INTRODUCTION
Over the past two or three decades, there has been a significant upsurge of interest in the use of fodder trees and shrubs as sources of protein and other nutrients for non-ruminant animals. Although considerable attention has been given to the use of leaf meals derived from these trees and shrubs, there is a growing realisation that the seeds of these species may serve as protein sources in their own right and limited work has been published in this area.
Much attention has been focused on leaf meals from Leucaena leucocephala and Manihot esculenta (cassava) but, more recently, interest has turned to leaf meals derived from other sources such as Gliricidia sepium, Robinia pseudoacacia (black locust or false acacia), Cajanus cajan (pigeon pea), Sesbania sesban, Prosopis spp. (mesquite) and Albizia spp. It is clear that the leguminous species predominate in this group due to their relatively high crude protein (CP) contents and to the ability of some species to thrive in adverse soil and climatic conditions. While the potential exists for the wider utilisation of the seeds of fodder trees in non-ruminant diets the limited data in the literature relate to the grain of just one legume: Cajanus cajan.
The purpose of this paper is to critically review current knowledge on the role of leaf meals and seeds derived from fodder trees and shrubs in monogastric nutrition, identify chemical constraints, formulate practical recommendations where appropriate and to identify research and development priorities where potential exists for exploitation of novel species with specific reference to developing countries. Chemical constraints to the use of browse species will also be considered.
FEEDING TRIALS WITH LEAF MEALS
The relatively high crude protein (CP) content of leaf meals in comparison with that of cereals is well recognised. However, the fibre component is also a major fraction of the dry matter (DM). In many instances the fibre content of leaf meals may equal or exceed CP concentrations. Consequently, digestibility of the CP fraction of many leaf meals is low which tends to depress overall CP digestibility when leaf meals constitute a significant proportion of the diet (Tangendjaja et al., 1990).
Although lysine concentrations in leaf meals are considerably higher than those of cereal grains and certain by-products such as coconut oil meal, they are somewhat inferior to those of soyabean meal and fish meal. It follows that leaf meals cannot be expected to fully replace high quality ingredients in diets for monogastric animals. Deficiencies of the sulphur containing amino acids add a further dimension to the nutritional limitations of leaf meals. For example, biological values (BV) for cassava leaf meal range from 0.49 to 0.57 which may be enhanced to 0.80 on supplementation with methionine (Eggum, 1970). Although this leaf meal confers a superior dietary amino acid profile relative to by-products such as coconut oil meal, this difference is not reflected in BV determinations with pigs (Ravindran et al., 1987).
Further insight into the nutritional value of leaf meals is provided by a consideration of metabolisable energy (ME) content. The limited data point to extremely low ME values for Leucaena (D'Mello and Acamovic, 1989) and Robinia as determined with poultry, but the ME content of cassava leaf meal appears to be appreciably higher, not only for poultry but also for pigs.
Leucaena
Consistent with the low digestibility of CP and the inadequate ME content, graded additions of Leucaena leaf meal induce dose-related depressions in growth of chicks even when maize oil is used to compensate for the low ME value of the leaf meal. The adverse effects of Leucaena on the growth of young chicks is now well established (see D'Mello and Acamovic, 1989). Moreover, with older broiler chicks, D'Mello et al. (1987) showed that diets containing 100g leaf meal/kg significantly reduced growth without affecting DM intakes. On the other hand pigs given diets containing Leucaena leaf meal at 100 g/kg grew significantly faster than control animals but live weight gain declined progressively with higher inclusions (Malynicz, 1974). It should be noted that in this study Leucaena was substituted for a commercial ration containing 180g CP/kg diet and, consequently, all Leucaena diets had higher CP concentrations than the control diet. Furthermore, all diets were supplemented with ferrous sulphate to counteract possible toxicity arising from the non-protein amino acid, mimosine, present in the leaf meal. The addition of ferrous sulphate to the mimosine-free control diet was unnecessary and may have depressed growth of pigs on this diet.
Despite the favourable digestibility of the CP fraction of Leucaena leaf meal for rabbits (Raharjo et al., 1986), graded additions of this legume to a control diet precipitated progressive depressions in growth such that, at the 400g/kg inclusion, weight gain was less than 40% of control values (Tangendjaja et al., 1990).
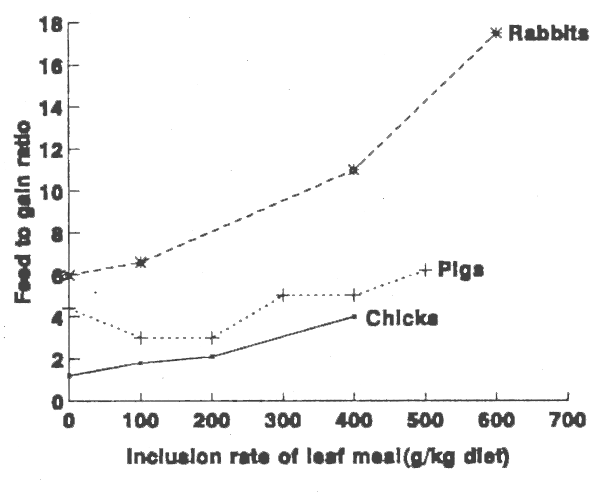
Concomitant with these growth responses is a consistent and striking increase in feed:gain ratios for the three classes of animals when dietary inclusion rates of Leucaena leaf meal exceed 200g/kg (Figure 1). The data for chicks relate to two separate studies by D'Mello and Acamovic (1982) and D'Mello et al. (1987) in which oil additions to diets were held at a constant level irrespective of inclusion rates of Leucaena. Although the studies of Malynicz (1974) show significant improvements in feed conversion ratios on inclusion of Leucaena at rates of up to 200 g/kg diet (Figure 1), more recent studies by Chen et al. (1981) indicate no differences in these ratios between the control diet and diets containing up to 160g leaf meal/kg. The decrease in feed efficiency of rabbits given high Leucaena diets (Figure 1; Tangendjaja et al., 1990) is all the more remarkable given the herbivorous nature of this species. It is apparent that coprophagy confers no special advantage as regards the ability of rabbits to utilise Leucaena-based diets.
FIGURE 1. Effect of Leucaena leaf meal on feed to gain ratios of growing chicks, pigs and rabbits.
Egg production in hens is also adversely affected by dietary inclusion of Leucaena leaf meal. For example Vohra et al. (1972) showed that while inclusion rates of 50 and 100g/kg diet elicited satisfactory laying performance, the diet containing 200g/kg precipitated severe reductions in food intake and egg production. These deleterious effects occurred despite adjustments to the ME content of the diet by addition of oil. It should be noted that the duration of this study was only five weeks. Earlier studies over eight weeks (Mateo et al., 1970) employing pair-feeding techniques indicated that the depression in egg production was specifically attributable to the intake of Leucaena leaf meal.
Cassava
Although cassava leaf meal has been accorded with a favourable ME value, graded inclusion of this leaf meal as a replacement for soyabean meal and maize in practical-type diets elicits progressive depressions in growth rates of both growing chickens and pigs (Ross and Enriquez, 1969; Ravindran, 1990). In addition, cassava leaf meal induces greater reductions in growth rate of chickens than dehydrated alfalfa meal at all inclusion levels from 50 to 200 g/kg diet (Ross and Enriquez, 1969). However, if substitutions of cassava leaf meal are accomplished at the expense of poor quality ingredients such as coconut oil meal in a control diet, then dietary inclusion rates of cassava leaf meal of up to 150g/kg may be employed without reducing growth of broiler chicks (Ravindran et al., 1986). Similarly, Ravindran et al., (1987) showed that performance of growing pigs was unaffected by cassava leaf meal concentrations of up to 267g/kg diet when the leaf meal was substituted for coconut oil meal. At the 400g/kg dietary inclusion, cassava leaf meal significantly reduced average daily gains of pigs. A different picture emerges, however, if this leaf meal is used as a replacement for good quality protein sources. Thus, Ravindran (1990) substituted cassava leaf meal at rates of 100, 200 and 300g/kg diet for maize and soyabean meal and observed significant reductions in live weight gains of pigs with each level of the leaf meal.
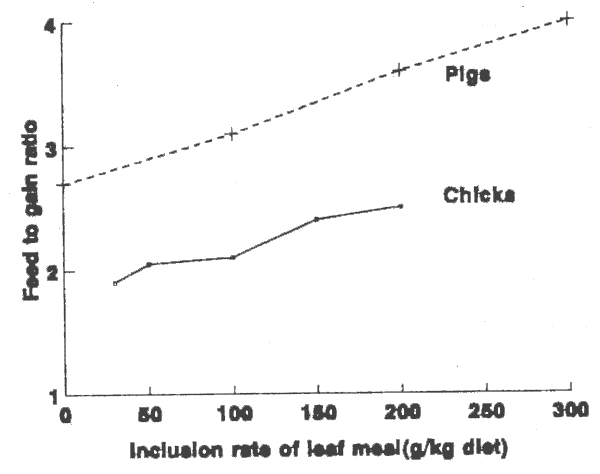
The effects of cassava leaf meal as a replacement for soyabean meal and maize are most clearly seen in the feed: gain ratios depicted in Figure 2.
FIGURE 2. Effect of cassava leaf meal on feed to gain ratios of growing chicks and pigs.
Other Leaf Meals
Limited work with leaf meals from Robinia, Sesbania and Gliricidia suggests poor replacement values for conventional ingredients in diets for poultry. In the case of Sesbania, there are signs of acute toxicity with inclusion rates as low as 100g/kg diet, with 100% mortality in chickens fed Sesbania at 300g/kg diet (Brown et al., 1987). Pigeon pea leaf meal may be more acceptable as a feed component for laying hens (Udedibie and Igwe, 1989). However, feed efficiency declines on inclusion of this leaf meal and body weight losses may also occur.
POTENTIAL OF GRAIN FROM FODDER TREES
On the basis of chemical composition, the grain of fodder trees should offer greater potential as protein and energy sources for non-ruminants than do their corresponding leaf meals. Thus CP concentrations have been reported to be over 30% higher in the seed than in the leaf of Leucaena leucocephala and, although CP concentrations in pigeon pea are similar in the seed relative to the leaf, fibre content of the former is considerably lower (D'Mello, 1987) leading to favourable ME values for poultry.
The feeding value of pigeon pea seed is reasonably well documented. In one investigation (Tangtaweewipat and Elliott, 1989), live weight gains of broilers were unaffected by dietary inclusion of raw pigeon pea seed even at levels of 500g/kg. Pigeon pea seed was substituted for maize and soyabean meal in the control diet. Feed conversion efficiency, however, deteriorated with diets containing 400 and 500g seed/kg. It should be noted that ME values for the seed were somewhat low at around 8 MJ/kg DM and consequently graded levels of oil were added with increasing levels of pigeon pea seed. On the other hand, the results of two trials with laying hens indicate that raw pigeon pea seed induces adverse effects on egg production and feed efficiency when inclusion rates of 300–400 g/kg diet are used (Tangtaweewipat and Elliott, 1989; Udedibie and Igwe, 1989). Body weight losses occurred consistently with diets containing 400g seed/kg, a response similar to that observed with lower dietary concentrations of the leaf meal of pigeon pea. Deleterious effects also occur in pigs fed raw pigeon pea seed at 300g/kg diet (Visitpanich et al., 1985).
The nutritive value of seeds from other fodder trees and shrubs has not been examined on any systematic basis but potential exists for exploitation of a variety of seeds, particularly those from leguminous browse species.
UPGRADING OF LEAF MEALS AND GRAIN
It will be apparent from the foregoing account that the observed nutritive value of leaves and seeds of fodder trees frequently falls considerably short of that expected from their chemical composition. The constraints to enhanced utilisation of these commodities reside chiefly in factors such as fibre content, the presence of anti-nutritive compounds and deficiencies of certain essential amino acids.
The lower fibre contents of seeds relative to leaf meals from fodder trees inevitably means superior quality of the former. Thus the seed of pigeon pea has an ME value for poultry and pigs, which is considerably in excess of the ME content of any of the leaf meals examined thus far.
The leaves and seeds of fodder trees and shrubs are known to contain a wide array of compounds which are capable of reducing performance of animals. A complex combination of different groups of compounds such as toxic amino acids, tannins, cyanogenic glycosides and proteins may occur in a single tree species. The presence of such a disparate range of deleterious compounds has provided the stimulus for much work on methods of detoxification and upgrading of both leaves and seeds. The production of leaf meals for non-ruminant feeding inevitably involves at least some measure of processing. Sun-drying is the method of choice in the tropics and this treatment alone can be an effective method for the removal of anti-nutritive substances in some leaf meals. Thus, sun-drying of cassava leaves may reduce HCN concentrations by as much as 90% (Ravindran et al., 1987).
Thermal processing is an effective method of upgrading plant products through the inactivation of most of the heat labile anti-nutritive substances. In this respect, Visitpanich et al. (1985) were able to reduce trypsin inhibitor activity by heating the grain of pigeon pea to 124°C for 15 minutes. This treatment eliminated all adverse effects on performance of pigs fed 300g seed/kg diet. Heat treatment is a less effective procedure for the upgrading of leaf meals derived from Leucaena, Robinia and Sesbania and, in the case of cassava leaves, boiling may reduce the availability of amino acids (Eggum, 1970).
Another strategy for upgrading leaf meals involves the use of additives. Ferric sulphate and polyethylene glycol (PEG) have been used to complex with mimosine and tannins respectively, with marked improvements on the growth of chicks fed Leucaena-based diets (D'Mello and Acamovic, 1989). Methionine supplementation appears to offer the most viable procedure for upgrading cassava leaf meal for poultry and pigs, in combination with energy-rich additives such as coconut oil (Ravindran, 1990). The response to methionine is presumably a reflection not only of the low content and availability of methionine but also of the need for this amino acid in detoxification of residual HCN.
RECOMMENDATIONS
The scope for leaf meals as protein sources in monogastric nutrition is limited primarily by their low contents of digestible CP, ME and sulphur amino acids and by increased bulkiness of these products. Dietary inclusion rates will depend to a significant extent on the protein sources they are intended to replace. Thus the replacement value of leaf meals is relatively low in diets based on good quality protein sources such as soyabean meal and fish meal. On the other hand, in diets based on poor quality raw materials such as coconut meal or cottonseed cake, higher substitution rates for leaf meals have been recommended. It is salutary to note, however, that, even under these conditions and despite their superior lysine content, leaf meals are unlikely to completely replace poor quality feedstuffs without deleterious consequences.
As regards Leucaena leaf meal, it would not be prudent to exceed inclusion rates of 50g/kg diet for broiler chicks and 100g/kg diet for laying hens, growing pigs and rabbits. In the latter cases, provision of additional energy from coconut oil and of ferrous sulphate may be necessary adjuncts, which may, nevertheless, still not promote optimum feed conversion ratios.
Recommended rates for cassava leaf meal are somewhat better if methionine and oil supplements are used. Up to 150g/kg diet may be used for broiler chicks, and diets for growing pigs may contain up to 300 g/kg without compromising feed efficiency. The available evidence precludes any role in monogastric nutrition, for leaf meals derived from Gliricidia, Robinia, pigeon pea or Sesbania as these are associated with adverse effects and even acute toxicity in some cases.
Considerable potential exists for the grain of pigeon pea in that heat processing offers an effective method of detoxification of protease inhibitors with consequent enhancement of nutritional value for poultry and pigs. Inclusion rates of up to 300g/kg diet have been found to be satisfactory for growing pigs providing that the seed is first subjected to thermal processing procedures. It should be stressed that the use of supplements and heating procedures should not be undertaken without an economic assessment of efficacy.
CONCLUSIONS
The exploitation of under-utilised feedingstuffs is destined to continue in an effort to mitigate regional shortages of protein sources. In this context, leaf meals may have an important but limited role to play in the feeding of non-ruminant animals. Although the presence of anti-nutritive substances undoubtedly contributes to the poor nutritional value of some leaf meals, it is likely that their high fibre content represents the major constraint for monogastric animals. However, the removal of the fibrous components in the production of leaf protein concentrates (LPC) from fodder trees and shrubs is not consistently accompanied by enhanced nutritional value (Cheeke et al., 1980). It appears unlikely that LPC production would be a commercially viable procedure for upgrading leaf meals even if nutritional value can be improved.
In the long-term, any expansion of the poultry and pig industries in the tropics cannot be sustained with the extended use of leaf meals alone and alternative strategies should be considered. One attractive option may be the exploitation of grains not only from fodder trees but also from under utilised or novel legumes such as Canavalia ensiformis (jack bean); Cyamopsis tetragonoloba (guar); Psophocarpus tetragonolobus (winged bean); and Vigna unguiculata (cowpea). These seeds are not without major limitations emanating from their anti-nutritive components but in many instances a relatively simple detoxification procedure can yield a product nutritionally comparable to soyabean meal (D'Mello et al., 1983; D'Mello and Walker, 1991). The extended utilisation of by-products such as coconut oil meal and rice bran should also be pursued with renewed impetus. These, together with the wider use by non-ruminants of protein sources from fodder trees and shrubs, merit high priority in the developing countries.
Bibliography
Brown, G.L., Barnes, D.A., Rezende, S.A. and Klasing, K.C. 1987. Yield, composition and feeding value of irrigated Sesbania sesban. Animal Feed Science and Technology 18: 247–255.
Cheeke, P.R., Telek, L., Carlsson, R. and Evans, J.J. 1980. Nutritional evaluation of leaf protein concentrates prepared from selected tropical plants. Nutrition Reports International 22: 717–721.
Chen, M.T., Chang, T. and Lin, L.S. 1981. Feeding efficiency of Leucaena leaf meal used in pig rations. Leucaena Research Reports 2: 46.
D'Mello, J.P.F. 1987. Underexploited tropical feedingstuffs for poultry. World Review of Animal Production 23: 37–43.
D'Mello, J.P.F. and Acamovic, T. 1982. Apparent metabolisable energy value of dried Leucaena leaf meal for young chicks. Tropical Agriculture (Trinidad) 59: 329–332.
D'Mello, J.P.F. and Acamovic, T. 1989. Leucaena leucocephala in poultry nutrition-a review. Animal Feed Science and Technology 26: 1–28.
D'Mello, J.P.F. and Walker, A.G. 1991. Detoxification of jack beans (Canavalia ensiformis): studies with young chicks. Animal Feed Science and Technology 33: 117–127.
D'Mello, J.P.F., Acamovic, T. and Walker, A.G. 1983. Nutrient content and apparent metabolisable energy values of full-fat winged beans (Psophocarpus tetragonolobus) for young chicks. Tropical Agriculture (Trinidad) 60: 290–293.
D'Mello, J.P.F., Acamovic, T. and Walker, A.G. 1987. Evaluation of Leucaena leaf meal for broiler growth and pigmentation. Tropical Agriculture (Trinidad) 64: 33–35.
Eggum, B.O. 1970. The protein quality of cassava leaves. British Journal of Nutrition 24: 761–768.
Malynicz, G. 1974. The effect of adding Leucaena leucocephala meal to commercial rations for growing pigs. Papua New Guinea Agricultural Journal 25: 12–14.
Mateo, J.P., Labadan, M.M., Abilay, T.A. and Alandy, R. 1970. Study of paired feeding of pullets using high levels of ipil-ipil (Leucaena leucocephala Lam de wit) leaf meal. The Philippine Agriculturist 54: 312–318.
Raharjo, Y.C., Cheeke, P.R., Pat ton, N.M. and Supriyati, K. 1986. Evaluation of tropical forages and by-product feeds for rabbit production. I. Nutrient digestibility and effect of heat treatment. The Journal of Applied Rabbit Research 9: 56–66.
Ravindran, V. 1990. Feeding value and digestibility of cassava leaf meal for growing pigs. Proceedings of the fifth Asian Australasian Animal Production Congress 3: 20.
Ravindran, V., Kornegay, E.T., Rajaguru, A.S.B., Potter, L.M. and Cherry, J.A. 1986. Cassava leaf meal as a replacement for coconut oil meal in broiler diets. Poultry Science65: 1720–1727.
Ravindran, V., Kornegay, E.T., Rajaguru, A.S.B. and Notter, D.R. 1987. Cassava leaf meal as a replacement for coconut oil meal in pig diets. Journal of the Science of Food and Agriculture 41: 45–53
Ross, E. and Enriquez, F.Q. 1969. The nutritive value of cassava leaf meal. Poultry Science 48: 846–853.
Tangendjaja, B., Raharjo, Y.C. and Lowry, J.B. 1990. Leucaena leaf meal in the diet of growing rabbits: evaluation and effect of a low-mimosine treatment. Animal Feed Science and Technology 29: 63–72.
Tangtaweewipat, S. and Elliott, R. 1989. Nutritional value of pigeon pea (Cajanus cajan) meal in poultry diets. Animal Feed Science and Technology 25: 123–135.
Udedibie, A.B.I. and Igwe, F.O. 1989. Dry matter yield and chemical composition of pigeon pea (C. cajan) leaf meal and the nutritive value of pigeon pea leaf meal and grain meal for laying hens. Animal Feed Science and Technology 24: 111–119.
Visitpanich, T., Batterham, E.S. and Norton, B.W. 1985. Nutritional value of chickpea (Cicer arietinum) and pigeon pea (Cajanus cajan) meals for growing pigs and rats. I. Energy content and protein quality. Australian Journal of Agricultural Research 36: 327–335
Vohra, P., Herrick, R.B., Wilson, W.O. and Siopes, T.D. 1972. The use of ipil-ipil (Leucaena leucocephala) in the diets of laying chickens and laying quail. The Philipine Agriculturist 56: 104–113.