IDENTITY
Biological features
Rostrum moderately long with 7-10 dorsal and 2-4 ventral teeth. In mature males petasma symmetrical and semi-open. Spermatophores complex, consisting of sperm mass encapsulated by sheath. Mature female has open thelycum. Six nauplii, three protozoeal, and three mysis stages. Coloration normally translucent white, but can change depending on substratum, feed and water turbidity. Maximum size 23 cm, with maximum CL of 9 cm. Females commonly faster growing and larger than males.
Images gallery
 |
 |
Female broodstock (Photo: Briggs, M.) |
Maturation tanks in Latin America (Photo: Briggs, M.) |
 |
 |
Extensive ponds (Photo: Briggs, M.) |
Harvesting (Photo: Briggs, M.) |
PROFILE
Historical background
The first spawning of this species was achieved in Florida in 1973 from nauplii spawned and shipped from a wild-caught mated female from Panama. Following good pond results and the discovery of unilateral ablation (and adequate nutrition) to promote maturation in Panama in 1976, commercial culture of Penaeus vannamei began in South and Central America. Subsequent development of intensive breeding and rearing techniques led to its culture in Hawaii, mainland United States of America, and much of Central and South America by the early 1980s. From this time, the commercial culture of this species in Latin America showed a rapidly increasing trend (with peaks every 3–4 years during the warm, wet 'el niño' years), punctuated by declines co-incident with disease outbreaks during the cold 'la niña' years. Despite these problems, production of P. vannamei from the Americas has been increasing – after declining from its earlier peak production of 193 000 tonnes in 1998 to 143 000 tonnes in 2000 it had grown to over 270 000 tonnes by 2004. Asia has seen a phenomenal increase in the production of P. vannamei. Although no production was reported to FAO in 1999, it was nearly 1 116 000 tonnes by 2004 and had overtaken the production of P. monodon in China, Taiwan Province of China and Thailand, due to a number of favourable factors. However, due to fears over importation of exotic diseases, many Asian countries have been reluctant to promote farming of P. vannamei, so that its culture remains officially confined to experimental testing only in Cambodia, India, Malaysia, Myanmar and the Philippines. Thailand and Indonesia both freely permit its commercial culture but have official restrictions, so that only SPF/SPR broodstock may be imported. Similarly, most Latin American countries have strict quarantine laws or bans to prevent importation of exotic pathogens with new stocks.
Main producer countries
The main producer countries of Penaeus vannamei are shown in the map, while the whole list include: China, Thailand, Indonesia, Brazil, Ecuador, Mexico, Venezuela, Honduras, Guatemala, Nicaragua, Belize, Viet Nam, Malaysia, Tawian P.C., Pacific Islands, Peru, Colombia, Costa Rica, Panama, El Salvador, the United States of America, India, Philippines, Cambodia, Suriname, Saint Kitts, Jamaica, Cuba, Dominican Republic, Bahamas.
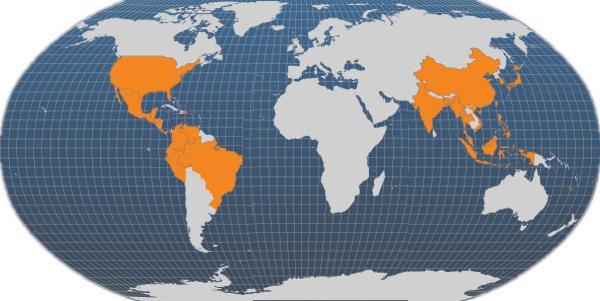

Main producer countries of Penaeus vannamei (FAO Fishery statistics, 2006)
Habitat and biology
The whiteleg shrimp is native to the Eastern Pacific coast from Sonora, Mexico in the North, through Central and South America as far South as Tumbes in Peru, in areas where water temperatures are normally >20 °C throughout the year. Penaeus vannamei live in tropical marine habitats. Adults live and spawn in the open ocean, while postlarvae migrate inshore to spend their juvenile, adolescent and sub-adult stages in coastal estuaries, lagoons or mangrove areas. Males become mature from 20 g and females from 28 g onwards at the age of 6–7 months. P. vannamei weighing 30–45 g will spawn 100 000–250 000 eggs of approximately 0.22 mm in diameter. Hatching occurs about 16 hours after spawning and fertilization. The first stage larvae, termed nauplii, swim intermittently and are positively phototactic. Nauplii do not feed, but live on their yolk reserves. The next larval stages (protozoea, mysis and early postlarvae respectively) remain planktonic for some time, eat phytoplankton and zooplankton, and are carried towards the shore by tidal currents. The postlarvae (PL) change their planktonic habit about 5 days after moulting into PL, move inshore and begin feeding on benthic detritus, worms, bivalves and crustaceans.
PRODUCTION

Captured wild seeds were used in Latin America for extensive pond culture of Penaeus vannamei until the late 1990s. Domestication and genetic selection programmes then provided more consistent supplies of high quality, disease free and/or resistant PL, which were cultured in hatcheries. Some were shipped to Hawaii in 1989, resulting in the production of SPF and SPR lines, leading to the industry in the United States of America and Asia.
There are three sources for broodstock P. vannamei:
Hatchery systems range from specialized, small, unsophisticated, often inland, backyard hatcheries to large, sophisticated and environmentally controlled installations, together with maturation units. Nauplii are stocked into flat, or preferably 'V' or 'U' shaped tanks with a volume of 4–100 m³, made from concrete, fibreglass or other plastic lined material. The larvae are either cultured to PL10–12 in a single larval rearing tank, or harvested at PL4–5 and transferred to flat-bottomed raceways/tanks and reared to PL10–30. Survival rates to PL10–12 should average >60 percent. Water is exchanged regularly (at 10–100 percent daily) to maintain good environmental conditions. Feeding normally consists of live food (microalgae and Artemia), supplemented by micro-encapsulated, liquid or dry formulated diets. From hatching, it takes about 21 days to reach harvest at PL12. Care is taken to reduce bacterial/pathogen contamination of the larval facilities using a combination of periodic dry-outs and disinfections, inlet water settlement, filtration and/or chlorination, disinfection of nauplii, water exchange and the use of antibiotics or (preferably) probiotics.
Most farming operations for P. vannamei do not use nurseries, but transport PL10–12 at reduced temperature either in plastic bags or oxygenated transportation tanks to the pond and introduce them directly. In some instances, nursery systems are used and comprise separate concrete nursery tanks or earth ponds, or even net pens or cages located within production ponds. Such nursery systems may be used for 1–5 weeks. Nurseries are useful in colder areas with limited growing seasons, where PL are nursed to a larger size (0.2–0.5 g) in heated tanks/ponds, before stocking into ponds. The use of super-intensive, temperature-controlled, greenhouse-enclosed, concrete or lined raceways have given good results in the United States of America.
Ongrowing techniques can be sub-divided into four main categories: extensive, semi-intensive, intensive and super-intensive, which represent low, medium, high and extremely high stocking densities respectively.
Extensive
Commonly found in Latin American countries, extensive grow-out of P. vannamei is conducted in tidal areas where minimal or no water pumping or aeration is provided. Ponds are of irregular shape, usually 5–10 ha (up to 30 ha) and 0.7–1.2 m deep. Originally, wild seeds entering the pond tidally through the gate, or purchased from collectors were used; since the 1980s hatchery reared PL are stocked at 4–10/m². Shrimp feed mainly on natural foods enhanced by fertilization, and once-daily feeding with low protein formulated diets. Despite low stocking densities, small shrimp of 11–12 g are harvested in 4–5 months. The yield in these extensive systems, is 150–500 kg/ha/crop, with 1–2 crops per year.
Semi-intensive
Semi-intensive ponds (1–5 ha) are stocked with hatchery-produced seeds at 10–30 PL/m²; such systems are common in Latin America. Regular water exchange is by pumping, pond depth is 1.0–1.2 m and aeration is at best minimal. The shrimp feed on natural foods enhanced by pond fertilization, supplemented by formulated diets 2–3 times daily. Production yields in semi-intensive ponds range from 500–2 000 kg/ha/crop, with 2 crops per year.
Intensive
Intensive farms are commonly located in non-tidal areas where ponds can be completely drained, dried and prepared before each stocking, and are increasingly being located far from the sea in cheaper, low salinity areas. This culture system is common in Asia and in some Latin American farms that are trying to increase productivity. Ponds are often earthen, but liners are also used to reduce erosion and enhance water quality. Ponds are generally small (0.1–1.0 ha) and square or round. Water depth is usually >1.5 m. Stocking densities range from 60–300 PL/m². Heavy aeration at 1 HP/400–600 kg of harvested shrimp is necessary for water circulation and oxygenation. Feeding with artificial diets is carried out 4–5 times per day. FCRs are 1.4–1.8:1.
Since the outbreak of viral syndromes, the use of domesticated disease free (SPF) and resistant (SPR) stocks, implementation of biosecurity measures and reduced water exchange systems have become commonplace. However, feed, water exchange/quality, aeration and phytoplankton blooms require carefully monitoring and management. Production yields of 7–20 000 kg/ha/crop, with 2–3 crops per year can be achieved, up to a maximum of 30–35 000 kg/ha/crop.
In the 'bacterial floc' system, the ponds (0.07–1.6 ha) are managed as highly aerated, recirculating, heterotrophic bacterial systems. Low protein feeds are fed 2–5 times per day, in an effort to increase the C:N ratio to >10:1 and divert added nutrients though bacterial rather than algal pathways. Stocking at 80–160 PL/m², the ponds become heterotrophic and flocs of bacteria are formed, which are consumed by the shrimp, reducing dependence on high protein feeds and FCR and increasing cost efficiency. Such systems have realized productions of 8–50 000 kg/ha/crop in Belize and Indonesia.
Super-intensive
Recent research conducted in the United States of America has focused on growing P. vannamei in super-intensive raceway systems enclosed in greenhouses, using no water exchange (only the replacement of evaporation losses) or discharge, stocked with SPF PL. They are thus biosecure, eco-friendly, have a small ecological footprint and can produce cost-efficient, high quality shrimp. Stocking 282 m² raceways with 300–450 0.5–2 g juveniles/m² and ongrowing for 3–5 months has realized production of 28 000–68 000 kg/ha/crop at growth rates of 1.5 g/week, survivals of 55–91 percent, mean weight of 16–26 g and FCRs of 1.5–2.6:1.
P. vannamei are very efficient at utilizing the natural productivity of shrimp ponds, even under intensive culture conditions. Additionally, feed costs are generally less for P. vannamei than the more carnivorous P. monodon, due to their lower requirement for protein (18–35 percent compared to 36–42 percent), especially where bacterial floc systems are used. Feed prices for P. vannamei range from USD 0.6/kg in Latin America and Thailand to USD 0.7–1.1/kg elsewhere around Asia; FCRs of 1.2–1.8:1 are generally obtained.
Extensive and semi-intensive ponds are harvested by draining the pond at low tide through a bag net installed in the outlet sluice gate. If the tide does not allow harvesting, the water can be pumped out. In some larger farms, harvesting machines pump shrimp and water up to the pond bank where they are dewatered. Intensive ponds may be harvested similarly and small 2–6 man seine nets are dragged around the pond to corral shrimp to the side of the pond from where they are removed by cast or dip net or perforated buckets.
Partial harvesting is common in Asian intensive culture after the first 3 months. In Thailand, artificial sluice gates are temporarily installed inside one corner of the pond to harvest closed system ponds. Shrimp are then trapped in nets attached to this temporary gate when the pond is pumped out.
In super-intensive systems, the shrimp are simply harvested with large scoop nets when required for processing.
If shrimp are sold directly to processing plants, specialized teams for harvesting and handling are commonly used to maintain shrimp quality. After sorting, shrimp are washed, weighed and immediately killed in iced water at 0–4 °C. Often sodium metabisulphate is added to the chilled water to prevent melanosis and red-head. Shrimp are then kept in ice in insulated containers and transported by truck either to processing plants or domestic shrimp markets. In processing plants, shrimp are placed in iced bins and cleaned and sorted according to standard export sizes. Shrimp are processed, quickly frozen at -10 °C and stored at -20 °C for export by ship or air cargo. Due to an increasing demand, no taxes and higher profit margins, many processing plants operate value-added product lines.
Production costs vary depending on many factors. Operational costs for seed production averages USD 0.5–1.0/1 000 PL, whilst sales prices vary from USD 0.4/1 000 PL8–10 in China and USD 1.0–1.2/1 000 PL12 in Ecuador to USD 1.5 3.0/1 000 PL12 around Asia. Lower feed costs and higher intensity levels result in mean production costs for ongrowing of approximately USD 2.5–3.0/kg for P. vannamei, compared to USD 3.0–4.0/kg for more extensive P. monodon culture.
Production cycle

Production cycle of Penaeus vannamei
Production systems
Seed supply
Captured wild seeds were used in Latin America for extensive pond culture of Penaeus vannamei until the late 1990s. Domestication and genetic selection programmes then provided more consistent supplies of high quality, disease free and/or resistant PL, which were cultured in hatcheries. Some were shipped to Hawaii in 1989, resulting in the production of SPF and SPR lines, leading to the industry in the United States of America and Asia.
Broodstock maturation, spawning and hatching
There are three sources for broodstock P. vannamei:
- Where they occur naturally, broodstock are sea-caught (usually at 1 year of age and weighing >40 g) and spawned.
- Cultured shrimp harvested from ponds (after 4–5 months at 15–25 g), are on-grown for 2–3 months and then transferred to maturation facilities at >7 months of age when they weigh 30–35 g.
- Purchased from tank-reared SPF/SPR broodstock from the United States of America, (at 7–8 months of age and weighing 30–40 g).
Hatchery production
Hatchery systems range from specialized, small, unsophisticated, often inland, backyard hatcheries to large, sophisticated and environmentally controlled installations, together with maturation units. Nauplii are stocked into flat, or preferably 'V' or 'U' shaped tanks with a volume of 4–100 m³, made from concrete, fibreglass or other plastic lined material. The larvae are either cultured to PL10–12 in a single larval rearing tank, or harvested at PL4–5 and transferred to flat-bottomed raceways/tanks and reared to PL10–30. Survival rates to PL10–12 should average >60 percent. Water is exchanged regularly (at 10–100 percent daily) to maintain good environmental conditions. Feeding normally consists of live food (microalgae and Artemia), supplemented by micro-encapsulated, liquid or dry formulated diets. From hatching, it takes about 21 days to reach harvest at PL12. Care is taken to reduce bacterial/pathogen contamination of the larval facilities using a combination of periodic dry-outs and disinfections, inlet water settlement, filtration and/or chlorination, disinfection of nauplii, water exchange and the use of antibiotics or (preferably) probiotics.
Nursery
Most farming operations for P. vannamei do not use nurseries, but transport PL10–12 at reduced temperature either in plastic bags or oxygenated transportation tanks to the pond and introduce them directly. In some instances, nursery systems are used and comprise separate concrete nursery tanks or earth ponds, or even net pens or cages located within production ponds. Such nursery systems may be used for 1–5 weeks. Nurseries are useful in colder areas with limited growing seasons, where PL are nursed to a larger size (0.2–0.5 g) in heated tanks/ponds, before stocking into ponds. The use of super-intensive, temperature-controlled, greenhouse-enclosed, concrete or lined raceways have given good results in the United States of America.
Ongrowing techniques
Ongrowing techniques can be sub-divided into four main categories: extensive, semi-intensive, intensive and super-intensive, which represent low, medium, high and extremely high stocking densities respectively.
Extensive
Commonly found in Latin American countries, extensive grow-out of P. vannamei is conducted in tidal areas where minimal or no water pumping or aeration is provided. Ponds are of irregular shape, usually 5–10 ha (up to 30 ha) and 0.7–1.2 m deep. Originally, wild seeds entering the pond tidally through the gate, or purchased from collectors were used; since the 1980s hatchery reared PL are stocked at 4–10/m². Shrimp feed mainly on natural foods enhanced by fertilization, and once-daily feeding with low protein formulated diets. Despite low stocking densities, small shrimp of 11–12 g are harvested in 4–5 months. The yield in these extensive systems, is 150–500 kg/ha/crop, with 1–2 crops per year.
Semi-intensive
Semi-intensive ponds (1–5 ha) are stocked with hatchery-produced seeds at 10–30 PL/m²; such systems are common in Latin America. Regular water exchange is by pumping, pond depth is 1.0–1.2 m and aeration is at best minimal. The shrimp feed on natural foods enhanced by pond fertilization, supplemented by formulated diets 2–3 times daily. Production yields in semi-intensive ponds range from 500–2 000 kg/ha/crop, with 2 crops per year.
Intensive
Intensive farms are commonly located in non-tidal areas where ponds can be completely drained, dried and prepared before each stocking, and are increasingly being located far from the sea in cheaper, low salinity areas. This culture system is common in Asia and in some Latin American farms that are trying to increase productivity. Ponds are often earthen, but liners are also used to reduce erosion and enhance water quality. Ponds are generally small (0.1–1.0 ha) and square or round. Water depth is usually >1.5 m. Stocking densities range from 60–300 PL/m². Heavy aeration at 1 HP/400–600 kg of harvested shrimp is necessary for water circulation and oxygenation. Feeding with artificial diets is carried out 4–5 times per day. FCRs are 1.4–1.8:1.
Since the outbreak of viral syndromes, the use of domesticated disease free (SPF) and resistant (SPR) stocks, implementation of biosecurity measures and reduced water exchange systems have become commonplace. However, feed, water exchange/quality, aeration and phytoplankton blooms require carefully monitoring and management. Production yields of 7–20 000 kg/ha/crop, with 2–3 crops per year can be achieved, up to a maximum of 30–35 000 kg/ha/crop.
In the 'bacterial floc' system, the ponds (0.07–1.6 ha) are managed as highly aerated, recirculating, heterotrophic bacterial systems. Low protein feeds are fed 2–5 times per day, in an effort to increase the C:N ratio to >10:1 and divert added nutrients though bacterial rather than algal pathways. Stocking at 80–160 PL/m², the ponds become heterotrophic and flocs of bacteria are formed, which are consumed by the shrimp, reducing dependence on high protein feeds and FCR and increasing cost efficiency. Such systems have realized productions of 8–50 000 kg/ha/crop in Belize and Indonesia.
Super-intensive
Recent research conducted in the United States of America has focused on growing P. vannamei in super-intensive raceway systems enclosed in greenhouses, using no water exchange (only the replacement of evaporation losses) or discharge, stocked with SPF PL. They are thus biosecure, eco-friendly, have a small ecological footprint and can produce cost-efficient, high quality shrimp. Stocking 282 m² raceways with 300–450 0.5–2 g juveniles/m² and ongrowing for 3–5 months has realized production of 28 000–68 000 kg/ha/crop at growth rates of 1.5 g/week, survivals of 55–91 percent, mean weight of 16–26 g and FCRs of 1.5–2.6:1.
Feed supply
P. vannamei are very efficient at utilizing the natural productivity of shrimp ponds, even under intensive culture conditions. Additionally, feed costs are generally less for P. vannamei than the more carnivorous P. monodon, due to their lower requirement for protein (18–35 percent compared to 36–42 percent), especially where bacterial floc systems are used. Feed prices for P. vannamei range from USD 0.6/kg in Latin America and Thailand to USD 0.7–1.1/kg elsewhere around Asia; FCRs of 1.2–1.8:1 are generally obtained.
Harvesting techniques
Extensive and semi-intensive ponds are harvested by draining the pond at low tide through a bag net installed in the outlet sluice gate. If the tide does not allow harvesting, the water can be pumped out. In some larger farms, harvesting machines pump shrimp and water up to the pond bank where they are dewatered. Intensive ponds may be harvested similarly and small 2–6 man seine nets are dragged around the pond to corral shrimp to the side of the pond from where they are removed by cast or dip net or perforated buckets.
Partial harvesting is common in Asian intensive culture after the first 3 months. In Thailand, artificial sluice gates are temporarily installed inside one corner of the pond to harvest closed system ponds. Shrimp are then trapped in nets attached to this temporary gate when the pond is pumped out.
In super-intensive systems, the shrimp are simply harvested with large scoop nets when required for processing.
Handling and processing
If shrimp are sold directly to processing plants, specialized teams for harvesting and handling are commonly used to maintain shrimp quality. After sorting, shrimp are washed, weighed and immediately killed in iced water at 0–4 °C. Often sodium metabisulphate is added to the chilled water to prevent melanosis and red-head. Shrimp are then kept in ice in insulated containers and transported by truck either to processing plants or domestic shrimp markets. In processing plants, shrimp are placed in iced bins and cleaned and sorted according to standard export sizes. Shrimp are processed, quickly frozen at -10 °C and stored at -20 °C for export by ship or air cargo. Due to an increasing demand, no taxes and higher profit margins, many processing plants operate value-added product lines.
Production costs
Production costs vary depending on many factors. Operational costs for seed production averages USD 0.5–1.0/1 000 PL, whilst sales prices vary from USD 0.4/1 000 PL8–10 in China and USD 1.0–1.2/1 000 PL12 in Ecuador to USD 1.5 3.0/1 000 PL12 around Asia. Lower feed costs and higher intensity levels result in mean production costs for ongrowing of approximately USD 2.5–3.0/kg for P. vannamei, compared to USD 3.0–4.0/kg for more extensive P. monodon culture.
Diseases and control measures
The major disease problems suffered by P. vannamei are shown in the table below. The availability of SPF and SPR broodstock provide a means of avoiding these diseases, although biosecurity procedures are also important, including:
In some cases antibiotics and other pharmaceuticals have been used in treatment but their inclusion in this table does not imply an FAO recommendation.
Suppliers of pathology expertise
Assistance can be provided from the following sources:
- Thorough drying/scraping of pond bottoms between cycles.
- Reducing water exchange and fine screening of any inlet water.
- Use of bird netting or scarers.
- Putting barriers around ponds.
- Sanitary procedures.
In some cases antibiotics and other pharmaceuticals have been used in treatment but their inclusion in this table does not imply an FAO recommendation.
| DISEASE | AGENT | TYPE | SYNDROME | MEASURES |
| White spot (WSD); also known as WSBV or WSSV | Part of the white spot syndrome baculovirus complex (recently renamed in a new family as a nimavirus) | Virus | Acutely infected shrimp show reduced food consumption; lethargy; high mortality of 100% within 3–10 days of onset of clinical signs; loose cuticles with white spots of 0.5–2.0 mm diameter, most apparent inside the carapace; moribund shrimp often have pink to reddish-brown colouration due to expansion of cuticular chromatophores & few if any white spots | Use SPF broodstock; wash & disinfect eggs/nauplii with iodine, formalin; screen broodstock, nauplii, PL & pond stages; avoid rapid changes in water quality; maintain water temperature >30 °C; avoid stress; avoid use of fresh feeds such as trash fish; minimize water exchange to prevent entry of virus carriers; treat infected ponds & hatcheries with 30 ppm chlorine to kill infected shrimp & carriers; disinfect associated equipment |
| Taura Syndrome (TS); also known as Taura syndrome Virus (TSV) or red tail disease | Single-stranded RNA virus (Picornaviridae) | Virus | Occurs during single moult in juvenile shrimp beginning 5–20 days after stocking, or has a chronic course over several months; weakness, soft shell, empty gut & diffuse expansion of red chromatophores in appendages; mortality varies 5–95%; survivors may have black lesions, & remain carriers for life | Use SPF & SPR broodstock; wash & disinfect eggs & nauplii; clean & disinfect contaminated vehicles & equipment; scare away birds (vectors); destroy all stock & thoroughly disinfect infected facilities |
| Infectious Hypodermal & Haematopoietic necrosis (IHHNV), causing Runt Deformity Syndrome (RDS) | Systemic parvovirus | Virus | Low mortality for resistant P. vannamei; however, reduced feeding, growth & feed efficiency; cuticular deformities (bent rostrum – RDS) occurs in <30% of infected populations, increasing variance of final harvest weight & reducing market value | Use SPF broodstock; wash & disinfect eggs & nauplii; if infected, culture facility must be completely & very carefully disinfected to avoid re-introduction |
| Baculoviral Midgut Gland Necrosis (BMN); also known as midgut gland cloudy disease, white turbid liver disease, & white turbidity disease | Non-occluded enteric baculovirus | Virus | Infects larval & early PL stages, causing high mortality; white turbidity of hepatopancreas caused by necrosis of tubule epithelium; larvae float inactively on surface; later stages show resistance; positive broodstock are source of infection | Separate eggs from faeces, wash eggs & nauplii with running clean seawater & disinfect with iodine &/or formalin; disinfect infected culture facility to avoid re-introduction |
| Vibriosis | Vibrio spp., particularly V. harveyi & V. parahaemolyticus | Bacteria | May cause various important syndromes, such as luminescence & the so-called zoea-2 & bolitas syndromes In hatchery; seen as luminescence in water &/or shrimp body; disruption of gut; fouling of body; reduced feeding & high mortality In ponds, high levels of vibrios are associated with red discoloration of shrimp (especially tails) & internal & external necrosis; low feeding & chronic mortality; often a secondary infection resulting from poor environmental management; weakens shrimp which become susceptible to viral infections |
Careful system management In hatcheries, disinfect facilities, equipment, water & workers; use bacteria-free live feeds; cover culture tanks with plastic sheet to prevent transfer In ponds, prevent by proper preparation; bloom control; good water & feed management; control stocking density & aeration to maintain optimal environmental conditions throughout culture cycle |
Suppliers of pathology expertise
Assistance can be provided from the following sources:
-
Prof. Lightner, D.
Aquaculture Pathology Section - Department of Veterinary Science - University of Arizona
Building 90, Room 202 - Tucson, AZ 85721, United States of America
Telephone: (+1) 520 6218414 - Fax: (+1) 520 6214899
E-mail: [email protected] -
Prof. Chen, S.N.
Department of Zoology Director, Institute of Fishery Biology - National Taiwan University
No. 1 Roosevelt Road, Section 4. - Taipei, Taiwan 10764, Taiwan, Province of China
Telephone: (+886) 2 3687101 - Fax: (+886) 2 3687122
E-mail: [email protected] -
Prof. Flegel, T.
Centex Shrimp, Chalern Prakiat Building - Faculty of Science - Mahidol University
Rama 6 Road - Bangkok, 10400, Thailand
Telephone: Personal (+66) 2 2015876 Mobile Phone (+66) 1 4035833 - Office (+66) 2 20158-70 or -71 or -72 - Fax: (+66) 2 2015873
E-mail: [email protected] -
Dr. Walker, P.
Associate Professor and Principal Research Scientist - CSIRO Livestock Industries
PMB 3 Indooroopilly - Queensland - 4068, Australia
Telephone: (+61) 7 32143758 - Fax: (+61) 7 32142718
E-mail: [email protected]
STATISTICS
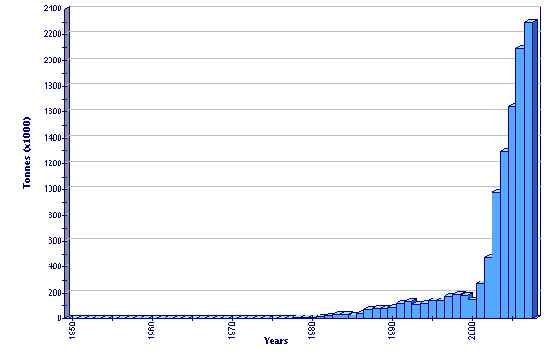
Production statistics
Global aquaculture production of Penaeus vannamei
(FAO Fishery statistics)
(FAO Fishery statistics)

Market and trade
Products
Frozen head-on, head-off, and peeled shrimp were formerly the major products for export to the main global markets of United States of America, European Union and Japan. The trend now is for the processing of value-added products. This is due to the lack of anti-dumping tariffs for processed products to the United States of America market, fewer people eating out and the desire for ready-to-cook or ready-to-eat products for home dining.
Prices and market statistics
The major market for shrimp is the United States of America, which was expected to import approximately 477 000 tonnes worth USD 3.1 billion in 2005, 1.8 times more than the 264 000 tonnes imported in 2000. The United States of America was traditionally supplied with small frozen or processed headless shrimp from Latin America. More recently, the United States of America has looked to Asia to supply its increasing demand (1.9 kg/capita in 2004). Major suppliers to the United States of America in 2005 were Thailand, Ecuador, India, China and Viet Nam. However, the rapidly increasing production of P. vannamei has led to serious price depression in the international markets. Similarly, farm gate value for 15–20 g size whiteleg shrimp has steadily decreased from USD 5/kg in 2000 to about USD 3.0–3.5/kg in 2005.
The next most important market is the European Union (importing 183 000 tonnes in the first half of 2005), which favours small (31/40 count), whole, frozen shrimp. Japan, whose market mainly requires large headless (16/20 count) shrimp, is typically supplied by P. monodon from large extensive Asian farms.
Market regulations
Standards for sanitation and the use of drugs and chemicals, and common food safety regulations for seafood (particularly shrimp) are already high in all major importing countries. However, the European Union market has more strict regulations (zero tolerance) on residues of chemicals and antibiotics, as well as the Generalized System of Preference (GSP) on import tax. The United States of America market enforces more strictly on a sanitary standard such as HACCP or Sensory Assessment, but has also instigated strict controls over banned antibiotics in shrimp. From June 2005, the final antidumping tariffs on cultured shrimp imported into the United States of America from 6 main shrimp producing countries were finalized and set (for the general rate) at approximately 113 percent for China, 26 percent for Viet Nam, 10 percent for India, 7 percent for Brazil, 6 percent for Thailand, and 4 percent for Ecuador. Mexico and Indonesia escaped these tariffs.
Frozen head-on, head-off, and peeled shrimp were formerly the major products for export to the main global markets of United States of America, European Union and Japan. The trend now is for the processing of value-added products. This is due to the lack of anti-dumping tariffs for processed products to the United States of America market, fewer people eating out and the desire for ready-to-cook or ready-to-eat products for home dining.
Prices and market statistics
The major market for shrimp is the United States of America, which was expected to import approximately 477 000 tonnes worth USD 3.1 billion in 2005, 1.8 times more than the 264 000 tonnes imported in 2000. The United States of America was traditionally supplied with small frozen or processed headless shrimp from Latin America. More recently, the United States of America has looked to Asia to supply its increasing demand (1.9 kg/capita in 2004). Major suppliers to the United States of America in 2005 were Thailand, Ecuador, India, China and Viet Nam. However, the rapidly increasing production of P. vannamei has led to serious price depression in the international markets. Similarly, farm gate value for 15–20 g size whiteleg shrimp has steadily decreased from USD 5/kg in 2000 to about USD 3.0–3.5/kg in 2005.
The next most important market is the European Union (importing 183 000 tonnes in the first half of 2005), which favours small (31/40 count), whole, frozen shrimp. Japan, whose market mainly requires large headless (16/20 count) shrimp, is typically supplied by P. monodon from large extensive Asian farms.
Market regulations
Standards for sanitation and the use of drugs and chemicals, and common food safety regulations for seafood (particularly shrimp) are already high in all major importing countries. However, the European Union market has more strict regulations (zero tolerance) on residues of chemicals and antibiotics, as well as the Generalized System of Preference (GSP) on import tax. The United States of America market enforces more strictly on a sanitary standard such as HACCP or Sensory Assessment, but has also instigated strict controls over banned antibiotics in shrimp. From June 2005, the final antidumping tariffs on cultured shrimp imported into the United States of America from 6 main shrimp producing countries were finalized and set (for the general rate) at approximately 113 percent for China, 26 percent for Viet Nam, 10 percent for India, 7 percent for Brazil, 6 percent for Thailand, and 4 percent for Ecuador. Mexico and Indonesia escaped these tariffs.
STATUS AND TRENDS
Research
The following are some of the highest priority areas for research into P. vannamei culture:
While the expansion of P. vannamei culture has been rapid in recent years, particularly in Asia, it has led to reduced value of harvested shrimp. This trend is expected to continue. Under such circumstances, the less efficient producers may not be able to compete with those capable of producing either more eco-friendly or cheaper products. Recent worldwide trends have been towards the integration of the industry, in response to the ever increasing requirement for traceability and control within the culture system.
Market
There has been a slowly increasing demand for shrimp in world markets, as capture fisheries stagnate and people became more affluent and conscious of healthy food choices. Despite the increased demand, the price for P. vannamei has been declining steadily. In the future, the market for P. vannamei is expected to become more competitive, due mainly to the saturation of export markets and reduction in world economic growth, as well as the emergence of non-tariff barriers in shrimp trade. Additionally, the industry will need to accommodate importing countries requirements on:
All shrimp farmers are becoming acutely aware of the growing need to farm shrimp in a responsible, traceable and low impact manner which can enhance biosecurity, and help protect the environment, whilst producing shrimp in a cost efficient manner. The newly developed intensive bacterial floc and super-intensive systems may have potential to address all of these concerns and should be investigated more thoroughly. In order to continue the growth of shrimp farming smoothly in the long term, domestic consumption should be promoted (as in China) to supplement the problematic export markets.
The following are some of the highest priority areas for research into P. vannamei culture:
- Continued development of SPR lines of P. vannamei for viruses including TSV, WSSV, IHHNV, BMNV and IMNV.
- Development of faster growing lines of SPF/SPR stocks.
- Continued development of biosecure, high density and low salinity culture systems.
- Vaccination and other effective treatments for shrimp viruses.
- Replacement of non-eco friendly and costly marine meals in shrimp feeds.
- Efficient water treatment and management systems for closed culture systems.
- Techniques for reducing bacterial loads in shrimp culture systems.
- Effective disinfection procedures for eggs, nauplii and PL in hatcheries.
- Effective replacements (i.e. probiotics and immunostimulants) for antibiotics.
While the expansion of P. vannamei culture has been rapid in recent years, particularly in Asia, it has led to reduced value of harvested shrimp. This trend is expected to continue. Under such circumstances, the less efficient producers may not be able to compete with those capable of producing either more eco-friendly or cheaper products. Recent worldwide trends have been towards the integration of the industry, in response to the ever increasing requirement for traceability and control within the culture system.
Market
There has been a slowly increasing demand for shrimp in world markets, as capture fisheries stagnate and people became more affluent and conscious of healthy food choices. Despite the increased demand, the price for P. vannamei has been declining steadily. In the future, the market for P. vannamei is expected to become more competitive, due mainly to the saturation of export markets and reduction in world economic growth, as well as the emergence of non-tariff barriers in shrimp trade. Additionally, the industry will need to accommodate importing countries requirements on:
- Chemical residues.
- Food safety.
- Certification.
- Traceability.
- Eco-labelling.
- Environmental sustainability.
All shrimp farmers are becoming acutely aware of the growing need to farm shrimp in a responsible, traceable and low impact manner which can enhance biosecurity, and help protect the environment, whilst producing shrimp in a cost efficient manner. The newly developed intensive bacterial floc and super-intensive systems may have potential to address all of these concerns and should be investigated more thoroughly. In order to continue the growth of shrimp farming smoothly in the long term, domestic consumption should be promoted (as in China) to supplement the problematic export markets.
MAIN ISSUES
The recent expansion of shrimp culture has generated many public debates over its effects on the environment and its sustainability, such as the perceived:
- Use of protective mangrove ecosystems for pond construction.
- Slash and burn style use of ponds for a few years, before moving to new areas.
- Salinization of groundwater and agricultural land.
- Pollution of coastal waters by pond effluents.
- Overuse of marine meals leading to inefficient use of vital protein sources and disruption of marine ecosystems.
- Biodiversity issues arising from collection of wild seed and broodstock and introduction of non-native species and their attendant pathogens.
- Social conflicts with other resource users.
- Farm discharges, causing self-pollution in shrimp growing areas.
Responsible aquaculture practices
Due to rapid expansion and increasing awareness of the negative impacts of shrimp farming practices on the environment and its own production, many shrimp producing countries are making sincere efforts to comply with the concept of responsible aquaculture as detailed in Article 9 of the FAO Code of Conduct for Responsible Fisheries (CCRF). The formulation and adoption of BMPs (or Good Aquaculture Practices – GAP) is gaining prevalence to enhance biosecurity, increase cost efficiency, reduce chemical residues and increase traceability. Organic certification for shrimp farming is being seriously considered. HACCP and ISO standards, already used in processing/feed plants, are being adopted in farms and hatcheries. FAO and other organizations have developed a system of guidelines and BMPs to help shrimp producing countries comply with the various aspects of the CCRF (FAO et.al, 2006).
REFERENCES
Bibliography
| Boyd, C.E. & Clay, J.W. 2002. Evaluation of Belize Aquaculture Ltd: A superintensive shrimp aquaculture system. Report prepared under the World Bank, NACA, WWF and FAO Consortium Program on Shrimp Farming and the Environment. Published by the Consortium and obtainable through NACA, Bangkok, Thailand. 17 pp. |
| Briggs, M., Funge-Smith, S., Subasinghe, R. & Phillips, M. 2004. Introductions and movement of Penaeus vannamei and Penaeus stylirostris in Asia and the Pacific. FAO Regional Office for Asia and the Pacific. RAP Publication 2004/10:1–12. |
| Browdy, C.L., Moss, S.M., Lotz, J.M., Weirich, C.R., Otoshi, C.A., Ogle, J.T., Macabee, B.J., Montgomeries, A.D. & Matsuda, E.M. 2003. Recent USMSFP advances in the development of biosecure environmentally sound superintensive shrimp production systems. p. 35 In: Abstracts of Aquaculture America 2003. World Aquaculture Society, Baton Rouge, Louisiana, USA. |
| FAO. 1995. Code of Conduct for Responsible Fisheries. FAO, Rome, Italy. 41 pp. |
| Macabee, B.J., Bruce, J.W., Weirich, C.R., Stokes, A.D. & Browdy, C.L. 2003. Use of super-intensive greenhouse-enclosed raceway systems for the production of juvenile Litopenaeus vannamei. p. 169 In: Abstracts of Aquaculture America 2003. World Aquaculture Society, Baton Rouge, Louisiana, USA. |
| Parker, J.C., Conte, F.S., MacGrath, W.S. & Miller, B.W. 1974. An intensive culture system for penaeid shrimp. Proceedings of the World Mariculture Society, 5:65–79. |
| Pérez Farfante, I. 1969. Western Atlantic shrimps of the genus Penaeus. Fishery Bulletin, 67(3): 461–591. |
| Pérez Farfante, I. & Kensley, B. 1997. Penaeoid and sergestoid shrimps and prawns of the world. Keys and diagnoses for the families and genera. Memoires du Museum National d'Historie Naturelle, Paris, France. 233 pp |
| Samocha, T.M., Lawrence, A.L., Collins, C.A., Castille, F.L., Bray, W.A., Davies, C.J., Lee, P.G. & Wood, G.F. 2004. Production of the Pacific white shrimp, Litopenaeus vannamei, in high-density greenhouse enclosed raceways using low-salinity groundwater. |
| Taw, N. 2005. Indonesia Shrimp Production. Presented in the Indonesian shrimp farmers session of World Aquaculture 2005, May 9–13, 2005, Nusa Dua, Bali, Indonesia. Charoen Pokphand, Jakarta Indonesia. 18 pp. |
| Wyban, J.A. & Sweeney, J.N. 1991. Intensive shrimp production technology. High Health Aquaculture, Hawaii, USA. 158 pp. |