I. Mueller-Harvey, A.B. McAllan, M.K. Theodorou and D.E. Beever
Institute of Grassland and Animal Production, Animal and Grasslands Research Station, Hurley, Berkshire, UK
Introduction
Occurrence of phenolics in plants
Classes of phenolic compounds
Analytical methods
The role of phenolics in plants and implications for breeding programmes
Mechanism of phenolics interaction with cell constituents and their digestion
Effect of polyphenolic compounds on voluntary feed intake
Effect of polyphenolics on the sites of carbohydrate and protein digestion
Effects of plant phenolics on rumen microbial activity
Effect of polyphenolics on animal performance
Conclusion
References
DISCUSSION
Crop residues are important animal feeds in many developing countries. Up to 80% of their dry matter is cell-wall polysaccharides. The utilisation of this energy source depends on both the physical properties and the chemical composition of the residues. Although our knowledge of cell wall chemistry is still incomplete, it is known that phenolic compounds are an integral part of the hemicellulose fraction (Mueller-Harvey et al, 1986). Some tropical crop residues contain about 3% simple phenolic acids by weight. Other types of more complex phenolic compounds are known to be present in cereal grains (Ramachandra et al, 1977) and cereal crop residues (Jambunathan et al, 1986).
This review first presents the various classes of phenolic compound found in plants and current methods of analysis. The 'chemical defense' hypothesis is examined critically in the light of recent findings. The purported roles of phenolics in plants are presented before the interactions between phenolics and other cell constituents (carbohydrates and proteins) are reviewed. This is followed by discussion of the apparent effects of phenolic compounds on ruminant nutrition and rumen microbial metabolism. The review concludes by highlighting areas in which more information is required.
Phenolics occur in several forms in plants: (1) as soluble compounds extractable with water (garlic acid esters) or with methanol and aqueous acetone (proanthocyanidins, flavonols, flavonolglycosides) and (2) in non-extractable forms. Phenolics may remain in the residue after extraction as a result of their inherent insolubility, e.g. due to their large molecular weights, or because they are covalently bonded to, or tightly bound in complexes with, other plant constituents (Bears et al, 1985b; Hartley and Buchan, 1979; Hartley and Keene, 1984; Mueller-Harvey et al, 1986).
Structures
Several types of phenolic compound affect the digestion of crop residues, including simple phenolic compounds, such as the cinnamic acids (I) or aldehydes (II) and polyphenolics, such as the 'condensed' (III) and 'hydrolysable' tannins (IV) (Haslam, 1981) (Figure 1).
Polyphenolics
"Vegetable tannins" are a group of polyphenolics, consisting of a large number of structurally very different compounds. The original definition of a "tannin" is a compound "able to convert hide to leather." All tannins contain phenolic groups, but not all phenolics are tannins. Many of these compounds have now been identified. It is increasingly obvious that even the modified definition of a tannin as "a compound that binds to proteins" (Swain and Bate-Smith, 1962) is imprecise and misleading and no longer serves any useful purpose. We will, therefore, try to avoid the word "tannin," except when referring to "condensed" and "hydrolysable tannins."
Qualitative analysis
The literature on the effects of phenolics in animal nutrition has many examples of inappropriate use of analytical methods resulting in unsupported conclusions. This often stems from a poor understanding of the reactions between phenolic compounds and their detection agent. However, it may also frequently be a result of the use of unsuitable preparation procedures. Different types of phenolic have been estimated using unspecific methods. Quantification of phenolics is problematic because of the lack of a general standard compound, together with the likelihood that different phenolic compounds have different nutritional effects. This makes comparative assessments of nutritional responses extremely difficult.
Precautions need to be taken when extracting phenolics (McLeod, 1974; Gartlan et al, 1980). Many phenolic compounds isomerise in sunlight (trans-cis conversions; Kahnt, 1967), react with oxygen in alkaline solution (quinone formation) and with methanol at room temperature and pH 6 (Haslam, 1966).
Plant phenolics have been measured colorimetrically using the Folin-Denis reagent (Swain and Hillis, 1959). This reagent reacts non-stoichiometrically with phenolic and other OH groups, and several reducing agents interfere (Singleton and Rossi, 1965). Despite the lack of specificity for polyphenolics, this reagent has often been used to measure tannin content in forage crops because of its ease of use (Burns, 1963) .
The Folin reagent as modified by Folin and Ciocalteau (1927) gives a better estimate of total phenolic groups (Single ton and Rossi, 1965). This reagent gives a greater colour response with phenols and a lesser response to non-phenolic compounds. Another general reagent for phenolic groups is Prussian blue (Price and Butler, 1977), but this has the disadvantage of a widely varying sensitivity for different compounds. Vanillin reacts under acidic conditions with one group of phenols, called flavanols. When used under the conditions recommended (Sarkar and Howarth, 1976) it is specific for a narrow range of flavanols (including condensed tannins) and dihydrochalcones (Swain and Hillis, 1959; Sarkar and Howarth, 1976).
Heating proanthocyanidins with hydrochloric acid and n- butanol produces strongly coloured anthocyanidins (Swain and Hillis, 1959). However, chlorophylls interfere and the reaction is not quantitative. The yield and type of anthocyanidins differ for each type of proanthocyanidin. Its advantage is its specificity.
Quantitative analysis
In order to assess the effects of phenolics in nutritional studies, quantitative estimates of the amounts present are required. Gallic acid (Singleton and Rossi, 1965), tannic acid (McLeod, 1974), catechin (Price et al, 1978) and chlorogenic acid (Walter and Purcell, 1979) have all been used as reference standards. However, tannic acid is a poor standard because it tends to be a mixture of different compounds, the relative proportions of which vary between samples (King and Pruden, 1970). Using catechin as a standard in the vanillin test over-estimates the concentration of condensed tannins because the reaction kinetics are different for the monomer and the oligomers (Price et al, 1978).
Gravimetric procedures (Temper, 1982) have been used to obtain a direct measure of phenolics thus avoiding the problems associated with quantification in colorimetric assays. The hide powder assay (ALCA, 1956) measures the amount of phenolics adsorbed by the powder. However, Laurent (1975) found that results were variable and Verzele et al (1986) criticised the choice of hide powder because it selectively binds only some compounds from tannic acid mixtures.
Reed et al (1985) developed a method for precipitating soluble phenolics from plant extracts with ytterbium acetate. Precipitation was complete when phenolics accounted for more than 16% of the dry matter. The precipitates contained flavan-3-ols, condensed tannins, garlic acid and hydrolysable tannins, catechin gallates, flavonols and their glycosides (Mueller-Harvey, Reed and Hartley, unpublished data). Little protein or chlorophyll appeared to be co precipitated with the phenolics. This method holds promise for the gravimetric determination of phenolic compounds and modifications are being made to obtain complete precipitation of all phenolics at lower concentrations.
High-performance liquid chromatography (HPLC) (Vance Casteele et al, 1982; Mueller-Harvey et al, 1987) is better able to distinguish among different types of phenolics but separations may be incomplete with complex mixtures of similar compounds, e.g. some mixtures of condensed and hydrolysable tannins.
The metabolic role of phenolics in the plant is still not clear. Feeny (1976) suggested that they serve as chemical defense agents through their astringent taste and by interfering with the digestive enzymes of the predators. Although a negative correlation was found between susceptibility to bird attack and polyphenolic content of sorghum grain (Bullard and Elias, 1980), Coley (1983) found no correlation between the extent of herbivore attack and phenolic content of mature leaves in a tropical forest. Similarly, Bernays (1981) stressed the variability of effects of polyphenolics on insect herbivores and warned against generalization concerning their ecological significance.
A systematic approach by Beart et al (1985a) noted that the principal biosynthetic thrust in plants is towards the product ion of higher molecular weight polyphenolics which are the end-product of the metabolism. Based on the "defense hypothesis" one might assume that such end-product are produced by plants as these compounds are most effective in precipitating the proteins of the herbivores. Beart et al (1985a) therefore investigated the binding strength between various phenolic precursors and end-products with a protein, bovine serum albumin (BSA). To their surprise, they found that one common precursor, pentagalloyl glucose, bound the protein much more strongly than most of the end-products. This led them to doubt the defense hypothesis.
In a similar context in micro-organisms, Bu'Lock (1980) suggested that the secondary metabolism serves to maintain basic metabolism in circumstances not propitious for growth (e.g. nutritional imbalances). This is supported by the observation that the condensed tannin content of Lotus species was higher under low soil fertility than high soil fertility conditions (Barry and Forss, 1983) and that high phenolic contents have been linked with high light intensity, high temperatures and severe drought (Burns, 1966). A further example is that sainfoin grown in the UK had a much lower extractable phenolic content than sainfoin grown in Ethiopia (J.D. Reed, personal communication).
The basic principles of the metabolism of phenolics must be better understood in order to develop strategies for the development of plants with higher nutritive value. For example, if stress conditions cause more phenolic precursors to be synthesised and specific plant enzymes cause their polymerisation, two screening approaches could be employed:
1. To assess the effect of stress on precursor synthesis in different varieties; and
2. To assess the differences in plant enzymes which produce the polyphenolics.
This approach is supported by the feet that different types of precursor occur in different sorghum plants (Watterson and Butler, 1983), and by the discovery of different end products within the Cinnamon species (Nonaka, et al, 1983). It should be possible to exploit such genetic differences in breeding programmes once the nutritional effects of the various phenolics are better understood.
Carbohydrates and simple phenols
Several simple phenolics occur in plants. The derivatives of cinnamic acid are the most abundant; derivatives of benzoic acid and aldehydes occur in smaller amounts (Jung et al, 1983a; Hartley and Keene, 1984). Ferulic and p-coumaric acids are esterified to carbohydrates in plant cell walls (Mueller-Harvey et al, 1986), whereas the aldehydes are apparently linked at their phenolic groups to cell-wall polysaccharides (Hartley and Keene, 1984). It has been suggested that phenolic compounds limit the digestion of carbohydrates (Hartley and Jones, 1978): digestibility of cell-wall carbohydrates is increased when phenolic compounds are released from graminaceous forages by treatment with alkali (Hartley and Jones, 1978).
Very little is known about the type of bonding between polyphenolics and carbohydrates. Beart et al (1985b) proposed that some proanthocyanidins may be covalently linked via ether bridges at C-4 to carbohydrates, in analogy to the C-C bridges in proanthocyanidins, but the evidence for such linkages is circumstantial. Other types of bonding between polyphenolics and carbohydrates have been demonstrated; H+-bridges and hydrophobic interaction binding are important in such complexes.
Ford (1978) observed low digestibilities of cell walls from Desmodium intortum and suggested that this was probably caused by proanthocyanidins complexing with cellulose. More detailed studies showed that some condensed and hydrolysable tannins adsorb to starch (Davis and Hoseney, 1979). McManus et al (1985) studied binding among several polysaccharides and polyphenolics and concluded that the molecular size of the polyphenol and its conformational flexibility are important to the binding, which seems to be pH independent. They also noticed that small changes in the structure of either the polyphenol or the polysaccharide resulted in marked changes in their affinity for each other. Where the association was primarily a surface effect, broad similarities were noted with the analogous complexation with proteins. However, where the polysaccharide was also capable of forming inclusion complexes, significant differences were observed relative to the binding of proteins.
Proteins
The interaction between polyphenolics and proteins has been recognised for much longer than that with polysaccharides. However, we still do not understand the causes of the differences in the effects of polyphenolics among digestion trials. Polyphenolics have been reported to have both beneficial effects (Reed and Soller, 198 7; McLeod, 1974) and detrimental effects (Reed and Soller, 1987; Reddy et al, 1985) on protein metabolism in ruminants.
The complexes formed by the interaction of proteins and phenolics in solution may be either soluble or insoluble (Van Buren and Robinson, 1969; Mole and Waterman, 1985). However, insoluble (non-extractable) phenolics can also complex proteins (Bate-Smith, 1977).
The effect of complex formation on the digestibility of proteins/phenolics and the activity of enzymes is not fully understood. Mole and Waterman (1985) reported that condensed tannins both stimulated and inhibited digestion of complexed proteins by trypsin. Although several enzymes are inhibited by polyphenolics (Butler et al, 1984 and references there in), others retain some or most of their activity whilst complexed (Goldstein and Swain, 1965; Davis and Hoseney, 1979). Thus, enzyme inhibition is not a good measure for so-called "tannins."
The formation of complexes depends on the concentration of both the polyphenolic and the protein, resulting in variable stoichiometries (e.g. ranging between 1:60 and 1:120 for the protein:polyphenol ratio) (McManus et al, 1981; Mole and Waterman, 1985). Precipitation is thought to occur when a hydrophobic outer layer is formed. Thus at appropriate concentrations even simple phenolics, such as pyrogallol and resorcinol, can precipitate proteins from solution (McManus et al, 1981). This example illustrates best why the term "tannin" is obsolete. pH also affects precipitation (Jones and Mangan, 1977; Martin and Martin, 1983). Precipitation is greatest at a pH within one unit of the isoelectric point of the protein (Hagerman and Butler, 1978).
Martin et al (1985) demonstrated that metal ions influence the extent of precipitation between hydrolysable tannins and leaf fraction I protein: Mg2+ and Ca2+ were more effective than Na+ and K+ at bringing about protein precipitation.
Our present understanding of the mechanism of interaction favours both H+ -bond formation and hydrophobic interactions (Goldstein and Swain, 1965; Haslam, 1974; Oh et al, 1980).
As indicated earlier for carbohydrates, complex formation is dependent on both solution conditions and the properties of the phenolics and proteins. Molecular size and conformational flexibility have major effects on the strength of binding between polyphenol and protein. For example, molecular flexibility of certain ellagitannins is less than that of gallotannins due to intramolecular crosslinking: as a result, the ellagitannins bind BSA more weakly than do gallotannins (Bears et al, 1985a; McManus et al, 1985). Proteins with open, loose conformations interact much more strongly with sorghum polyphenolics (Asquith et al, 1987).
Thus, the great specificity of some polyphenolics for certain proteins (Becker and Martin, 1982; Butler et al, 1984; Asquith et al, 1987) can be likened to the specificity between enzymes and substrates. It follows that some proteins can be preferentially precipitated out of solution even in the presence of excess amounts of other proteins (Butler et al, 1984).
It seems reasonable to assume that the strength of binding in such complexes will have important implications on the degradability of complexes at different pH values (Jones and Mangan, 1977), by enzymes or micro-organisms (Martin and Martin, 1983).
Studies are also needed to assess any reactions within the polyphenol-protein complex that may occur during digestion. Beart et al (1985b) predicted that covalent bonds could be formed in such complexes in the ruminant.
Intake of feed containing large amounts of condensed tannins its low (Barry and Duncan, 1984). Barry and Duncan (1984) recorded an increase in both metabolisable energy intake (MEI) and digestible organic matter intake (DOMI) in sheep fed high-polyphenolic Lotus in response to decreasing condensed tannin content when polyethylene glycol (PEG) was used to bind the condensed tannins (Barry and Forss, 1983). Digestibilities of OM, cellulose, hemicellulose and nitrogen also increased. Thirty-two percent of the increase in DOMI could be attributed to increased intake of digestible fibre and a further 32% to increased intake of digestible crude protein. However, an increase in intake of digestible crude protein does not necessarily increase supply of amino acids to the animal, as shown by Thomson et al (1971) working with dried lucerne and sainfoin fed to sheep.
The presence of condensed phenolics in sainfoin (Thomson et al, 1971; Egan and Ulyatt, 1980) and Lotus species (Barry et al, 1986b) has been associated with increased nitrogen retention in sheep. This has been attributed to an increased supply of amino acids to the small intestines as a result of protection of the plant protein from proteolysis in the rumen (Reid et al, 1974).
In Lotus species condensed phenolic contents up to 25 g kg- DM appear to have little effect on rumen carbohydrate digestion but concentrations between 25 and 100 g kg- DM reduce carbohydrate digestion in the rumen in a dose-dependent manner (Barry and Manley, 1986; Barry et al, 1986b).
Barry and Manley (1986) found that, in Lotus based feed, increased polyphenolic content significantly reduced the extent of digestion of OM in the rumen The digestive behaviour of the high-polyphenolic Lotus was compared with the predicted behaviour of the same crop assuming it was a low-polyphenolic crop. The predicted increased flow of organic matter (measured as the sum of its individual components) was 99 g d 1, of which 42% was fibre and the remainder largely crude protein. This supported the suggestion by Barry and Duncan (1984) that polyphenolics both reduce digestion of carbohydrates in the rumen and increase protein outflow. The amount of fibre excreted in the farces was equal to the amount of extra fibre entering the small intestine suggesting that dietary fibre not digested in the rumen was irreversibly bound by polyphenolics. In contrast, approximately 60% of the extra protein entering the duodenum was digested in the small or large intestine. However, in further similar experiments Barry et al (1986b) found that the reduction in carbohydrate digestion in the rumen was compensated by increased post-ruminal digestion and whole tract digestibility appeared unaffected.
In another study, Thomson et al (1971) examined the digestion of dried sainfoin (high condensed tannin) and lucerne (low condensed tannin) in mature sheep. Although the crops were harvested at similar stages of growth and had similar total N contents, cellulose digestibility was significantly higher on sainfoin (78 vs 67) and the extent of cellulose digestion in the rumen was unaffected. In contrast, apparent N digestibility was significantly lower in sainfoin (68 vs 51). However, duodenal amino acid supply from sainfoin was 49% greater (178 vs 119 g d-1) and, despite a small increase in total amino acid flow at the ileum (sainfoin 64, lucerne 52 g d-1), availability of amino acids in the small intestine was 46 g d-1 greater from sainfoin than from lucerne. This response was seen despite a significant reduction in apparent N digestibility (whole tract) and indicates (a) the futility of using apparent N digestibility as a measure of protein value, and (b) a marked positive effect of condensed tannins on protein supply to the host animal.
In this study, no real at tempts were made to determine the origin of duodenal protein on the two diets, but in a parallel publication Harrison et al (1973), using DAPA, reported that bacterial N contributed 37% (sainfoin) and 79% (lucerne) to total duodenal N. suggesting that undegraded dietary protein made a major contribution to the overall increased protein flow on sainfoin. Irrespective of its origins, the apparent avail ability of amino acids in the small intestine appeared to be unaffected by the presence or absence of tannins.
In a subsequent study, Beever and Siddons (1985) examined the effect of sainfoin tannins on 'protein protection' in the rumen, and the possible effect of a small proportion of sainfoin on the digestion of a tannin-free legume such as red clover.
All diets had similar total N contents, but soluble N contents were noticeably lower on the sainfoin-rich diets (Table 1). Duodenal amino acid flows were less than amino acid intake (as expected on fresh forages), but were 11-28% higher on the sainfoin diet than on all other diets (Table 2). Similarly, the flow of microbial protein was highest on the sainfoin diet and it was concluded that much of the increased duodenal (1985). amino acid was due to increased microbial synthesis. This contrasts with the findings of Harrison et al (1973), but in their study the diets were artificially dehydrated prior to feeding. Of possibly greater consequence, is that there was no positive interaction between sainfoin and red clover, from which it may be concluded that the polyphenolics of saifoin are specific (both physically and chemically) to sainfoin protein or that they were not present in sufficient quantity to exert any real effect on the red clover proteins.
Table 1. Nitrogenous fractions of diets containing varying proportions of sainfoin and red clover.
|
Percentage contribution (DM basis) | ||||
|
Sainfoin |
100 |
40 |
20 |
0 |
|
Red clover |
0 |
60 |
80 |
100 |
|
Dietary characteristics (g kg-1 DM) | ||||
|
Total N |
34 |
37 |
38 |
38 |
|
Rumen liquor TCA soluble N |
12 |
18 |
21 |
22 |
|
Buffer soluble N |
8 |
11 |
14 |
15 |
Source: Beever and Siddons (1985).
Table 2. Duodenal digesta amino acids flows and efficiencies of microbial synthesis in sheep receiving diets containing varying proportions of sainfoin and red clover.
|
Percentage contribution (DM basis) | ||||
|
Sainfoin |
100 |
40 |
20 |
0 |
|
Red clover |
0 |
60 |
80 |
100 |
|
Duodenal amino acid flow (g g-1 intake) | ||||
|
Total |
0.83 |
0.65 |
0.75 |
0.67 |
|
Microbial |
0.41 |
0.33 |
0.35 |
0.32 |
|
Feed |
0.32 |
0.25 |
0.32 |
0.28 |
|
Microbial synthesis (g g-1 degraded amino acid) |
0.61 |
0.44 |
0.51 |
0.44 |
Source: Beever and Siddons
The effects of condensed tannins on carbohydrate digestion have been attributed, at least in part, to complexing of microbial extracellular enzymes. Condensed tannins inhibit in vitro proteolytic, cellulolytic and general fermentative activities of rumen microbes (Schaffert et al, 1974; Tagari et al, 1965) and in vivo microbial multiplication (Sadanandan and Arora, 1979).
No information appears to be available on the effects of condensed tannins on protozoal or fungal fermentative capacities, both of which may contribute considerably to digestion of structural carbohydrates. However, the reduction in rumen digestibility in animals receiving diets high in condensed tannins may simply reflect insufficient rumen-degradable nitrogen (RDN) for maximum microbial activity. This is supported to some extent by the responses of rumen digestion to additional non-protein nitrogen. Adding 3% urea to a diet of wheat straw and deoiled sal seed meal fed to calves increased total nutrient digestibility from 48 to 61% (Sinha and Nath, 1982). Similar effects were observed in vitro where adding urea to high-and low-condensed-tannin sorghum grains increased in vitro dry matter digestibility (IVDMD) from 73 to 93% in low-condensed-tannin cultivars and from 46 to 79% in high-condensed-tannin cultivars (Schaffert et al, 1974).
It is not known whether urea destabilises the bonding between protein and condensed tannins, releasing protein for microbial use, or if the urea simply acts as a source of RDN. The latter would seem to be more probable: Schaffert et al (1974) observed no effect of urea on in vitro protein degradabilities of sorghum grains despite increased IVDMD. In vivo studies also tend to support the idea that lack of RDN reduces digestion. Egan and Ulyatt (1980) associated the higher N retention in sheep receiving sainfoin than in those receiving clover or ryegrass diets with an increased rate of recycling of nitrogen (presumably as urea) into the rumen. Barry et al (1986b) reported rumen ammonia levels of about 16 mM in sheep receiving Lotus diets containing 95 g condensed tannins per kg DM and Reed and Soller (1987) found that sheep receiving acacia browse, which is high in condensed tannins, excreted significantly less nitrogen in the urine than sheep receiving Sesbania sesban or vetch hay, which are low in condensed tannins. The latter authors suggested that changes in urinary nitrogen excretion could be the result of increased microbial utilisation of endogenous nitrogen in the rumen.
Digestibility of structural carbohydrates in the rumen is greater with protein supplementation than with NPN (urea) supplementation and responses to different protein sources differ (McAllan and Smith, 1983). However, supplementing diets containing large amounts of condensed tannins with protein may not be economical and other ways to reduce the adverse effects of phenolics must be explored.
Other studies on nitrogen utilisation of feeds high in phenolic contents have shown both positive and negative effects (Reed and Soller, 1987), indicating the need for caution in extrapolating reported effects of phenolics on digestion or utilisation parameters from one plant to another. Reed et al (1985) examined the effects of phenolic compounds from a variety of plants in an in vitro cellulase system a and found that the degree of inhibition varied from about 2 to 70%: this variation was probably related to the types and amounts of phenolic compounds present. Other workers have also reported markedly different responses in rumen microbial activity to phenolics from different sources (Tagari et al, 1965).
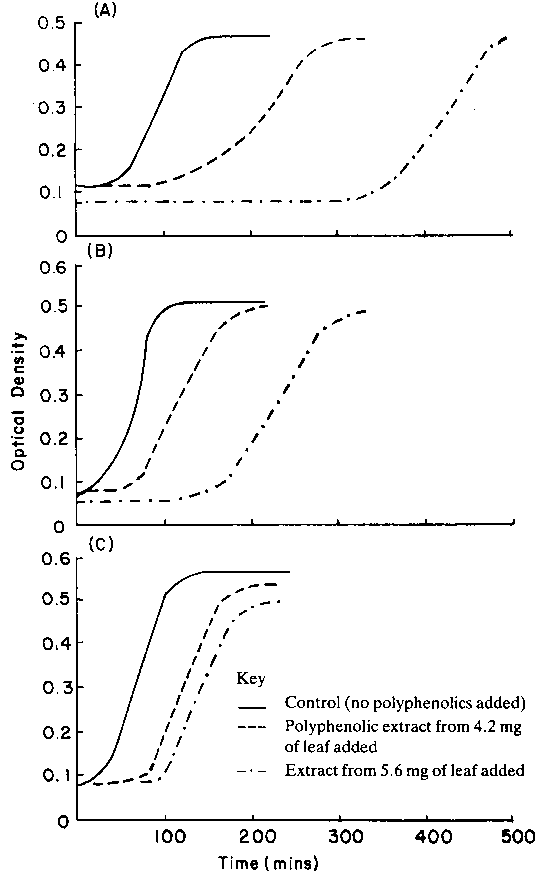
Recent experiments (Mueller-Harvey, Theodorou, Hitchin, Dhanoa, unpublished data) assessed the anti-microbial and physiological effects of several types of polyphenolic compound from Ethiopian browse plants on Streptococcus bovis. In general, all extracts increased lag times and reduced biomass yields. Growth rates were also reduced, but only in cultures grown with insufficient organic nitrogen. Large differences were observed in the extent of these anti-microbial effects when comparable concentrations of phenolics from Acacia nilotica, Euclea schimperi and Pterolobium stellatum were added to S. bovis cultures. Extracts from A. nilotica were substantially more toxic, confirming that polyphenolics are not a uniform group of chemicals having similar effects.
The effect of polyphenolics on S. bovis was reduced by increasing the organic nitrogen concentration in the culture medium (Figure 2). Other workers have also reported markedly different responses in rumen microbial activity to polyphenolics from different plant sources (Tagari et al, 1965).
The toxicity of free phenolic acids has been demonstrated in a wide range of rumen bacteria (Chesson et al, 1982). Bacteriocidal and bacteriostatic effects have been demonstrated in cellulolytic bacteria, such as Ruminococcus albus, R. flavifaciens and Bacteroides succinogenes, in the presence of 10 mM or less of p-coumaric acid and ferulic acid (Chesson et al, 1982). Akin (1982) also observed that cellulolytic and xylanolytic bacteria were inhibited by p-coumaric acid and ferulic acid and that p-coumaric acid reduced motility in entodinomorph, but not holotrich, protozoa. Ferulic acid and sinapic acid had lesser effects on protozoa (Akin, 1982).
Figure 2. Effect of polyphenolics from Acacia nilotica on the growth of Streptococcus bovis at different concentrations of organic nitrogen. Cultures were grown on glucose in medium B (Lowe et al, 1985). A: no organic nitrogen (yeast extract and trypticase omitted). B: half the amount of yeast extract and trypticase C: full amount of yeast extract and trypticase.
Vanillin and, to a lesser extent, cinnamic acid also inhibit cellulolysis by rumen bacteria, apparently by preventing them from attaching to cellulose particles. Similar observations have been reported by Akin et al (in press). Differences in responses to phenolic acids may reflect the dominant bacterial species in the rumen, because individual species of rumen cellulolytic bacteria are affected differently by a given phenolic acid (Chesson et al, 1982). Akin et al (in press) reported changes in sub-group populations, from ruminococcus-like to bacteroides-like morphotypes, in the presence of phenolic acids. Some adaptation by bacteria can occur (Akin et al, in press) and changes in the proportions of VFAs produced over extended incubation periods indicate possible changes in the microbial population.
The effect of phenolic acids on fibre digestion by mixed populations of rumen microorganisms is less clear. Jung and Fahey (1983) reported that a range of phenolic acids, including p-coumaric acid, ferulic acid, protochatecuic acid and vanillin, have a negative effect on the extent of in vitro cellulose digestion but the concentrations required to cause inhibition were in excess of those normally encountered in the rumen. Degradation of forage cell walls by rumen microorganisms alters the amounts of phenolic acids recoverable from plant materials (Theander et al, 1981). Several reports have presented evidence for the breakdown or modification of free monomeric phenolic acids or more complex phenolics under strictly anaerobic conditions (e.g. Chen et al, 1985). In general, microbial consortia are required to modify these then although some pure cultures of rumen bacteria degrade phenolics, with a greater apparent degradation of ferulic than coumaric acid (Theander et al, 1981). Low recoveries of phenolic acids added to in vitro incubations have also been observed by a number of workers (Chesson et al, 1983; Jung and Fahey, 1983; Jung et al, 1983a).
Since cellulolytic bacteria are closely associated with structural polysaccharides in the rumen (Akin, 1976) they may encounter locally high concentrations of potentially toxic bound and free phenolic acids as degradation proceeds. No obvious relationship was found between depressions of cellulose and hemicellulose digestibilities and changes in substitutions to the aromatic ring of the phenolics although aldehydes were found to be more inhibitory than related acids (Jung, 1985). Hemicellulolytic bacteria appear to be more tolerant of phenolic acids than cellulolytic bacteria (Jung, 1985), presumably because these phenolics are linked to the hemicellulose fraction of the cell walls and hemicellulolytic bacteria are adapted to these phenolics.
Experiments with some forages have shown a negative correlation between the phenolic constituents of cell walls and apparent digestibility (Hartley, 1972); removal of phenolic acids from cell walls increases digestibility (Chesson, 1981; Hartley and Jones, 1978; Jung and Fahey, 1981). Ford and Elliott (1987), however, found no relationship between the concentration of any cell-wall constituent and degradability. These latter authors suggested that variability in biodegradability of cell walls is more probably a result of structural features such as cross linking between polymers than concentration of any particular cell-wall constituent.
Barry et al (1986a) examined the effects of tannins on nutrient utilisation in sheep fed high-condensed-tannin Lotus. They observed increased levels of growth hormone and their results suggested an increase in the ratio of lipolysis to lipogenesis. Similar trends were reported by Purchas and Keogh (1984), who found lower carcass fat levels in lambs grazing Lotus than in those grazing white clover. This could be due to dilution of fat content by increased N retention, while increased lipolysis may have been mediated by increased secretion of growth hormone.
Plant phenolics are a diverse group of chemicals. Each group can have different effects on the nutritive value of plants for feeding ruminants. polyphenolics are very reactive and precautions are needed to avoid reactions during handling that affect quantitative and qualitative analysis. The choice of methods for phenolic analysis is difficult and requires consideration of the specificity of each method.
There is sufficient information to question the hypothesis that plants produce polyphenolics in order to defend themselves against insect and herbivore attack (the "chemical defense hypothesis"). An alternative hypothesis is that plants produce phenolics in response to stress conditions such as low soil fertility, drought, high tempera sure, high light intensity and grazing pressure. This hypothesis needs testing by plant physiologists.
A proper understanding of the metabolism of phenolics is needed to assist plant breeding programmes. The variability in enzymatic pathways that lead to the production of phenolic precursors and end-products needs to be investigated. The differences between phenolic compounds which have positive effects on ruminant nutrition and those which have negative effects need to be determined. The difference may depend on the chemical structure of the phenolic and the specificity of the interaction of the phenolics with proteins and carbohydrates.
Akin D E. 1976. Ultrastructure of rumen bacterial attachment to forage eel 1 walls. Applied and Environmental Microbiology 31:562-568. Akin D E. 1982. Forage cell wall degradation and p-coumaric, ferulic and sinapic acids. Agronomy Journal 74:424- 428.
Akin D E, Rigsby L L, Theodorou M K and Hartley R D. (in press). Population changes of fibrolytic rumen bacteria in the presence of phenolic acids and plant extracts. Animal Feed Science and Technology.
ALCA (American Leather Chemists Association). 1956. Sub-committee report. Journal of the American Leather Chemists Association 51:353.
Asquith T N. Uhlig J. Mehansho H. Putman L, Carlson D M and Butler L G. 1987. Binding of condensed tannins to salivary proline-rich glycoproteins: The role of carbohydrates. Journal of Agricultural and Food Chemistry 35:331-334.
Barry T N and Duncan S J. 1984. The role of condensed tannins in the nutritional value of Lotus pedunculatus for sheep. 1. Voluntary intake. British Journal of Nutrition 51:485491.
Barry T N and Forss D A. 1983. The condensed tannin content of vegetative Lotus pedunculatus, its regulation by fertiliser application and effect upon protein solubility. Journal of the Science of Food and Agriculture 34:1047-1056.
Barry T N and Manley T R. 1986. Interrelationships between the concentrations of total condensed tannins, free condensed tannins and lignin in Lotus sp. and their possible consequences in ruminant nutrition. Journal of the Science of Food and Agriculture 37:248254.
Barry T N. Allsop T F and Redecop C. 1986a. The role of condensed tannins in the nutritional value of Lotus pedunculatus for sheep. 5. Effects on the endocrine system and on adipose tissue metabolism. British Journal of Nutrition 56:607-614.
Barry T N. Manley T R and Duncan S J. 1986b. The role of condensed tannins in the nutritional value of Lotus pedunculatus for sheep. 4. Sites of carbohydrate and protein digestion as influenced by dietary reactive tannin concentration. British Journal of Nutrition 55:123-137.
Bate-Smith E C. 1977. Astringent tannins of Acer species. Phytochemistry 16:1421-1426.
Beart J E, Lilley T H and Haslam E. 1985a. Plant polyphenols - secondary metabolism and chemical defense: Some observations. Phytochemistry 24:33-38.
Beart J E, Lilley T H and Has lam E. 1985b. Polyphenol interactions. Part 2. Covalent binding of procyanidins to proteins during acid-catalysed decomposition; observations on some polymeric proanthocyanidins. Journal of the Chemistry Society, Perkin Transactions 2: Physical and Organic Chemistry 1439-1443.
Becker P and Martin H S. 1982 . Protein-precipitating capacity of tannins in Shorea (Dipterocarpaceae) seedling leaves. Journal of Chemical Ecology 8:1353-1367.
Beever D E and Siddons R C. 1 985. Digestion and metabolism in the grazing ruminant. In: L P Milligan, W L Grovum and A Dobson (eds), Control of digestion and ' metabolism in ruminants. Prentice-Hall, New Jersey, USA.
Bernays E A. 1981. Plant tannins and insect herbivores: An appraisal. Ecological Entomology 6:353-360.
Bullard R W and Elias D J. 1980. Sorghum polyphenols and bird resistance. In: Polyphenols in cereals and legumes. Symposium proceedings. 36th Annual Meeting of the Institute of Food Technologists, St. Louis, Missouri, 10-13 June 1979. International Development Research Centre, Ottawa, Canada.
Bu'Lock J D. 1980. In: P S Stein (ed.), The biosynthesis of mycotoxins. Academic Press, London and New York. pp. 1-6.
Burns R E. 1963. Methods of tannin analysis for forage crop evaluation. Technical Bulletin N.S. 32. Georgia Agriculture Experiment Station.
Burns R E. 1966. Tannin in Sericea lespedeza. Technical Bulletin N.S. 164. Georgia Agriculture Experiment S Station.
Butler L G, Riedl D J. Lebryk D G and Blytt H J. 1984. Interaction of proteins with sorghum tannin: Mechanism, specificity and significance. Journal of the American Oil Chemists' Society 61:916-920.
Chen W. Supanwong K, Ohmiya K, Shimizu S and Kawakami H. 1985. Anaerobic degradation of veratrylglycerol-B-guaiacyl ether and guaiacoxyacetic acid by mixed rumen bacteria. Applied and Environmental Microbiology 50:1451-1456.
Chesson A. 1981. Effect of sodium hydroxide on cereal straws in relation to enhanced degradation of structural polysaccharides by rumen microorganisms. Journal of the Science of Food and Agriculture 32:745-758.
Chesson A, Gordon A H and Lomax J A. 1983. Sustituent groups linked by alkali labile bonds to arabinose and xylose residues of legume, grass and cereal straw cell walls and their fate during digestion by rumen microorganisms. Journal of the Science of Food and Agriculture 34:1330-1340.
Chesson A, Stewart C S and Wallace R J. 1982. Influence of plant phenolic acids on growth and cellulolytic activity of rumen bacteria. Applied and Environmental Microbiology 44:597-603.
Coley P D. 1983. Herbivory and defensive characteristics of the tree species in lowland tropical forests. Ecological Monographs 53:209-233.
Davis A B and Hoseney R C. 1979. Grain sorghum condensed tannins. I. Isolation, estimation, and selective adsorption by starch. Cereal Chemistry 56:310-314.
Egan A R and Ulyatt M J. 1980. Quantitative digestion of fresh herbage by sheep. VI. Utilisation of nitrogen in five herbages. Journal of Agricultural Science (Cambridge) 94:47-56.
Feeny P. 1976. Plant apparency and chemical defense. In: J W Wallace and R L Mansell (eds), Biochemical inter actions between plants and insects. Recent Advances in Phytochemistry Volume 10 . Plenum, New York.
Folin O and Ciocalteau V. 192 7. On tyrosine and tryptophane determination in proteins. Journal of Biological Chemistry 73:627-650.
Ford C W. 1978. In vitro digestibility and chemical composition of three tropical pasture legumes, Desmodium intortum cv. Greenleaf, D. tortuosum and Macroptilium atropurpureum cv. Siratro. Australian Journal of Agricultural Research 29:963-974.
Ford C W and Elliott R. 1987. Biodegradability of mature grass cell walls in relation to chemical composition and rumen microbial activity. Journal of Agricultural Science (Cambridge) 108:201-209.
Gartlan J S. McKay D D, Water man P G. Mbi C N and Struhsackers T T. 1980. A comparative study of the phytochemistry of two African rain forests. Biochemical Systematics and Ecology 8:401-422.
Goldstein J L and Swain T. 1965. The inhibition of enzymes by tannins. Phytochemistry 4:185192.
Hagerman A E and Butler L G. 1978. Protein precipitation method for the quantitative determination of tannins . Journal of Agricultural and Food Chemistry 26:809-812.
Harrison D G. Beever D E, Thomson D J and Osbourn D F. 1973. The influence of diet upon the quantity and types of amino acids entering and leaving the small intestine of sheep. Journal of Agricultural Science (Cambridge) 81:391-401.
Hartley R D. 1972. p-Coumaric and ferulic acid components of cell walls of ryegrass and their relationships with lignin and digestibility. Journal of the Science of Food and Agriculture 23:1347-1354.
Hartley R D and Buchan H. 1979. High-performance liquid chromatography of phenolic acids and aldehydes derived from plants or from the decomposition of organic matter in soil. Journal of Chromatography 180:139-143.
Hartley R D and Jones E C. 1978. phenolic components and degradability of the cell walls of the brown midrib mutant, bmz, of Zea mays. Journal of the Science of Food and Agriculture 29:777-789.
Hartley R D and Keene A S. 1984. Aromatic aldehyde constituents of graminacious cell walls. Phytochemistry 23:1305-1307.
Haslam E. 1966. Chemistry of vegetable tannins. Academic Press, London and New York.
Haslam E. 1974. Polyphenol-protein interactions. Biochemical Journal 139:285-288.
Haslam E. 1981. Vegetable tannins. In E E Conn (ed.), The biochemistry of plants. Vol. 7. Academic Press, London and New York. pp. 527-556.
Jambunathan R. Butler L G. Bandyopadhyay R and Mughogho L K. 1986. Polyphenol concentrations in grain, leaf, and callus tissues of mould-susceptible and mould-resistant sorghum cultivars. Journal of Agricultural and Food Chemistry 34:425-429.
Jones W T and Mangan J L. 1977. Complexes of the condensed tannins of sainfoin (Onobrychis viciifolia Scop.) with Fraction 1 leaf protein and with submaxillary mucoprotein, and their reversal by polyethylene glycol and pH. Journal of the Science of Food and Agriculture 28:126-136.
Jung H-J G. 1985. Inhibition of structural carbohydrate fermentation by forage phenolics Journal of the Science of Food and Agriculture 36:74-80.
Jung H G and Fahey Jr G C. 1981. Effect of phenolic compound removal in vitro forage digestibility. Journal of Agricultural and Food Chemistry 29:817-820.
Jung H G and Fahey Jr G C. 1983. Interactions among phenolic monomers and in vitro fermentation. Journal of Dairy Science 66:1255-1263.
Jung H G. Fahey Jr G C and Garst J E. 1983. Simple phenolic monomers of forages and effects of in vitro fermentation on cell wall phenolics Journal of Animal Science 57:1294-1305.
Kahnt G. 1967. Trans-cis-equilibrium of hydroxy cinnamic acids during irradiation of aqueous solutions at different pH. Phytochemistry 6:755-758.
King H C G and Pruden G. 1970 Lower limits of molecular weights of compounds excluded from Sephadex G-25 eluted with aqueous acetone mixtures. Application of the results to the separation of the components of tannic acid. Journal of Chromatography 52:285-290.
Laurent S. 1975. Etude comparative de différentes méthodes d'extraction et de dosage des tanins chez quelques pteridophytes. Archives Internationales de Physiologie et de Biochimie 83:735-752.
Lowe S E, Theodorou M K, Trinci A P and Hespell R B. 1985. Growth of anaerobic rumen fungi on defined and semi-defined media lacking rumen fluid. Journal of General Microbiology 131:2225-2229.
Martin J S and Martin M M. 19 33. Tannin assays in ecological studies. Precipitation of ribulose-1,5-biphosphate carboxylase/oxygenase by tannic acid, quebracho, and oak foliage extracts. Journal of Chemical Ecology 9: 285-294.
Martin M M, Rockholm D C and Martin J S. 1985. Effects of surfactants, pH, and certain cations on precipitation of proteins by tannins . Journal of Chemical Ecology 11:485494.
McAllan A B and Smith R H. 1983. Factors influencing the digestion of dietary carbohydrates between the mouth and abomasum in steers. British Journal of Nutrition 50:445-454.
McLeod M N. 1974. Plant tannins - their role in forage quality. Nutrition Abstracts and Reviews 44:803-815.
McManus J P. Davis K G. Lilley T H and Haslam E. 1981. The association of proteins with polyphenols. Journal of the Chemical Society, Chemical Communications 309-311.
McManus J P. Davis K G. Beart J E, Gaffney S H. Lilley T H and Haslam E. 1985. Polyphenol interactions. Part 1. Introduction: Some observations on the reversible complexation of polyphenols with proteins and polysaccharides. Journal of the Chemical Society, Perkin Transactions 2: Physical Organic Chemistry 1429-1438.
Mole S and Waterman P G. 1985. Stimulatory effects of tannins and cholic acid on tryptic hydrolysis of proteins: Ecological implications. Journal of Chemical Ecology 11:13231332.
Mueller-Harvey I, Hartley R D, Harris P J and Curzon E H. 1986. Linkage of p-coumaroyl and feruloyl groups to cell wall polysaccharides of barley straw. Carbohydrate Research 148:71-85.
Mueller-Harvey I, Reed J D and Hartley R D. 1987. Characterisation of phenolic compounds, including flavonoids and tannins, of ten
Ethiopian browse species by high performance liquid chromatography. Journal of the Science of Food and Agriculture 39:1-14.
Nonaka G. Morimotos and Nishioka I. 1983. Tannins and related compounds. Part 13. Isolation and structures of trimeric, tetramerie, and pentameric proanthocyanidins from cinnamon. Journal of the Chemical, Society, Perkin Transactions 1: Organic and Bio-Organic Chemistry 2139-2145.
Oh H I, Hoff J E, Armstrong G S and Hoff L A. 1980. Hydrophobic inter action in tannin-protein complexes. Journal of Agricultural and Food Chemistry 28:39 4-398.
Price M L, Van Scoyoc S and Butler L G. 1978. A critical examination of the vanillin reaction as an assay for tannin in sorghum grain. Journal of Agricultural and Food Chemistry 26:1214-1218.
Price M L and Butler L G. 197 7. Rapid visual estimation and spectrophotometric determination of tannin content of sorghum grain. Journal of Agricultural and Food Chemistry 25:1268-1273.
Purchas R and Keogh R. 1984. Fatness of lambs grazed on 'Grasslands Maku' lotus and 'Grasslands Huia' white clover. Proceedings of the New Zealand Society of Animal Production 44:219-221.
Ramachandra G. Virupaksha T K and Shadaksharaswamy M. 1977. Relationship between tannin levels and in vitro protein digestibility in finger millet (Eleusine coracana Gaertn.). Journal of Agricultural and Food Chemistry 25:11011104.
Reddy N R. Pierson M D, Sathe S K and Salunkhe D K. 1985. Dry bean tannins: A review of nutritional implications . Journal of the American Oil Chemists' Society 62:541-549.
Reed J and Soller H. 1987. phenolics and nitrogen utilization in sheep fed browse. In: M Rose (ed.), Herbivore nutrition research. Second International Symposium on the Nutrition of Herbivores, Queensland, July 1987. An occasional publication. Australian Society of Animal Production. pp. 47-48.
Reed J D, Horvath P J. Allen M S and Van Soest P J. 1985. Gravimetric determination of soluble phenolics including tannins from leaves by precipitation with trivalent ytterbium. Journal of the Science of Food and Agriculture 36:255-261.
Reid C S W, Ulyatt M J and Wilson M J. 1974. Plant tannins, bloat and nutritive value. Proceedings of the New Zealand Society of Animal Production 34:82-93.
Sadanandan K P and Arora S P. 1979. Influence of tannins on rumen metabolism, Journal of Nuclear Agriculture and Biology 8:1-8.
Sarkar S K and Howarth R E. 1976. Specificity of the vanillin test for flavonols. Journal of Agricultural and Food Chemistry 24:317-320.
Schaffert R E, Lechtenberg V L, Oswalt D L, Axtell J D, Pickett R C and Rhykerd C L. 1974. Effect of tannin on in vitro dry matter and protein disappearance in sorghum grain. Crop Science 14:640-643.
Singleton V L and Rossi J A Jr. 1965. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. American Journal of Enology and Viticulture 16:144158.
Sinha R P and Nath K. 1982. Effect of urea supplementation on nutritive value of deoiled sal meal in cattle. Indian Journal of Animal Sciences 52:1165-1169.
Swain T and Bate-Smith E C. 1962. Flavonoid compounds. In: M A Florkin and H S Mason (eds), Comprehensive Biochemistry 3:755-809. Academic Press, New York.
Swain T and Hillis W E. 1959. The phenolic constituents of Prunus domesticus. 1. The qualitative analysis of phenolic constituents. Journal of the Science of Food and Agriculture 10:63-68.
Tagari H. Henis Y. Tamir M and Volcani R. 1965. Effect of carob pod extract on cellulolysis, proteolysis, deamination and protein biosynthesis in an artificial rumen. Applied Microbiology 13:437-443.
Tempel A S. 1982. Tannin-measuring techniques. Journal of Chemical Ecology 8:1289-1297.
Theander D, Uden P and Aman P. 1981. Acetyl and phenolic acid substituents in timothy of different maturity and after digestion with rumen microorganisms or a commercial cellulase. Agriculture and Environment 6:127-133.
Thomson D J. Beever D E, Harrison D G. Hill I W and Osbourn D F. 1971. The digestion of dried lucerne and sainfoin by sheep. Proceedings of the Nutrition Society 3:14A.
Van Buren J P and Robinson W B. 1969. Formation of complexes between protein and tannic acid. Journal of Agricultural . and Food Chemistry 17:772-777.
Vande Casteele K, Geiger H and Van Sumere C F. 1982. Separation of flavonoids by reversed-phase high-performance liquid chromatography. Journal of Chromatography 240:81-94.
Verzele M, Delahaye P and Van Damme F. 1986. Determination of the tanning capacity of tannic acids by high-performance liquid chromatography. Journal of Chromatography 362:362-374.
Walter W M and Purcell A E. 1979. Evaluation of several methods for analysis of sweet potato phenolics. Journal of Agricultural and Food Chemistry 27:942-946.
Watterson J J and Butler L G. 1983. Occurrence of an unusual leucoanthocyanidin and absence of proanthocyanidins in sorghum leaves. Journal of Agricultural and Food Chemistry 31:41-45.
Jenkins: Could you clarify whether phenolics are produced under stress or do they provide protection under stress conditions?
Mueller - Harvey: Under stress conditions normal metabolism is affected and light energy which would be converted to carbohydrate has to be absorbed in another way. The plant responds by producing phenolics.
Van Soest: Lignin production decreases under stress conditions but tannin levels increase. In alfalfa isoflavones are produced.
Reed: If stovers are sun-dried the phenolics may polymerize and thus have a lower inhibitory effect on bacteria. How ever, in the process of polymerisation some nutrients may be complexed and they become less available.
Aboud: If phenolics are produced in response to stress conditions, why do plants such as tea contain substantial amounts of tannins?
Mueller - Harvey: Some plants routinely produce phenolic compounds.