1.1 Introduction
1.2 Parasite groupings
1.3 Identification procedure
As a first step in the investigation of helminth infections of ruminants, it is important to establish what parasite species are present in an area, country or region. This may already be well documented, in which case this step is not necessary. However, the dominant parasites in an area can change, particularly as livestock management practices change, so existing parasite inventories as well as distribution data based on old studies may require updating.
Initial surveys should be kept extremely simple. They are intended to identify parasites present rather than to determine their importance, a later procedure.
While it may be relatively easy to identify some helminth parasites of ruminants simply on the basis of the tissues and organs in which they are located, it may be difficult to identify gastro-intestinal parasites. Many of the gastro-intestinal nematodes look alike but they can be identified down to a species level by microscopical examination. In addition, most of them live in specific sites in the intestinal tract, which helps in the identification process. Since different species have different pathogenic effects, it is important to know which broad groups are present in a herd or area. Furthermore, some of these parasites have very different development times, both outside and inside the host, a knowledge of which is important for effective control measures.
1.2.1 Nematodes
1.2.2 Cestodes
1.2.3 Trematodes
1.2.4 Protozoa
Helminth parasites can be classified into four broad groups.
|
Haemonchus |
Bunostomum (hookworms) |
|
Ostertagia |
Strongyloides |
|
Trichostrongylus |
Oesophagostomum |
|
Mecistocirrus |
Chabertia ovina |
|
Cooperia |
Trichuris (whipworms) |
|
Nematodirus |
Dictyocaulus |
|
Protostrongylus |
Parafilaria |
|
Muellerius |
Onchocerca |
|
Toxocara |
Setaria |
|
Stephanofilaria |
Thelazia |
|
Monezia |
Cysticercus bovis |
|
Avitellina |
Cysticercus tenuicollis |
|
Thysaniezia |
Coenurus cerebralis |
|
Stilesia |
Hydatid cysts |
|
Fasciola |
Paramphistomum |
|
Dicrocoelium |
Schistosoma |
Coccidia* (Eimeria)
* Members of this family Eimeriidae are referred to here as Coccidia
As the protozoan parasites usually referred to as coccidia are commonly found in the intestines of ruminants, this manual will cover the main features of the epidemiology, diagnosis and control of these parasites.
1.3.1 Post-mortem examination
1.3.2 Identification of parasite eggs in faecal samples from live animals
The identification of parasites present in an area can be carried out in the following two ways:
· a post-mortem examination of animals(a) that have died from acute or chronic diseases or
(b) that have been slaughtered at a slaughterhouse/slaughter place· the identification of parasite eggs and larvae present in faecal samples from live animals.
1.3.1.1 Gastro-intestinal tract
1.3.1.2 Liver
1.3.1.3 Lungs
1.3.1.4 Other organs and tissues
Generally the most useful data will be acquired from young animals and those that have not been recently dewormed. The entire gastro-intestinal tract (rumen to rectum) should be obtained from slaughterhouses/places, butchers, veterinary diagnostic centres, etc., for the purpose of calculating the total number of parasites present (see a description of this procedure in section 4.3), as well as identifying the species found. This procedure should be performed towards the end of the rainy season.
Table 1.1 PARASITES LOCATED IN THE GASTRO-INTESTINAL TRACT AND THEIR EFFECT
|
Site |
Host species |
Parasites |
Action |
|
Rumen |
Cattle |
Paramphistomum |
Mucosal damage |
|
Abomasum
|
Cattle, sheep, goats
|
Haemonchus |
Blood sucking |
|
Mecistocirrus |
Blood sucking |
||
|
Ostertagia |
Mucosal damage |
||
|
Trichostrongylus axei |
Mucosal damage |
||
|
Small intestine
|
Cattle, sheep, goats
|
Trichostrongylus |
Mucosal damage |
|
Bunostomum |
Blood sucking |
||
|
Cooperia |
Mucosal damage |
||
|
Nematodirus |
Mucosal damage |
||
|
Strongyloides |
Mucosal damage |
||
|
Paramphistomum larva |
Mucosal damage |
||
|
Coccidia |
Mucosal damage |
||
|
Monezia |
Minimal |
||
|
Large intestine
|
Cattle, sheep, goats
|
Trichuris |
Blood sucking |
|
Oesophagostomum |
Mucosal damage, nodules |
||
|
Sheep
|
Coccidia |
Mucosal damage |
|
|
Chabertia ovina |
Minimal |
The identification of parasite species is described in Chapter 4.
Most gastro-intestinal parasites live in distinct sites of the intestinal tract.
The location of parasites in the intestinal tract of different ruminant species
is shown in Table 1.1. Parasites vary also in their geographical distribution
which depends particularly on climate (especially rainfall), vegetation and
livestock density.
The liver should be examined for migratory tracts (caused by immature Fasciola and Cysticercus tenuicollis larvae), swellings (hydatid cysts and Schistosoma nodules), enlarged bile ducts (adult Fasciola) and relatively large transparent cysts attached to the surface of the liver (Cysticercus tenuicollis). The presence of parasites associated with these lesions can be confirmed by incisions in the liver parenchyma. Following incision, Dicrocoelium and Stilesia may be observed in the bile ducts.
Table 1.2 PARASITES LOCATED IN THE LIVER AND THEIR EFFECT
|
Site |
Host species |
Parasites |
Action |
|
Parenchyma
|
Cattle, sheep, goats
|
Immature flukes |
Destruction of tissue, fibrosis |
|
Larval stages of Cysticercus tenuicollis |
Fibrotic tracts, calcified nodules |
||
|
Eggs of schistosomes |
Granulomas, destruction of tissue |
||
|
Hydatid cysts |
Pressure atrophy |
||
|
Bile duets
|
Cattle, sheep, goats
|
Mature flukes |
Blood sucking, destruction of bile ducts, fibrosis |
|
Dicrocoelium |
Minimal (fibrosis) |
||
|
Stilesia |
Minimal |
||
|
Liver capsule |
Cattle, sheep, goats |
Cysticercus tenuicollis cysts |
Minimal |
Several species of lungworms may cause pathological changes and the post-mortem examination of the lungs may reveal signs of bronchitis, pneumonia, pleuritis, swellings and nodules. Adult Dictyocaulus species are found in the trachea and the main bronchi, Protostrongylus in the terminal bronchioles and Muellerius are usually embedded in grey nodules formed around the alveoli. The lungs are an organ in which hydatid cysts may be located and associated swellings can often be seen or palpated. Occasionally nodules containing liver flukes may be found in the lungs.
Table 1.3 PARASITES LOCATED IN THE LUNGS AND THEIR EFFECT
|
Site |
Host species |
Parasite |
Action |
|
Trachea |
Cattle, sheep, goats |
Dictyocaulus |
Tracheitis, |
|
Bronchi |
Cattle, sheep, goats |
Dictyocaulus |
Bronchitis pneumonia |
|
Bronchioles |
Sheep, goats |
Protostrongylus |
Pneumonia, pleuritis |
|
Alveoli |
Sheep, goats |
Muellerius |
Nodules |
|
Lung tissue |
Cattle, buffalo, sheep, goats |
Hydatid cysts |
Tissue atrophy |
The larvae of some cestode species may be found in muscle tissues of cattle (Cysticercus bovis) and sheep (Cysticercus ovis). The cysts are 6-9 mm in size and semi-transparent when young. As a result of the host's immune response they gradually degenerate, becoming caseous and eventually calcified. The predilection sites are the heart muscle, the tongue, the masseters and the diaphragm.
Some of the filarial worms are found in nodules located in ligaments and tendons (Onchocerca species), in muscles (Onchocerca) and in the skin or subcutaneous tissue (Onchocerca, Parafilaria, Stephanofilaria). Others are found living in the body cavities (Setaria).
Parasites of the conjunctival sac and/or lachrymal duct (Thelazia) may be very prevalent in some areas.
Fresh faecal samples should be taken from a small number of animals. These samples should be taken preferably towards the end of the rainy season from young animals and those that have not recently been dewormed.
Collected faecal samples should be subjected to a flotation and sedimentation procedure for separating and concentrating parasite eggs (see section 3.3) and examined microscopically. In addition the sample should be subjected to a Baerman examination for isolation of lungworm larvae (see section 3.6). The eggs of some parasites are easy to differentiate. The following are examples of these parasites.
Table 1.4 PARASITES LOCATED IN VARIOUS ORGANS AND TISSUES AND THEIR EFFECT
|
Site |
Host species |
Parasite |
Action |
|
Muscle |
Cattle, buffalo, |
Cysticercus |
Minimal |
|
Ligaments, tendons |
Cattle, buffalo |
Onchocerca |
Minimal |
|
Skin and subcutaneous tissue |
Cattle, buffalo |
Onchocerca, |
Minimal |
|
Body cavities |
Cattle, buffalo |
Setaria |
Minimal |
|
Central nervous system
|
Cattle, buffalo |
Setaria larvae |
Neurological disturbance |
|
Sheep, (goats, |
Coenurus cerebralis |
Neurological disturbance |
|
|
Circulatory system |
Cattle, sheep, goats |
Schistosomes |
Minimal * |
|
Eye |
Cattle, sheep goats |
Thelazia |
Minimal |
* Note: Most of the pathogenic effect of schistosomes is related to the migration of the eggs.
Coccidia oocysts
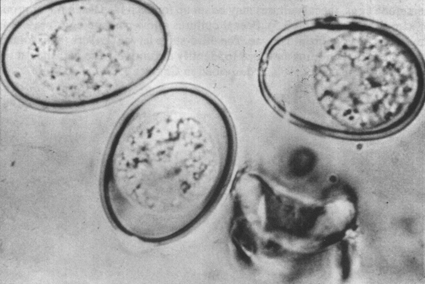
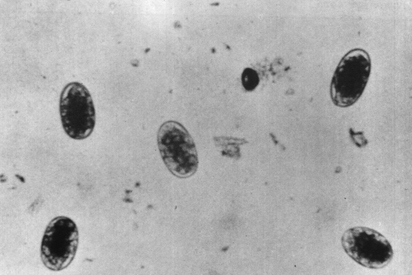
The eggs of other parasites, however, are similar in size and structure and cannot easily be differentiated. These include the Trichostrongyles, Oesophagostomum and Bunostomum. To differentiate and identify these nematode eggs, faecal cultures may be set up for each faecal sample or group of samples (see section 3.5). Faecal cultures allow parasite eggs present in the faeces to develop into larvae. The third-stage larvae (L3) are then isolated from the faecal cultures and used to identify definitively the parasites at the species or genus level. This is described in Chapter 3.