operate value-added product lines.
Production costs always vary depending on the site, season, scale of production, water management system (such as water exchange versus closed system), irregular production yield affected by culture problems, outbreak of diseases, etc. Operational costs for seed production regionally average at about USD 2.5/1 000 PL.
Production costs for adult shrimp are summarized as follows (USD/kg):
Production cycle

Production cycle of Penaeus monodon
Production systems
Seed supply
Due to their larger size and better survival, captured wild seeds were used commonly in south Asia for extensive ponds, which require a minimal amount of seed for stocking. However, the use of wild seeds has been reduced, due to overfishing and the outbreak of white spot disease in shrimp nursery grounds. Therefore most Penaeus monodon grow-out farms now rely solely on hatchery-produced seeds.
Broodstock
Healthy females (25-30 cm body length and 200-320 g weight) and males (20-25 cm; 100-170 g) captured from the wild are preferably used as broodstock in the induced ovarian maturation process. Broodstock from greater depths (60-80 m), or more than 20 miles offshore, are preferable due to the lower prevalence of shrimp diseases, which are higher in coastal shrimp farming areas. Once the shrimp have recovered from transport stress for a few days, they are stocked in a circular maturation tank that is normally covered and kept in a dark room. The same stocking density (2-3/m²) is used for both females and males. Shrimp are subsequently induced to moult by manipulating the salinity of the water. After mating has occurred, which is easily determined by the presence of a spermatophore in the thelycum and hardening of the shell, the eyestalk of females is unilaterally ablated for endocrine stimulation. Broodstock are fed with squid, mussel or cockle meat, supplemented by polychaete or Artemia biomass to enhance reproductive performance.
The early stage of ovarian development can be first observed within a week after ablation. Later, gravid females with ripe stage eggs, which can be observed by the opaque diamond-shaped ovary under torchlight, are collected and transferred into spawning tanks. After spawning, these females can be re-used in the maturation process a few times, while males can be further used for several months, depending on prawn health and tank conditions.
Whether the spawners are caught directly from the sea or from an induced maturation tank, they generally spawn on the first or second night in the hatchery. However, spawning can be delayed for long distance or overnight transportation if each spawner is placed tightly in a PVC pipe to straighten its body. Gravid females should be placed individually in a small spawning tank to avoid the spread of disease that may occur in mixed spawning.
After spawning, eggs are generally kept in the same tank for fertilization until hatching. Nauplii are then collected and cleaned (rinsed with flowing seawater to remove the fat and debris released by the spawner) for transfer to larval rearing tanks or for transportation to other remote hatcheries. In Thailand, thousands of specialized small-scale or backyard hatcheries in inland areas buy nauplii and grow them to PL 12-15 because they cannot conduct the costly broodstock maturation operation. Broodstock maturation facilities located at the coast require a large volume of clean, clear seawater, while closed system larval rearing generally needs much less seawater. Therefore the inland hatcheries, whose land costs are much lower, are able to operate economically by purchasing seawater or brine that has been transported by trucks from the sea or from salt pans.
Hatchery
Smaller indoor concrete tanks (4-5 tonnes) than those originally used have now proved more efficient and manageable for larval rearing, particularly during the application of closed systems for disease prevention. If an outdoor system is unavoidable, due to economic constraints, tanks should be covered by black cloth or roof tiles in order to avoid the diurnal fluctuation of water temperature, and also to reduce light intensity. Nauplii are usually stocked at 100 000/tonne and cultured up to late mysis or early PL with approximately 70-80 percent survival rate. They are then transferred to a new tank and cultured up to PL 12-15; a further 70-80 percent survival rate is achieved in this stage. Diatoms (either Chaetoceros, or Skeletonema or Tetraselmis) that have been reared in monoculture, are fed at an approximate density of 30 000-50 000 cells/ml, starting from the protozoea stage and continuing until early PL (4-5). Diatoms can be replaced by micro-encapsulated diets or dry formulated feeds if their production is interrupted by rain. Artemia nauplii, at an average of 50 g of cysts per 100 000 larvae are given from mysis up to early PL stage. Artemia flakes are also used to supplement Artemia nauplii for cost reduction. From PL 4 to PL 15, artificial diets are commonly used to reduce the deterioration in water quality that occurs when fresh feed is applied. From hatching, it takes about 26 days to reach PL 15.
Nursery
Due to its benthic habit, the harvesting of nursed juveniles in separate earthen ponds is difficult; thus nursing of hatchery produced postlarvae is impractical. Nursing in concrete tanks also yields poor survival, due to the cannibalistic behaviour of PL at high stocking densities. Since intensive ponds are well treated to eliminate all fish predators, it is safe to stock PL 15 prawns directly into the grow-out ponds. If the pond has not been well-prepared in time, or some predators have been observed, or the PL seem weak, PL 15 may be acclimatized by impoundment in nets, pens or small enclosure within grow-out ponds for less than a week before release.
In semi-intensive ponds, where postlarvae are not fully fed by artificial diets and some fish predators still remain, postlarvae are commonly nursed for a few weeks in an earthen compartment (5-10 percent of pond area) within the grow-out ponds. This enables the feed to be concentrated in this small nursing area, resulting in juveniles that are larger and thus better able to escape from the remaining fish predators after release into the grow-out pond.
Ongrowing techniques
There are three on-growing culture practices: extensive, semi-intensive and intensive, which represent low, medium, and high stocking densities respectively. Due to its benthic feeding habit Penaeus monodon is commercially cultured only in earthen ponds, under widely varying salinities from 2 to 30‰.
Extensive
Commonly found in Bangladesh, India, Indonesia, Myanmar, the Philippines and Viet Nam, extensive grow-out of shrimp is conducted in tidal areas where water pumping is unnecessary. Ponds with an irregular shape according to land boundaries are generally larger than five hectares and easily constructed by manual labour for cost reduction. Wild seeds, which either enter the pond through the gate by the tide or are purchased from collectors, are usually stocked at a density not exceeding 2/m². Shrimp feed on natural foods that enter the pond regularly on the tide and are subsequently enhanced by organic or chemical fertilizers. If available, fresh fish or molluscs may be used as supplementary feed. Due to the low stocking densities, larger sized shrimp (>50 g) are commonly harvested within six months or more. The yield is lowest in these extensive systems, at 50-500 kg/ha/yr. Due to the increase in land costs and the shortage of wild seeds, almost no new extensive farms are being constructed today. After gaining experience in shrimp farming, many farmers have upgraded their ponds to semi-intensive systems to provide better incomes.
Semi-intensive
Semi-intensive ponds (1-5 ha) are commonly stocked with hatchery-produced seeds at the rate of 5 to 20 PL/m². Water exchange is regularly carried out by tide and supplemented by pumping. The shrimp feed on natural foods enhanced by pond fertilization, supplemented by artificial diets. Production yields range from 500 to 4 000 kg/ha/yr.
Intensive
Intensive farms are commonly located in non-tidal areas where ponds can be completely drained and dried before each stocking. This culture system is found in all Penaeus monodon producing countries and is commonly practiced in Thailand, the Philippines, Malaysia and Australia. Ponds are generally small (0.1 to 1.0 ha) with a square or rectangular shape. Stocking density ranges from 20 to 60 PL/m². Heavy aeration, either powered by diesel engines or electric motors, is necessary for internal water circulation and oxygen supply for both animals and phytoplankton. Feeding with artificial diets is carried out 4-5 times per day followed by feed tray checking. Final FCR is normally between 1.2:1 and 2.0:1. Since the outbreak of white spot disease, reduced water exchange and closed systems have become commonplace, due to their lower risk of introducing viral diseases through intake water. However, feed and phytoplankton blooms need to be carefully monitored and managed to avoid deterioration of the pond bottom and water quality due to wastes. P. monodon has a habit of slowly nibbling feed on the pond bottom; this causes substantial nutrient losses because pellet stability is generally not longer than two hours. Efficient feed management is the major criteria for a successful crop, since feed represents over 50 percent of the production costs in intensive systems. Water quality parameters such as pH, salinity, dissolved oxygen, alkalinity, Secchi disc, H2S and unionised ammonia are regularly measured. If closed system culture is applied, stocking should be minimized, otherwise the pond must be harvested earlier (within 3.5 months instead of 4-5 months) and smaller shrimp will be yielded (20 g, instead of 30-35 g as achieved in semi-intensive and intensive systems with water exchange). Production yields of 4 000 to 15 000 kg/ha/yr are commonplace.
Feed supply
As shrimp feed technology has been readily available, each major producing country has developed its own commercial feed factories instead of relying on expensive imported feed. Prolonged storage of imported feed caused by sea transport or because of the necessity to import economically large volumes in each order, tends to reduce feed quality due to the rancidity.
Harvesting techniques
Bamboo traps are traditionally used for the partial harvest of selected large shrimp in extensive culture. Semi-intensive ponds are commonly harvested by draining the pond by tide through a bag net installed at the outlet sluice gate. Intensive ponds are normally harvested similarly to semi-intensive ponds. If the tide does not allow harvesting, the drainage canal can be blocked to allow the water to be pumped out to reduce the water level. It is still necessary to pick the remaining shrimp by hand after the pond has been drained.
In Thailand, artificial sluice gates are temporarily installed inside the pond for the harvesting of many closed system ponds where a sluice gate for water exchange is not necessary. Shrimp are then trapped in this artificial gate during the pumping out of the water. For the live shrimp market, ponds are partially harvested by cast net in the early morning. Due to its burrowing habit, a drag net is not practical unless it is installed with electric shock gear to stimulate the shrimp to jump.
Handling and processing
If shrimp are sold directly to processing plants, specialized teams for harvesting and handling are commonly used to ensure the first grade quality of their raw materials. After rough sorting, shrimp are washed, weighed and immediately killed in iced water at 0 °C. The most difficult job is to clean the shrimp picked by hand from pond bottoms at the end of the harvest, because they contain a lot of mud, organic matter and debris. Shrimp are then kept in ice in insulated containers and transported by small pick-up trucks for short distances or by large insulated trucks over long distances, either to processing plants or shrimp markets. For transportation of live shrimp from farms directly to aquarium tanks in restaurants, the shrimp are kept in aerated plastic containers at a density of 0.2-0.3 kg/litre of water. The containers are typically placed on small pick up trucks with a roof. For the export of live shrimp from Thailand to Hong Kong and China, water temperature is gradually reduced to 16-17 °C until the shrimp become inactive. The dormant shrimp are then packed alternately in layers of chilled sawdust or polystyrofoam beads in insulated boxes for export by air. This dry packing can minimize freight costs and shrimp can survive for 12-15 hours. Domestic markets mainly require chilled product supplied directly from farms or from shrimp markets.
In processing plants, shrimp are properly cleaned and sorted according to export standard sizes. Depending on market requirement, shrimp are processed in several categories before quick freezing at -10 °C and stored below -20 °C for further export by ship or air cargo. Due to an increasing demand and higher profit margin, many processing plants increasingly operate value-added product lines.
Production costs
Production costs always vary depending on the site, season, scale of production, water management system (such as water exchange versus closed system), irregular production yield affected by culture problems, outbreak of diseases, etc. Operational costs for seed production regionally average at about USD 2.5/1 000 PL.
Production costs for adult shrimp are summarized as follows (USD/kg):
| Extensive | Semi-intensive | Intensive | |
| Seed | 0.53 | 0.58 | 0.59 |
| Feed | – | 1.41 | 2.02 |
| Labour | 0.85 | 0.20 | 0.19 |
| Electricity & fuel | 0.21 | 0.36 | 0.33 |
| Chemical, materials & supplies | 0.16 | 0.18 | 0.26 |
| Overheads | – | 0.13 | 0.37 |
| Depreciation | 0.20 | 0.66 | 0.52 |
| Total | 1.95 | 3.52 | 4.28 |
Diseases and control measures
The major disease problems are included in the table below. There are no chemicals or drugs available to treat the viral infections listed but good management of pond, water, feed and health status of stock inputs can reduce their virulence. Outbreaks of the most serious virus (WSD) always occur after dramatic changes in water parameters such as temperature, salinity caused by heavy rain, DO2, hardness, and the stress to shrimp caused by deterioration in water quality and pond bottom environment. Pond preparation by proper bottom cleaning or regular scraping of the fouled layer is also a key factor for prevention of the shrimp stress caused by built up waste and toxic gases, and also for the elimination of virus carriers, particularly crustaceans. For confirmation, the Polymerase Chain Reaction (PCR) test for white spot disease or other viruses is widely used for screening of broodstock before spawning, nauplii before larval rearing, late PL before pond stocking, and shrimp in ponds for regular monitoring.
In some cases antibiotics and other pharmaceuticals have been used in treatment but their inclusion in this table does not imply an FAO recommendation.
Suppliers of pathology expertise
No specific institutes or laboratories named, but shrimp pathology expertise is now readily available.
In some cases antibiotics and other pharmaceuticals have been used in treatment but their inclusion in this table does not imply an FAO recommendation.
| DISEASE | AGENT | TYPE | SYNDROME | MEASURES |
| White spot (WSD)Otherwise known as WSBV, WSSV | Part of the white spot syndrome baculovirus complex | Virus | Acutely infected shrimp show rapid reduction in food consumption; lethargy; high mortality rates with cumulative mortalities reaching 100 percent within 3 to 10 days of the onset of clinical signs; acutely infected shrimp often have loose cuticle with white spots (which represent abnormal deposits of calcium salts by the cuticular epidermis) of 0.5 - 2.0 mm in diameter that are most apparent on the inside surface of the carapace; in many cases moribund shrimp display a pink to reddish-brown colouration due to expansion of cuticular chromatophores & few if any white spots | Screening of broodstock, nauplii, PL & grow-out stages; avoiding rapid changes in water conditions; avoiding shrimp stress; avoid use of fresh feeds, particularly crustacean; minimizing water exchange to prevent virus carriers entering the pond; treating infected ponds or hatcheries with 30 ppm chlorine to kill infected shrimp & carriers; disinfect associated equipment |
| Yellowhead (YHD) Also known as Yellow-head shrimp disease, Yellow-head virus (YHV), Yellow-head baculovirus (YBV), Yellow-head disease baculovirus (YHDBV) | Not yet described | Virus | Acute epizootics with high cumulative mortalities that may reach 100 percent within 3-5 days after appearance of clinical signs; infection is horizontally transmitted; PL 15 have been found to be resistant but PL 20-25 & on-growing juveniles through to sub-adults are highly susceptible; initially, feeding increases, followed by reduced feeding in later stages of the disease; pale body; yellowish swollen cephalothorax & hepatopancreas; whitish-yellowish-brownish gills; presumptive diagnosis can be made on basis of pond history, clinical signs, gross changes & histopathology | Screening of broodstock before hatchery operation & PL before stocking in pond; avoiding rapid changes in water pH, alkalinity, & dissolved O2; avoiding fresh aquatic feeds; proper cleaning of pond bottom before stocking; infected ponds & hatcheries must be disinfected similar to WSV (see above) |
| Baculoviral Midgut Gland Necrosis (BMN)Also known as midgut gland cloudy disease, white turbid liver disease, and white turbidity disease | Baculovirus | Virus | Generally infects larvae & early postlarval stages in which it can cause high mortalities; apparent white turbidity of the hepatopancreas caused by necrosis of tubule epithelium & possibly also the mucosal epithelium; larvae affected but later stages (late postlarvae) tend to show resistance; source of infection documented as wild-caught female spawners; larvae float inactively on the surface & exhibit a white midgut line through the abdomen | Wash fertile eggs through a soft gauze by running clean seawater to remove excrement or faeces of spawner; if infected, culture facility must be disinfected to avoid re-introduction of virus |
| Nuclear Polyhedrosis BaculovirosesAlso known as Monodon baculovirus disease (MBV) | Baculovirus | Virus | Lethargy, anorexia, dark coloured shrimp; reduced feeding & growth rates; often increased surface & gill fouling with various epibiotic & epicommensal organisms; severely affected larvae & postlarvae may exhibit a white midgut line through the abdomen; acute MBV causes loss of hepatopancreatic tubule & midgut epithelia &, consequently, dysfunction of these organs, often followed by secondary bacterial infections; linked with high mortalities (>90%) in late postlarvae & juvenile shrimp in many culture facilities; usually juvenile & adult P. monodon are more resistant to MBV than larval shrimp; MBV may predispose infected shrimp to infections by other pathogens; | Reduce stocking density, use of chemicals & environmentally induced stress; prevent contamination of fertilized eggs from spawner faeces by washing in formalin or iodophore treated seawater; if infected, culture facility must be disinfected & stock should be removed & sterilized |
Suppliers of pathology expertise
No specific institutes or laboratories named, but shrimp pathology expertise is now readily available.
STATISTICS
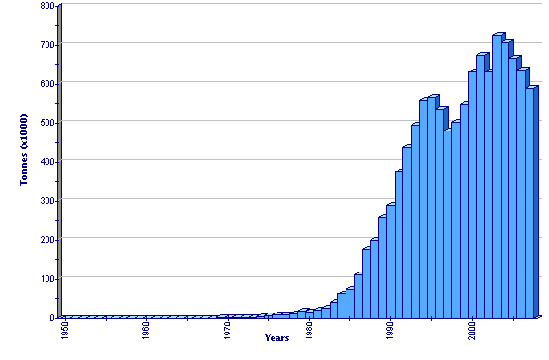
Production statistics
Global aquaculture production of Penaeus monodon
(FAO Fishery statistics)
(FAO Fishery statistics)
Market and trade
Products
Frozen head-on, head-off, and peeled shrimp used to be the major products for export to the main markets, which are USA, EU and Japan. Later, value-added products, such as microwavable or ready-to-cook tempura, sushi, shaomei, hargao, straightened, skewered, battered and breaded, spring roll and balls mainly processed in Thailand, have become increasingly popular. This has been because tight economic conditions in many developed countries limit frequent dinner in restaurants, and the time for cooking at home is scarce. Chilled product, which is sold in domestic markets, is generally non-exportable grade and shares less than 10 percent of all markets. Live product, which is mainly for domestic Chinese restaurants with some exports to Hong Kong and China, also shares less than 2 percent.
Prices and market statistics
In financial value, Penaeus monodon is the most important traded aquaculture commodity in Asia. C&F prices in Japan, whose market mainly requires large headless (16/20 size) shrimp from extensive and semi-intensive farms in Indonesia, India and Viet Nam, varied from USD 9-14/kg during 2001-2004. The US market purchased mainly small headless (21/25 size) shrimp (both peeled and shell-on) from intensive farms in Thailand and India at C&F prices ranging from USD 7-13/kg during the same period. The EU market, which mainly requires small head-on shrimp (31/40 size) from South East Asian intensive farms, paid C&F prices between USD 4.7 and 9.0/kg during 2001-2004.
Market regulations
Sanitary standards, standards for the uses of drugs and chemicals, and common food safety regulations for seafood (particularly shrimp) are already high in all major import countries. However, the EU market has more strict regulations (zero tolerance) on residues of chemicals and antibiotics, as well as the trade privilege or Generalized System of Preference (GSP) on import tax and HACCP. The US market enforces more strictly on a sanitary standard such as HACCP or Sensory Assessment. There are also additional regulations in the US regarding anti-dumping of imported shrimp, and the application of Turtle Excluder Devices (TEDs) on wild shrimp fishing fleets in exporting countries.
Frozen head-on, head-off, and peeled shrimp used to be the major products for export to the main markets, which are USA, EU and Japan. Later, value-added products, such as microwavable or ready-to-cook tempura, sushi, shaomei, hargao, straightened, skewered, battered and breaded, spring roll and balls mainly processed in Thailand, have become increasingly popular. This has been because tight economic conditions in many developed countries limit frequent dinner in restaurants, and the time for cooking at home is scarce. Chilled product, which is sold in domestic markets, is generally non-exportable grade and shares less than 10 percent of all markets. Live product, which is mainly for domestic Chinese restaurants with some exports to Hong Kong and China, also shares less than 2 percent.
Prices and market statistics
In financial value, Penaeus monodon is the most important traded aquaculture commodity in Asia. C&F prices in Japan, whose market mainly requires large headless (16/20 size) shrimp from extensive and semi-intensive farms in Indonesia, India and Viet Nam, varied from USD 9-14/kg during 2001-2004. The US market purchased mainly small headless (21/25 size) shrimp (both peeled and shell-on) from intensive farms in Thailand and India at C&F prices ranging from USD 7-13/kg during the same period. The EU market, which mainly requires small head-on shrimp (31/40 size) from South East Asian intensive farms, paid C&F prices between USD 4.7 and 9.0/kg during 2001-2004.
Market regulations
Sanitary standards, standards for the uses of drugs and chemicals, and common food safety regulations for seafood (particularly shrimp) are already high in all major import countries. However, the EU market has more strict regulations (zero tolerance) on residues of chemicals and antibiotics, as well as the trade privilege or Generalized System of Preference (GSP) on import tax and HACCP. The US market enforces more strictly on a sanitary standard such as HACCP or Sensory Assessment. There are also additional regulations in the US regarding anti-dumping of imported shrimp, and the application of Turtle Excluder Devices (TEDs) on wild shrimp fishing fleets in exporting countries.
STATUS AND TRENDS
Research
The following are urgent issues for study:
Expansion in aquaculture production of Penaeus monodon has not been as great as was originally expected, due to a number of causes, including major problems with viral disease outbreaks, shortages of broodstock, market competition and trade barriers. In addition, many farmers that originally reared Penaeus monodon have replaced this species with Litopenaeus vannamei, for which culture and domestication technologies are much simpler. L. vannamei disease problems are less severe, particularly for culture in inland freshwater ponds. Due to its lower price, this new species can be increasingly sold in domestic markets, which ensures stable incomes for farmers instead of only relying on the unstable export price. Shrimp farming will be more sustainable if farmers can shift production to other species when the existing cultured species faces problems. Decreasing P. monodon production can also improve the status of its broodstock in the wild in the future because less will be caught and less disease from grow-out ponds will be introduced into the sea. Due to this alternative species, the growth of P. monodon production is predicted to slow in the immediate future. Later, it may increase again if the research needs outlined above are addressed, thus improving the sustainability of production and reducing operational costs.
Market
In general, Penaeus monodon is the most prominent farmed crustacean product in international trade and has driven a significant expansion in aquaculture in many developing countries in Asia. Market prices during its early development were quite good due to little competition and strong demand from the Japanese market. International markets appear to have become almost saturated since global production reached 600 000 tonnes/yr. The price for P. monodon has since fallen, particularly during the booming of the production of Litopenaeus vannamei in Asia from 2001-2004. However, its price is still higher than L. vannamei. In the future, the market for P. monodon is expected to be less bright than it was in 1990s, due mainly to the saturation of export markets and reduction in world economic growth, as well as the emergence of non-tariff barriers in shrimp trade (such as anti-dumping rules), chemical residues, food safety, certification and eco-labelling in some importing countries. There has been increasing change in consumer preference from shrimp to marine fish, due to the lower cholesterol and higher omega-3 in fish.
Recommendations
In order to continue the growth of shrimp farming smoothly in the long term, domestic consumption should be promoted to avoid the problematic export markets. However, the domestic price should be reduced in order to encourage local consumption, through the use of advanced, efficient and sustainable culture systems. This is similar to the advanced chicken or salmon farming systems that have reduced production costs and guaranteed survival. Shrimp farmers in Asia should select whether to stock either P. monodon or L. vannamei according to the foreseen market and operational problems such as competition, climate and disease outbreak season.
The following are urgent issues for study:
- Domestication technology, which also leads to the efficient development of disease-free broodstock similar to those for Litopenaeus vannamei, is a major topic for on-going and future research in various institutions, including private sector organizations all over the world.
- Vaccination and effective treatment of shrimp viruses.
- Replacement of non-environmental friendly and costly fishmeal and Artemia in shrimp feeds.
- Efficient water treatment system for closed systems.
Expansion in aquaculture production of Penaeus monodon has not been as great as was originally expected, due to a number of causes, including major problems with viral disease outbreaks, shortages of broodstock, market competition and trade barriers. In addition, many farmers that originally reared Penaeus monodon have replaced this species with Litopenaeus vannamei, for which culture and domestication technologies are much simpler. L. vannamei disease problems are less severe, particularly for culture in inland freshwater ponds. Due to its lower price, this new species can be increasingly sold in domestic markets, which ensures stable incomes for farmers instead of only relying on the unstable export price. Shrimp farming will be more sustainable if farmers can shift production to other species when the existing cultured species faces problems. Decreasing P. monodon production can also improve the status of its broodstock in the wild in the future because less will be caught and less disease from grow-out ponds will be introduced into the sea. Due to this alternative species, the growth of P. monodon production is predicted to slow in the immediate future. Later, it may increase again if the research needs outlined above are addressed, thus improving the sustainability of production and reducing operational costs.
Market
In general, Penaeus monodon is the most prominent farmed crustacean product in international trade and has driven a significant expansion in aquaculture in many developing countries in Asia. Market prices during its early development were quite good due to little competition and strong demand from the Japanese market. International markets appear to have become almost saturated since global production reached 600 000 tonnes/yr. The price for P. monodon has since fallen, particularly during the booming of the production of Litopenaeus vannamei in Asia from 2001-2004. However, its price is still higher than L. vannamei. In the future, the market for P. monodon is expected to be less bright than it was in 1990s, due mainly to the saturation of export markets and reduction in world economic growth, as well as the emergence of non-tariff barriers in shrimp trade (such as anti-dumping rules), chemical residues, food safety, certification and eco-labelling in some importing countries. There has been increasing change in consumer preference from shrimp to marine fish, due to the lower cholesterol and higher omega-3 in fish.
Recommendations
In order to continue the growth of shrimp farming smoothly in the long term, domestic consumption should be promoted to avoid the problematic export markets. However, the domestic price should be reduced in order to encourage local consumption, through the use of advanced, efficient and sustainable culture systems. This is similar to the advanced chicken or salmon farming systems that have reduced production costs and guaranteed survival. Shrimp farmers in Asia should select whether to stock either P. monodon or L. vannamei according to the foreseen market and operational problems such as competition, climate and disease outbreak season.
MAIN ISSUES
In recent years, the development of shrimp farming has generated many public debates over environmental impact, such as the:
Overfishing of wild seeds and broodstock has been relaxed due to disease prevention measure and the shift to alternative species that can be domesticated. Shrimp farming in Asia does not create as many social conflicts with local communities as in Latin America, where large-scale farms are commonplace, because it is mostly operated by small-scale farmers that originate from coastal communities and own less than 5 ha of land. The shrimp industry also employs hundreds of thousands of rural people for farm operations and supply industries, as well as for shrimp processing and distribution. Enriched nutrients in shrimp farm effluent have proved to enhance the growth of aquatic animals and mangroves.
- Use of mangrove ecosystems for pond construction.
- Salinization of groundwater and agricultural land.
- Pollution of coastal waters due to pond effluents.
- Biodiversity issues arising from collection of wild seed and broodstock.
- Social conflicts with other users of resources.
- Farm discharges, causing self-pollution in shrimp growing areas as well as viral disease outbreaks.
Overfishing of wild seeds and broodstock has been relaxed due to disease prevention measure and the shift to alternative species that can be domesticated. Shrimp farming in Asia does not create as many social conflicts with local communities as in Latin America, where large-scale farms are commonplace, because it is mostly operated by small-scale farmers that originate from coastal communities and own less than 5 ha of land. The shrimp industry also employs hundreds of thousands of rural people for farm operations and supply industries, as well as for shrimp processing and distribution. Enriched nutrients in shrimp farm effluent have proved to enhance the growth of aquatic animals and mangroves.
Responsible aquaculture practices
Due to rapid expansion and increasing awareness of negative impacts of shrimp farming practices on the environment and its own production, many shrimp producing countries in Asia, particularly Thailand, have complied with the concept of responsible aquaculture in Article 9 of the FAO Code of Conduct. The Thai Government, with the agreement of stakeholders, used this concept, together with CODEX, ISO 14001/(EMS), to develop a shrimp farm certification programme under the Code of Conduct (COC) and Good Aquaculture Practice (GAP) for sustainable shrimp aquaculture. Conceptual guidelines, good practice and standards, together with training and auditing systems, have been established. The programme is running in all shrimp farming provinces and voluntary membership is increasing. This programme will be one of the models for further development of COC in other shrimp producing countries. However, certification and eco-labelling systems are a heavy task, because traceability of qualifying products through small-scale farms and hatcheries, which are the major producers in the region, seems to be difficult.
REFERENCES
Bibliography
| Chaitiamvong, S. & Supongpan, M. 1992. A Guide to Penaeoid Shrimps Found in Thai Waters. Australian Institute of Marine Science, Townsville, Australia. 77 pp. |
| Chanratchakool, P. 1993. Health Management in Shrimp Ponds. Aquatic Animal Health Research Institute, Bangkok, Thailand. 111 pp. |
| FAO. 2007. Improving Penaeus monodon hatchery practices. Manual based on experience in India. FAO Fisheries Technical Papers T446. Rome, FAO. |
| Kongkeo, H. 1997. Comparison of intensive shrimp farming systems in Indonesia, Philippines, Taiwan and Thailand. Aquaculture Research, 28:789-796. |
| Kongkeo, H.2001. Current status and development trends of aquaculture in Asia Region. pp. 267-293 In R.P. Subasinghe, P. Bueno, M.J. Phillips, S.E. McGladdery & J.R. Arthur (eds), Aquaculture in the Third Millennium. Technical Proceedings of the Conference on Aquaculture in the Third Millennium, Bangkok, Thailand, 20-25 February 2000. NACA, Bangkok, Thailand and FAO, Rome, Italy. |
| Kungvankij, P. 1986. Shrimp hatchery design, operation and management. NACA Training Manual Series No.1. NACA, Bangkok, Thailand. 88 pp. |
| Limsuwan, C. & Chanratchakool, P. 2004. Shrimp farming industry in Thailand. National Research Council of Thailand, Bangkok, Thailand. 206 pp. |
| NACA/FAO. 2001. Asia diagnostic guide to aquatic animal diseases. FAO Fisheries Technical Paper, 402/2. FAO, Rome, Italy. 237 pp. |
| Rosenberry, B. 1996. World Shrimp Farming 1996. Shrimp News International, San Diego, USA. 164 pp. |

