| Prepared at the 53rd JECFA (1999) and published in FNP 52 Add 7 (1999), superseding specifications prepared at the 35th JECFA (1989) and published in FNP 49 (1990). ADI "not specified" established at the 35th JECFA in 1989. | |
| SYNONYMS | INS No. 297 |
| DEFINITION | |
| Chemical names | trans-Butenedioic acid, trans-1,2-Ethylene-dicarboxylic acid |
| C.A.S. number | 110-17-8 |
| Chemical formula | C4H4O4 |
| Structural formula | 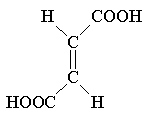 |
| Formula weight | 116.07 |
| Assay | Not less than 99.0% calculated on the dried basis |
| DESCRIPTION | Odourless, white crystalline powder or granules |
| FUNCTIONAL USES | Acidity regulator, flavouring agent (see "Table of specifications of flavouring agents") |
| CHARACTERISTICS | |
| IDENTIFICATION | |
| Solubility (FNP 5) | Soluble in ethanol; slightly soluble in water and in diethyl ether |
| pH (FNP 5) | 2.0 - 2.5 (1 in 30 solution) |
| 1,2-Dicarboxylic acid | Place 50 mg of the sample in a test tube, add 2 to 3 mg of resorcinol and 1 ml of sulfuric acid, shake, heat at 130� for 5 min and cool. Dilute with water to 5 ml and add sodium hydroxide solution (2 in 5) dropwise to render the solution alkaline, cool and dilute with water to 10 ml. A greenish blue fluorescence is observed under an ultraviolet lamp. |
| Test for double bond | Add 10 ml of water to 0.5 g of the sample and dissolve by boiling. Add 2 or 3 drops of bromine TS to the hot solution. The colour of bromine TS disappears. |
| PURITY | |
| Loss on drying (FNP 5) | Not more than 0.5% (120�, 4 h) |
| Melting range (FNP 5) | 286 - 302� (closed capillary, rapid heating) |
| Sulfated ash (FNP 5) | Not more than 0.1% Test 2 g of the sample (Method I) |
| Maleic acid (FNP 5) | Not more than 0.1% |
| Lead (FNP 5) | Not more than 2 mg/kg Prepare a sample solution as directed for organic compounds in the Limit Test and determine the lead content by atomic absorption spectroscopy (FNP 5). |
| METHOD OF ASSAY | Transfer about 1 g of the sample, accurately weighed, into a 250-ml Erlenmeyer flask, add 50 ml of methanol, and dissolve the sample by warming gently on a steam bath. Cool, add phenolphthalein TS, and titrate with 0.5 N sodium hydroxide to the first appearance of a pink colour that persists for at least 30 sec. Perform a blank determination and make any necessary correction. Each ml of 0.5 N sodium hydroxide is equivalent to 29.02 mg of C4H4O4. |