1. IDENTITY
2. DISTRIBUTION
3. POPULATION STRUCTURE
4. GROWTH
5. HARVESTING, BIOMASS RECOVERY AND ECONOMICS
6. AQUACULTURE
7. PROTECTION AND MANAGEMENT OF THE RESOURCE
8. UTILIZATION
9. REFERENCES
APPENDIX I EXCERPTS FROM: MARINE PLANTS/ATLANTIC FISHERY REGULATIONS
by
Glyn Sharp
Invertebrate and Marine Plants Division
Fisheries Research Branch
Fisheries and Oceans Canada
Halifax, Nova Scotia, Canada
|
ABSTRACT Ascophyllum nodosum, a major North Atlantic seaweed resource, is distributed widely from the Arctic Circle to 40 degrees North Latitude. In eastern Canada 45,000 T of wet Ascophyllum are considered harvestable. The present annual harvest ranges is 5/000 to 9,000 wet tons. Hand harvesting has been largely replaced by mechanical harvesting in the last 15 years. Recovery of biomass following harvest is dependent on site productivity/distribution of standing crop, and degree of harvest. After mechanical harvesting residual biomass ranges from 80 percent to 40 percent of pre-harvest standing crop. The economics of harvesting methods were compared, and cost per harvested ton ranged from Canadian dollars 10.65 to Canadian dollars 38.71 per wet ton. Ascophyllum from eastern Canada has been used for a range of agricultural, industrial, and human food products. However, alginate accounts for 95% of the end product. |
1.1 Nomenclature
1.2 Taxonomy
1.3 Morphology, anatomy & reproductive structures
1.4 Life cycle and sex composition
Ascophyllum nodosum was originally placed in the genus Fucus as Fucus nodosus L. It has numerous synonyms and has been treated various genera as follow:
Ascophylla laevigata Stackh
Ozothallia nodosa Decaisne and Thuret
Physocaulon nodosum Kutz
Halidrys nodosa Lyngb
Fucodium nodosum J. Ag.
Helicoccus nodosus Aresch.
The present accepted name was proposed by Le Jolis in 1863.
Ascophyllum nodosum is a member of the order Fucales. These are defined as growing from an apical cell group. Superficial conceptables carry the sporangia where gametes are released and combine outside of the plant. The family Fucaceae have dichotomous or pinnate branches, in some cases with numerous marginal slits or pits (cryptostomata Taylor, 1957). The genus Ascophyllum is distinguished from Fucus by branches un-differentiated into a midrib and a blade. Ascophyllum nodosum has a linear cartilaginous thallus with dichotomous primary branching and pinnate lateral or secondary branching (Fig. 1). Single vesicles expand the thallus at varying intervals. Receptacles are oval and yellow when mature.
Ascophyllum is an intertidal algae conspicuous due to its ability to float with the changing water surface. Vesicles (air bladders) support the thallus allowing the growing apices to be exposed maximumally to light energy (Fig. 1).
The plants are held to the substratum by discoid holdfasts which in dense populations frequently coalesce. The morphological terminology used to describe Ascophyllum has varied with authors. Cousens, (1981a) re-examined these terms and his definitions are adopted for this report as follows with additions to account for harvest related morphology (Fig. 1).
Shoot - the axis which results from growth of an apical meristem.
Primary shoot - originating from the holdfast (includes basal shoot of Baardseth).
Lateral shoot - arising from a lateral pit on another shoot. cryptostomata of Taylor/1957).
Figure 1. Ascophyllum nodosum fronds and associated terminology: A, apical tip; B, basal shoot; H, holdfast; I, internode; L/lateral shoot; P, primary shoot; R, receptacle; S, stump; V, vesicle.
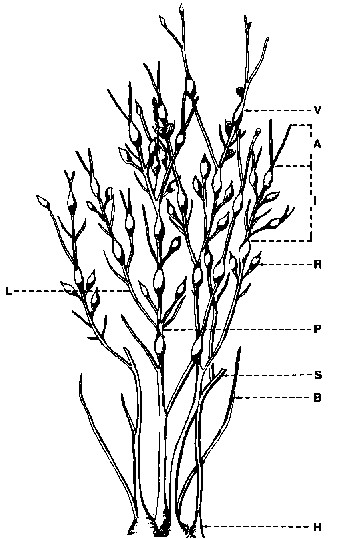
Stump - a shoot lacking a apical meristem.
Intact shoot - a shoot with at least one apical meristem.
Internode - the portion of a shoot between two adjacent vesicles.
Apical tip - the portion of a shoot distal to the last vesicle. Frond - the system of shoots derived from a single meristematic protrusion of the holdfast.
Plant - an assemblage of fronds arising from a common holdfast.
Stand - a group of plants within a defined area.
Vesicles - dilations of the shoot produced at intervals related to rate of annual shoot elongation.
Receptacles - fertile lateral shoots first appearing April -June maturing in one year and being shed April to June.
Shoot morphology varies greatly between sites and some of this variation can be attributed to environmental gradients. Vesicles are enlarged at sites exposed to wide variations in salinity.
Receptacles can vary in shape and size in the same locality (Baardseth/1970). Receptacles at the most extreme wave exposures were more spherical and had slender long stalks. The most apical internode of unbroken primary shoots reach maximum length at sites of intermediate exposure (Cousens, 1982). Overall length of shoots in relation to water movement increases with velocity up to 1 m/sec but declines thereafter (Mathieson et al 1977). Pigment concentrations increase with wave exposure changing from yellow in sheltered areas to green on the open sea coast (Cousens 1982). A fuller discussion of plant size is contained in Section 3.
Ascophyllum reproduces sexually through the union of eggs and sperms produced in the conceptacular cavities of ripe receptacles. The ratio of male to female plants was reported in early literature as approximating 50/50 (Baarsdeth/1970). Printz, (1956), determined shoot sex by coloration of receptacles; male yellow, female/greenish yellow and described female conceptacles as having larger openings (ostioles) than males. Although there were numerous investigations of reproductive periodicity/no further work has been reported on geographical or vertical distribution of male and female shoots.
Receptacles are initiated in April to June grow and mature for gamete release (also) in April to June. In Maine peak reproduction was April and March of successive years/however/the level of reproduction increased 3-fold in the second year (Mathieson et al. 1976). Mature receptacles were noted in monthly collections from the Bay of Fundy in March and April (Edelstein et al. 1970).
Receptacle initiation is stimulated by photoperiods of 8:16 to 12:12, longer periods of darkness inhibit initiation (Terry and Moss, 1980). The proportion of stand biomass comprised of receptacles ranges from 12.4 to 35.7% in a gradient from wave exposed to wave sheltered (Cousens, 1981a). Maximum dry weight per receptacle is at intermediate wave exposures (Cousens, 1982). However, reproductive effort is greatest at exposed sites, 61.2-60.8% (Cousens, 1981a).
The biomass of the receptacles is equivalent to that of the vegetative tissues during peak reproduction (Josselyn and Mathieson, 1978). The decomposition of the A. nodosum receptacles is rapid; 50% of their original ash free weight being lost within 50 days. Thus the release of A. nodosum receptacle tissue during the spring reproductive period makes a significant contribution to the estuarine detrital pools.
a) Eastern Canada
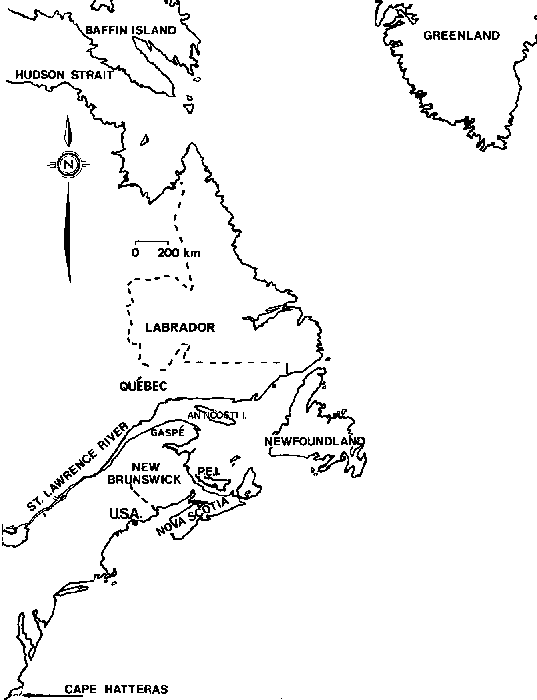
Until 1933 records of Ascophyllum nodosum in Eastern Canada were limited to individual widely dispersed single collections in localized areas. Collections from a series of 93 sites scattered over the Bay of Fundy, Atlantic and Gulf shores of Nova Scotia, and the coast of Prince Edward Island listed Ascophyllum as abundant in the Bay of Fundy and Atlantic shores and occasionally around Prince Edward Island (Fig. 2) (Bell and MacFarlane 1933). Although there have been more recent collections from the Atlantic and Fundy coasts of Canada, (Edelstein and McLachlan, 1966; Neish, 1971) no new extensions of the species range have been reported.
De la Pylaie visited Newfoundland, St. Pierre et Miquelon in 1816 and 1819 and recorded the species of Fucales (Fig. 3). Until 1970 records from this area were very limited (Hariot, 1889), (Wilce, 1959) (Lee, 1968). An intensive distributional study of Ascophyllum nodosum and its ecads found the attached form at 50 sites representing all coasts of Newfoundland (South and Hill, 1970).
In the Gulf of St. Lawrence, Bay de Chaleur and Gaspe, Ascophyllum nodosum was common in collections from 46 stations (Cardinal, 1967). Ascophyllum nodosum was reported from 28 of 48 stations along the north and south shores of the St. Lawrence estuary to within 20 km of Quebec City (Cardinal and Villalard, 1971) (Fig. 3).
b) World distribution
Ascophyllum is widely distributed in the North Atlantic from Northern Norway and the White Sea to Portugal in the Eastern Atlantic. The arctic circle in the Western Atlantic marks the northern limits and the 40° N latitude the southern edge of Ascophyllum nodosum's range (Fig 3). Although more northern exploration has occurred since the description of range by Baardseth (1970) no new northern extension was reported (Lee, 1980).
c) Vertical distribution
Ascophyllum nodosum is primarily a mid to low intertidal species. Free living ecads can be found in the upper intertidal and attached non floating forms occur in the subtidal (South and Hooper, 1980). In southwestern Nova Scotia A. nodosum occurs from the mean highwater spring tide line +3.5 m to -6 m (Fig. 4) (Pringle and Semple, 1980). Within estuaries the vertical distribution of attached A. nodosum is restricted to the mid to lower intertidal. (Chock and Mathieson, 1983). The distribution of biomass is examined in Section 4.3.
The upper limits of Ascophyllum nodosum distribution are controlled by its ability to resist desiccation and high temperatures (Schonbeck and Norton 1978). Ascophyllum nodosum dominates the mid intertidal on shores where it is present. Removal of A. nodosum temporarily allows other fucoids to become established for one or two years. (Hawkins and Hartnoll, 1985: Keser et al, 1981). Ice scour in the St. Lawrence river estuary controls the mix or dominance of A. nodosum and Fucus vesiculosus (Archami-bault and Bourget 1983). In areas of frequent ice scour F. vesiculosus dominates and A. nodosum is dominant in areas without or infrequent ice scour. Juvenile Ascophyllum plants are able to persist under canopies in the field. Once established the shoots are long lived, 5-15 years exceeding the life span of competitive fucoids. Ascophyllum is less susceptible to grazing than are other macrophytes. This is due to the low level of preference of Littorinids for Ascophyllum relative to other fucoids (Menge, 1975).
Figure 3. The eastern seaboard of Canada and the United States.
The factors controlling the lower limits of Ascophyllum in Atlantic Canada have been attributed to grazing pressure in sheltered environments. In exposed locations grazers are less abundant and lower limits are marked by 90% cover of Chondrus crispus. Chondrus survives under the A. nodosum canopy but will bleach if exposed to full light. If C. crispus is removed including the holdfast in its area of dominance, Fucus will colonize the bare surface (Lubchenco; 1980). However/if only Chondrus fronds are removed Ascophyllum re-establishes dominance within two years.
The minimum age of Ascophyllum nodosum shoots has been determined by the number of vesicles on the longest unbroken shoot; assuming one vesicle is formed annually. The time for the formation of the first vesicle is indeterminate and the length of the first internode varies greatly within a site (Cousens, 1981a). Maximum ages beyond the first vesicle are reported as 16 years in Maine and 12 years in Wales (Keser et al. 1981; David, 1943). In southwestern Nova Scotia maximum age beyond the first vesicle was 15 years (Sharp, unpublished data). Repeated breakage of primary or lateral shoots precludes a meaningful description of age structure. Instead we must examine length weight distributions of these populations. The mean length of primary shoots ranged from 18.6 + 19.9 cm to 11.9 + 14 cm at 5 sheltered to semi-sheltered sites in southwestern Nova Scotia (Sharp, 1981).
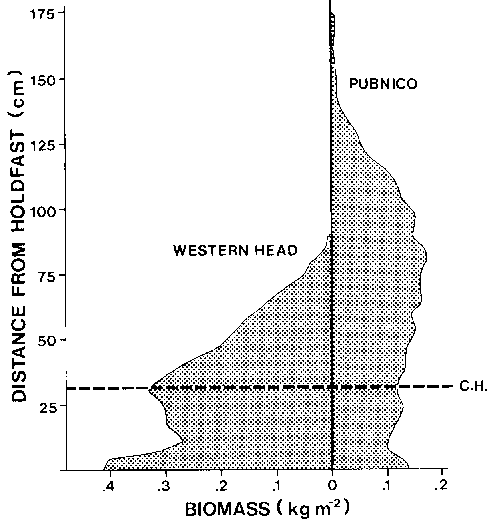
The length frequency of primary shoots is extremely skewed to the 0-10 cm class, (Fig. 5). The weight of unbroken primary shoots is a power function of shoot length (Fig. 6). Shoots above 30 cm in length contribute the majority, of stand biomass. Wave exposure affects the level of biomass within a stand. At exposed sites biomass is concentrated in shoots below 50 cm in length (Cousens 1981a) (Fig. 7) compared to sheltered sites where the majority of biomass is above 50 cm from the holdfast (Fig. 7). The proportion of shoot density contributing to 50% of the biomass at sheltered sites is 2.5% versus 10.5% in exposed sites (opp. cit).
Figure 5. The distribution of lengths for intact primary (normal) shoots from a wave sheltered site in southwestern Nova Scotia.
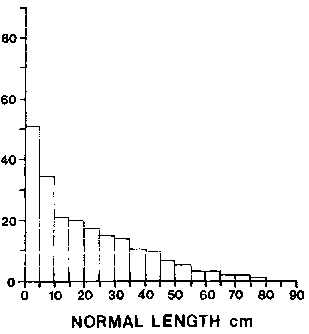
Figure 6. The length to wet weight relationship of intact primary shoots from a sheltered site in southwestern Nova Scotia.

Figure 7. The distribution of Ascophyllum biomass within stand canopies at a wave exposed site (Western Head) and a sheltered site (Pubnico) southwestern Nova Scotia, (Cousens, 1981a). CH, mean cutting height of mechanical harvesters.
The magnitude of natural mortality for Ascophyllum varies annually as significantly as between physical factors of tidal level and wave exposure (Breton Provencher, 1976). Mortality of tagged shoots over the summer was 9% and 16% in successive years in the St. Lawrence River estuary (opp. cit.). Survivorship of shoots (shoot type not identified) in a Maine estuarine site was 75% after one year (Vadas et al. 1976).
If survivorship of each shoot type is examined separately patterns related to environmental gradients are apparent. Survivorship of primary shoots to when an additional vesicle was present increased with increasing wave shelter (Cousens, 1981a). At exposed sites maximum survival was 5 years beyond the first vesicle for primary shoots; in wave sheltered areas the maximum survival was 11 years (opp. cit.). The position of the lateral shoot on a primary relative to the holdfast affects survivorship. Independent of age class laterals in the 0-30 cm (basal) portion of a shoot, had a survivorship of 90% after one year. Mortality of laterals decreased with distance from the holdfast/although mortality increased with age of lateral. The loss of laterals was primarily due to loss of the primary shoot rather than individual lateral shoot loss (opp. cit).
The germling is the most vulnerable stage in Ascophyllum's life. The initial settlement of germlings in an area denuded of Ascophyllum in Maine was 50/000 shoots m-2. Fifty percent mortality occurred within one year of settlement. Shoot densities reached a minimum >8/000 m-2 in 2 years covering 60% of the substratum (Keser and Larson, 1984).
Sheltered sites in the North Atlantic are frequently ice covered. Movement and abrasion of ice can be a significant cause of mortality. In the Great Bay Estuary of New Hampshire ice effects removed 1.04 kg m-2 dry weight of Ascophyllum fragments per year (Mathieson et al. 1982). In sheltered areas the Ascophyllum zone is a habitat for Littorina species, all potential grazers. However, microscopic A. nodosum was low in the food preference rank in a two way choice presentation of 39 red green and brown algae (Lubchenco/1978). If no other foods were available Ascophyllum was consumed.
Elevated water temperatures caused either by natural or man made phenomena can affect growth or survival of Ascophyllum nodosum . Periodic bursts of high (>20°C) temperature water have inhibited growth for 1.5 years in Maine. In other areas growth was initiated earlier than normal in the growing season (Vadas et al. 1976). Electrical impedence measurements of A. nodosum tissues, an indication of ion movement in extracellular channels, show cell death occurs at -20°C and/or 70% water loss (MacDonald et al. 1974). A. nodosum loses 70% of its fresh weight excluding vesicles after 7.5 hours at 22°C and relative humidity of 40-45% (Dorgelo, 1976). This rate was slower than adjacent Fucus vesiculosus. However, in general, the rate of water loss did not correspond to the tidal level of individual species (opp. cit.).
The upper limits of A. nodosum distribution on the shore were shown in field experiments to be its physiological limits resulting from desiccation factors at these levels (Schonbeck and Norton, 1978). Exposure to long periods of drying resulted in visible tissue damage of A. nodosum after 21 to 28 days (opp. cit.). Winter conditions and rainfall were not critical factors. Transplants of A. nodosum above the normal upper levels of distribution suffered mortalities (opp. cit.).
Ascophyllum nodosum occurs in estuarine locations which are frequently urbanized and industrialized. In approaches to pollution problems, A. nodosum has been utilized in two ways. The ecological approach was to assess the effects of effluents on the distribution, productivity, and reproduction of A. nodosum. The use of A. nodosum as a bioindicator for specific water-soluble pollutants has resulted from its bio-accumulation abilities and perennial nature.
A series of ecological base line studies is available for Oslofjord, Norway, over the period of increasing (1943 to present times) urban and industrial influences on water quality in this fjord. Over this period of increasing pollution, particularly sewage, Ascophyllum nodosum became less common in the inner fjord and at present it does not exist there (Rueness, 1973). Transplantation of mature A. nodosum plants from the outer fjord to the inner polluted area found no significant differences in vegetative or reproductive growth. It was assumed the germlings of A. nodosum could not compete with the Enteromorpha carpet present in the polluted areas of the inner basin (Rue ness opp. cit.). Similar ecological studies were conducted in Iceland (Gunnarsson and Porrison, 1976). Ascophyllum nodosum was among the higher diversity of species which occurred solely away from the sewage outfall.
A decrease in macro-algal species occurred towards a source of pulpmill wastes in the Iddefjord, Norway (Lein et al. 1974). Transplantation of Ascophyllum nodosum and F. vesiculosus to the inner fjord adversely affected their growth and development. Sedimentation and toxicity resulting from this pollution controlled inner boundaries of species rather than salinity in the fjord. Comparisons of Ascophyllum tissues from polluted and unpolluted estuarine areas found lower dry matter, mannitol, and higher phosphate and protein contents in polluted than in unpolluted ones (Munda, 1967).
Monitoring tagged plants, periods of maximum growth in length of 3.6 to 3.4 cm month1 were reported for April and July 1973, respectively in the Great Bay Estuary System of New Hampshire-Maine (Mathieson et al. 1976). Distinct yearly variations were found both in time of/maximal growth and rate of growth. Maximum growth rates were 2.3 and 2.2 cm month-1 in May and October respectively of the second measurement year (opp. cit.). An average of 0.8 cm month-1 was recorded for a Maine population by Vadas & Keser (1972). Three successive years of growth (elongation) measurements of Ascophyllum nodosum in the upper Ascophyllum zone at a site in Maine were 17.4+4.7, 11.5+5.5 and 12.2+5.5 cm (Keser and Larsen, 1984). Breton Provencher (1976) observing A. nodosum elongation in the St. Lawrence river estuary reported 26.4 cm and 24.8 cm in successive years. Nor-mandeau Assoc. Inc. (1977) found spring maximum growth rates of 2.2 cm month-1 and winter minimums of 1.2 cm month-1 in Maine. MacFarlane (1932) measured inter-vesicle length of Nova Scotian populations and calculated growth rates of 0.33 to 1.2 cm/month.
Maximum rates of shoot elongation were recorded in the central part of the Ascophyllum zone but differences between zones did not exceed annual variation (Keser and Larsen, 1984). The length of the most distal internode (growth in year from formation of last vesicle) on unbroken primary shoots reaches a maximum at sites of intermediate wave exposure (Cousens, 1982). Growth rate increases with the length of intact primary shoots up to 15 cm at more exposed sites and 70 cm at more sheltered sites (Cousens, 1981a). The extremes of exposure were suggested to be suboptimal for growth due to conditions of desiccation/water turnover, and light (opp. cit.).
A relative growth rate comparison was made between Ascophyllum nodosum and Fucus vesiculosus over seven-day periods at depths of 0 to 4 m. Growth in length and fresh weight decreased with depth except except for an increase near compensation depth (Ramus et al. 1977). Ascophyllum nodosum and F. vesiculosus grew in the same pattern but A. nodosum grew at a higher rate (opp. cit.). Maximum photosynthetic activity occurs in tissues of 2-3 years old in shoots with an average age of five to nine years (Khailov, 1976a). This peak of photosynthetic activity was correlated to the maximum dry weight for the 2-3 year age class of tissues (opp. cit.).
A series of unique growth studies was conducted using short term measurements of apical growth in Ascophyllum nodosum with laser diffraction. Irradiance and temperature were correlated with hourly measurements of the growing tip to 1 m m. At artificial light of 12.5 Wm-2 and below, plants showed a direct relationship between growth and irradiance (Stromgren, 1977). Irradiance of 35 to 40 Wm-2 resulted in a reduced growth rate after 21 day periods. The fucoid species compensation point of 3 Wm-2 is only reached in the most extreme low light conditions during winter overcast days under a canopy (Schonbeck and Norton 1980). The saturation point 30-50 Wm-2 is not reached at the surface under similar conditions (opp. cit.). Response of apices to temperature was rapid between 2.5 to 30°C for the first few hours, however, experiments of two to three weeks duration showed the optimal temperature or growth was below 17°C (Stromgren, 1976). These laboratory studies correspond to field observations of maximum growth rates in moderate (6-10°C) temperatures and light 150-250 1y d-1 (Mathieson et al. 1976). Elongation of Ascophyllum apices is linearly correlated with rapid air temperature increases to 35°C over 1 to 4 hours (Stromgren, 1983). The duration of exposure at low tide and ambient air temperature can account for a large portion of shoot elongation and annual variability in growth.
Growth continues during low nutrient conditions because of direct use of nitrogen from seawater during low ambient nitrogen levels and optimal light (Asare and Harlin, 1983). Tank cultured Ascophyllum nodosum growth is in agreement with previous lab and field observations reaching a maximum of 2.5% d-1 (length) at 15°C. Growth was a linear function of light up to 20-30 yEm-2 5-1 (Fortes and Luning, 1980).
Growth of zygotes settled on stone and brick substrata and transferred to the sea after ten days was found to be only 1 to 0.2 cm after one year and 0.5 to 1.5 cm after the second year (Sundene; 1973). Maximum growth rates for young Ascophyllum nodosum (10-80 mm) were 7 mm, 5 mm and 10 mm for the periods Sept.- Nov., Nov.-Feb. and Feb.-May in Scotland (Schonbeck and Norton, 1980). Studies of thallus growth immediately after germination in culture showed an increase with light intensity to 10,000 lux with temperature to 20°C (Sheader and Moss, 1975). After 30 days, the thallus was 0.27 mm, corresponding to a growth of 0.32 cm year-1 similar to the in-situ values of Sundene, 1973.
In a gradient of wave exposure, annual production (measured by standing crop differences) increases with wave shelter (Cousens, 1981b), Table 1. However the maximum production per unit biomass occurred on semi-exposed locations corresponding with an increase of reproductive effort. The turnover time for vegetative biomass ranged from 2.9 years at semi-exposed sites to 10.98 years at extreme wave exposed sites. Annual production varied by a factor of 2 within a 10 km section of coast line (opp. cit.).
Table 1. Standing crop and production (kg dry wt m-2 at sites of different exposures to wave action. Sites 1-6 Polly Cove, Site 7 Dover Soi. P/B was calculated from the Modified Baardseth production estimates. (Cousens 1981b).
|
Site |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
|
April Standing |
0.67 |
1.50 |
1.39 |
2.04 |
3.25 |
3.95 |
3.52 |
|
Crop (+2 s.e.) |
+0.34 |
+0.07 |
+0.09 |
+0.13 |
+0.25 |
+0.97 |
+0.61 |
|
Modified Baardseth method |
0.15 |
0.35 |
1.10 |
1.25 |
1.37 |
1.66 |
1.72 |
|
Standing Crop |
0.05 |
0.26 |
0.95 |
0.94 |
0.84 |
1.07 |
1.05 |
|
Difference Method P/B |
0.22 |
0.71 |
0.79 |
0.61 |
0.42 |
0.42 |
0.49 |
Differences between the annual maximum and minimum standing crops obtained in seasonal surveys of Ascophyllum nodosum biomass were used to calculate annual production for a number of widely separated sites (Cousens 1984, Table 2). The maximum value of annual production was found in southwestern Nova Scotia equivalent to 1,015 g c m-2 (Cousens, 1981a).
Table 2. Annual production and production to biomass ratios for Ascophyllum nodosum populations (Cousens 1964).
|
Location |
t dry wt ha-1 |
P/B |
|
New England |
15 |
.86 |
|
White Sea |
13 |
.65 |
|
Spain |
23 |
.64 |
|
Nova Scotia (pre-harvest) |
20-26 |
.33-.25 |
|
Nova Scotia |
6.1-28.2* |
.22-.79 |
*Corrected for growth loss prior to sampling Cousens 1964.
Biomass density in southwestern Nova Scotia is among the highest in the world (Table 3). Maine and southwestern Nova Scotia values are similar but Norwegian biomass density is closer to the levels in the St. Lawrence Estuary (Table 3). Survey methodology varies greatly between studies from selected samples in the Ascophyllum zone (McFarlane, 1952; Topinka et al. 1981) to a volume "eyeball" estimate (Pielou, 1981).
Table 3. Biomass density of Ascophyllum nodosum populations in the North Atlantic.
|
Source |
|
Location |
Tonnes/Hectare wet weight |
|
McFarlane |
1952 |
Southwestern Nova Scotia |
198a |
|
Sharp |
1981 |
Southwestern Nova Scotia |
120 ± 110b |
|
Cousens |
1981a |
St. Margarets Bay Nova Scotia |
96 ± 45a |
|
Breton- Provencher |
1976 |
St. Lawrence Estuary Quebec |
20 ± 5a |
|
Topinka et al |
1981 |
Lincoln County Maine |
80ac |
|
Chock & Mathieson |
1976 |
Great Bay Estuary New Hampshire |
72a |
|
Keser et al |
1981 |
Average of six sites Boothbay, Maine |
84 ± 60a |
|
Baardseth |
1970 |
South Norway |
6.9 ± 1.3 |
|
|
Central Norway |
24.1 ± 4.0 |
|
|
|
North Norway |
14.4 ± 1.7 |
|
|
Munda |
1978 |
Iceland |
69 |
aunharvested stock
bharvested stocks
cincludes all Fucoids
Surveys and spot biomass values on the Atlantic coast of Nova Scotia and New Brunswick were combined with existing air photo analysis to estimate standing crops for Ascophyllum nodosum (Smith and Loucks, 1980). Some areas, e.g. the Nova Scotia Eastern Shore, have no ground truthing and other areas had poor quality air photos due to high tides. Taking a conservative estimate from the range of probable values the Atlantic and Fundy coasts of Canada support 442,000 t of A. nodosum wet weight (Table 4). A standing crop of 180,000 tons for southwestern Nova Scotia was estimated prior to harvesting and was based on selective sampling methods (McFarlane, 1952).
Mean fucoid biomass per meter of a shoreline in Maine was 352.17 kg m-1 versus 540.0 kg m-1 in southwestern Nova Scotia (Topinka et al., 1981). The primary difference between these areas was a bed width of 45 m in Nova Scotia versus 22 m in Maine.
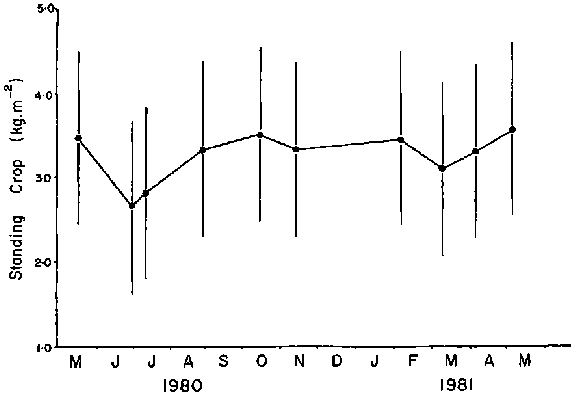
Seasonal variation in Ascophyllum biomass was significant at a New Hampshire site (Chock and Mathieson, 1983). Peak biomass occurred in August 105-135 g· 0.1m-2 and declined to 25-42 g· 0.1m-2 in September. However, at a southern Nova Scotian site the only significant change in standing crop occurred (Fig. 8) in June-July after the loss of receptacles (Cousens, 1981a).
Table 4. Total standing crop of A. nodosum from the Atlantic and Bay of Fundy shores of Nova Scotia and New Brunswick (Smith and Loucks 1980).
|
Marine Plant District Area |
Standing crop wet t |
Fucoids estimated harvestable annual yield wet t |
|
14, Southern New Brunswick |
51,000 |
3,800 |
|
12, Minas Basin to Chebogue |
42.000+ |
4,200 |
|
12, Chebogue to Cape Sable Island |
125.000+ |
15,000 |
|
12, Cape Sable |
100.000+ |
10,000 |
|
11, Medway to Chedabucto Bay |
154,000 |
11,500 |
|
Total Atlantic & Fundy Coasts of Mainland Nova Scotia & New Brunswick |
472,000 |
44,500 |
|
From Smith & Loch 1980 |
|
|
+ - Discounted by the author due to sampling methodology and assumptions of estimate.
Figure 8. Seasonal variation in standing crop (dry weight) at Polly Cove, southeastern Nova Scotia. The same plants were measured each time by a non-destructive method (Pielou, 1981). Vertical line indicates the mean ± 2 s.e. (Cousens, 1981a).
Biomass density decreases at both extremes of its vertical distribution on the shore (Chock and Mathieson, 1983; Cousens 1981a; Pringle and Semple, 1980) (Fig. 4). The spatial distribution of biomass is related to substratal types. In an area of mixed rock and gravel, patches of Ascophyllum averaged 1.5 to 2 m in maximum dimension. Rock and cobble substrata supported larger 2.0-2.5 m patches of A. nodosum (Black1, unpublished data). Biomass density increases with greater quantities of stable substrata (Fig. 9) (Topinka, 1980).
¹R.Black. Univ. Western Australia, Perth, Australia.
5.1 Harvesting
5.2 Regrowth
5.3 Economics of harvesting techniques
a) methods
Commercial harvesting of Ascophyllum nodosum in southwestern Nova Scotia has been active for 20 years by Scotia Marine Products Limited, Lower Woods Harbour (Fig. 2). Harvesting methodology made a transition from hand to mechanical methods in the early 1970's (McArthur, 1971). Maximum annual tonnage was 7,000 wet weight averaging between 5,000 and 6,000 t over a 8-9 month operating period. For the first 15 years of operations cutting was managed in a series of 2.2 km by 2.6 km grids, time and tonnage of yields were logged for each grid (Table 5).
Figure 9- Standing crop of Ascophyllum on gradients of wave exposure and proportion of stable substrate in Maine (Topinka, 1980).

Table 5. Annual harvests from selected 2.2 kg by 2.6 km grid areas by southwestern Nova Scotia. Grid areas were not always completely harvested within one year.
|
Location |
Harvest history by year () = tonnage | |||||
|
Northwest Roberts Is. |
1972a |
1974b |
(745) |
1975c |
(747) |
1978c (818) |
|
Raspberry Is. Woods Harbour |
|
1974c |
|
1977c |
|
1978c |
|
Eastern Wilson Is. |
1972a |
1974a&c |
(307) |
1976c |
(814) |
1977a |
|
Tuna Wharf Wedgeport |
|
1974a&c |
(224) |
1976c |
(414.8) |
1977a |
|
South Mikes Island |
|
1974c |
|
1976c |
(94) |
|
aPossible hand harvest
bPartial harvest
cMechanical harvest
The reharvest interval was 3 years except in areas where hand harvesting was used to reach portions of the resource inaccessible to mechanical harvesting. In the last 5 years the area has been divided in two and reharvested every 2 years. Certain areas exhibiting exceptional growth near the processing plant were harvested annually. (Reharvest intervals are discussed relative to harvester efficiency, Section 5.2).
The existence of only one processor/buyer in southwestern Nova Scotia restricts harvesting to the operational period of the plant. Mechanical harvesters operate 7-9 months of the year dependent upon winter weather conditions. High winds (>30 km h-1) and freezing spray prevent winter operation of harvesters. Hand harvesters use the summer months only since their product must be sun dried to allow its storage for use when mechanical harvesters are unable to operate.
The peak standing crop of Ascophyllum nodosum occurs in the late spring (Section 4.3) corresponding to the maximum size of reproductive structures. Harvesting and processing economics prevent any change in harvesting strategy relative to reproductive periodicity or standing crop.
Hand harvesting of Ascophyllum for commercial use is accomplished with cutting implements sickles and knives. The floating material is encircled by a seine net and loaded by hand or conveyor belt on a vessel (MacArthur, 1971). Later a cutter rake was used consisting of a rake (Fig. 10) with widely spaced tines and triangular cutting edges between the tines (Sharp, 1981). These rakes permitted harvesting and loading in one operation.
Figure 10. An Ascophyllum cutter rake as used in southwestern Nova Scotia. A, cutter tooth from a hay mower; B, rake tine constructed from a boat nail.

In Canada hand harvesting techniques account for less than 20% of the annual landings since the introduction of the Aqua Marine2 H650 harvester to harvest Ascophyllum in 1971. These are a modified aquatic weed harvester 10.5 m long by 4.9 m wide and 2.9 m high driven by paddle wheels through hydraulic motors powered by a diesel engine (Fig. 11). A reciprocating cutter blade cuts a 2.4 m swath; shoots are picked up by a conveyor belt and moved into a 1.4 t capacity storage bay. The operator controls cutting height by raising the forward conveyor belt. Once the holding capacity is reached the vessel moves to a barge and off-loads.
2Aqua Marine Corp., Wakuseka, Wisconsin.
In 1985 a Norwegian suction/cutter harvester was introduced to southwestern Nova Scotia. This harvester is approximately 5 m long by 2.2 m wide and 2.3 m high. The vessel is propelled by water jets near the stern which point sideways, rearward and forward (Fig. 12). A bladed impeller at the end of 25 cm diameter steel suction pipe simultaneously draws up and cuts shoots (Fig. 13). The operator hydraulically controls lifting and lowering of the suction head. Water and cut shoots are discharged into a net bag of approximately 1 t capacity (Fig. 11). The full bag is ejected and towed behind the vessel or moored together awaiting a collecting vessel.
There are few restrictions on the operational area for hand harvesting techniques. Any area reachable by a 5 m boat can be harvested by hand. The intensity of harvesting is largely dependent upon the acceptable level of catch per unit effort. Areas previously cut by mechanical cutters have provided sufficient yield to allow hand harvesting. The mean cutting height of the cutter rake is 23.8+12.9 cm, residual biomass ranges from 10 to 20% of the original values (Sharp, 1981).
Figure 13. Forward view of a Norwegian suction cutter Ascophyllum harvester. A, hydraulic motor drive for impeller; B, cutter/suction head; C, intake pipe and elevation mechanism.

Mechanical harvesters have physical limitations which define the accessibility of a resource. Vessel draft/operational depth of cutter and the seaworthiness of the vessel are the major variables. The ability to operate in wave exposed environments or under high winds is limited due to the danger of cutter impact with rocks. Propulsion systems must be powerful and efficient to overcome wind loads. The Aquamarine harvester has large wind exposed surfaces (15 m²) and an inefficient propulsion system which prevents operations in winds above 30 km h-1. The Norwegian harvester is capable of operating in higher wind velocities due to lower exposed surface (8 m²) but is limited by wave action (>.7 m) (M. Gilje pers. comm3).
3M. Gilje Harvest Manager PROTAN A/S Haugesund, Norway.
The harvesting area of southwestern Nova Scotia has a wide range of wave exposures. In some areas wave exposed shores are close to wave sheltered shores allowing the area of operations to be changed with weather conditions. However extreme wave exposed shores are rarely harvested. (R. Nickerson pers. comm.4).
4R. Nickerson, Harvest supervisor, Scotia Marine Products Limited.
The draft of the vessel determines the maximum height accessible on the shore and the duration of daily harvesting. The upper and lower most portions of the Ascophyllum zone are vulnerable to harvesting for 2 hours of a six hour operational period on a 0.3 m low tide. Frequently the upper portions of the zone are unharvested (Fig. 14). Due to the higher percentage of Fucus in the upper part of the Fucoid zone in southwestern Nova Scotia it is avoided by harvesters (R. Nickerson/pers. comm.).
Figure 14. The harvesting pattern and intensity of the Aquamarine harvester in a trial at a sheltered site, southwestern Nova Scotia. Incidence of harvest in.25 m by 1 in plots in 3 continuous transects from the upper to lower limits of Ascophyllum distribution. Height is relative to chart datum.

The incidence of harvest (proportion of sampled areas 0.25 m² which show evidence of harvest) ranged from 0.27 to 0.61 of areas with Ascophyllum present for the Aquamarine harvester (Fig. 14). The Norwegian harvester left evidence of harvest in 0.16 to 0.29 of the areas with Ascophyllum in them (Fig. 15). Both types of mechanical harvester leave an uneven or patchy distribution.
An area test harvested by the Norwegian harvester had a preharvest biomass (including the upper inaccessible area) of 15.8+11.6 kg m-2 and a post harvest biomass of 11.0+9.6 kg m-2. Excluding the inaccessible zone and areas without biomass, the preharvest biomass was 14.7 kg m-2 compared to an 8.7 kg m-2 post harvest. In a similar test area the Aquamarine harvester did not significantly change the standing crop as assessed in a strip transect method (Sharp, 1981). In the Norwegian harvester test area, 30.0 t wet weight was removed; it was estimated the Aqua- marine harvester would yield 6 t in this area (R. Nickerson, pers. comm.). The mean cutting height of the Aquamarine harvester was 35.2+.6 cm above the holdfast (Fig. 16a). The Norwegian harvester cuts at 29.4+14.9 cm above the holdfast (Fig. 16b). Stump length is normally distributed around the mean value in each case.
Figure 16a. The distribution of stump lengths following the harvest of a sheltered site by an Aquamarine harvester.

Figure 16b. The distribution of stump lengths following the harvest of a sheltered Ascophyllum bed by a Norwegian suction cutter harvester.

The Norwegian harvester removes more biomass from an area though the incidence of harvest is less overall than the Aquamarine harvester because of the cutting head and its manipulation. The Aquamarine harvester propels itself forward thru the bed while the operator moves the cutter head up or down in its 2.4 m swath (Fig. 11). The forward movement of the harvester pushes plants down and away from the cutter thus only a small percentage of shoots is cut in one pass. The machine must make repeated passes over the same area to complete the harvest.
The Norwegian harvester operator lowers the cutter head into a patch of Ascophyllum cutting all shoots within the diameter of the suction pipe (Fig. 13). The Norwegian harvester is very maneuverable allowing targeting of the cutter head on specific patches. In Pubnico, N.S., 50% of the biomass is 65-70 cm above the bottom (Cousens, 1981a,b). If an entire area was harvested at the 30 cm level, 73% of the biomass would be removed (Fig. 7). The Norwegian harvester removed a maximum of 60% of available biomass in such a test area. Shoots are rarely erect in the harvesters path due to the wide range of tide levels. This prevents consistent cropping.
As described in Section 5.1 the harvesting of A. nodosum does not leave the entire population cropped at an even height. Mechanical harvesters leave a remnant biomass of 40%. This remnant biomass consists of stands unreachable by the harvester; shoots uncropped within the path and cutting height of the harvester, shoots below the harvester cutting height, and shoots truncated by the harvester (stumps) (Fig. 17). Recovery depends largely on elongation of basal shoots and renewed lateral branching on stumps. In sheltered areas a lower proportion of shoots would be truncated for the equivalent yield at exposed sites (Fig. 7). Intermediate wave exposure results in maximum productivity and shortest biomass recovery times (Table 1). A recovery of harvestable biomass after one year has been observed at some sites in southwestern Nova Scotia over the 20 years of harvesting. The large amount of residual biomass (40%) is markedly different from the 2% (carrying capacity) biomass remaining in Norway after hand harvest (Seip, 1980). Harvesting models based upon lower degrees of harvesting intensity projected an increase of yield by 40% to 70% on a 2 year reharvest with larger amounts of residual biomass (opp. cit.).
Homogeneous cutting of Ascophyllum at 15-25 cm in 2 of 8 sheltered sites of Maine recovered biomass within one year (Keser et al. 1981). Within sixteen months, following complete denudation of a portion of the Ascophyllum zone and subsequent incursion of Fucus spp., Ascophyllum shoots in the 3-4 cm size class covered 52% of the area (Keser and Larson, 1984). Although in commercial harvesting complete denudation does not occur, over-harvesting using hand cutting methods did occur in a few sites in southwestern Nova Scotia in the early years of the industry (McArthur, pers. comm.5). Normally harvested Ascophyllum populations have a large capacity for biomass recovery. Frond densities average 529 m-2 in Maine and 957-5655 m-2 on Nova Scotian sites (Keser et al. 1981; Cousens, 1981a; Sharp, unpublished). At exposed and sheltered locations on the Atlantic coast of Nova Scotia 10.47% and 2.48% respectively of these shoots carry 50% of the biomass (Cousens, 1981a). Therefore, at cutting heights above 25 cm the shoot density will be relatively unaffected. Prior to harvest primary apices and lateral apices are 60 times shoot density (Keser et al. 1981). Primary growing points will not be reduced in proportion to shoot truncation since the majority of primary shoots are below 15 cm (Fig. 5). Lateral apices are more significantly affected because few laterals are below 15 cm, (Cousens, 1981a). Cutting may provide growth stimulation enhancing biomass recovery. Removal of the Ascophyllum canopy above 15 cm at a semi-sheltered Atlantic site increased primary shoot linear growth by 75% and lateral density per centimeter of shoot by 300% (Cousens 1981a). This effect was attributed to greater light intensity rather than removal of apical dominance (opp. cit.).
5A. McArthur, former Manager, Scotia Marine Products Ltd., Woods Harbour, Nova Scotia.
Figure 17. The major factors affecting recovery of Ascophyllum biomass.

Harvesting Ascophyllum with sickles or cutter rakes yields 2-3 wet tons per individual per tide (4-6 hours). Capital investment is dependent on the size and type of vessel used for harvest and transport. A 3-5 m outboard powered boat costs were Canadian $18.50 d-1 in 1975 dollars. Most operations require a larger boat or barge for several harvesters and such may cost Canadian $32.50 d-1 if amortized over 10 years (Sharp and Neish, 1976). To complete drying of the raw material a transport vehicle and a drying field are added to the costs (Table 6). The return to the harvester was assumed to be Canadian $4.00 hr however, due to inflation and competition for labor, present day costs would be at least doubled. One wet ton required 2.3 hours of labor costs in travel/harvest and unloading (Table 7). Exclusive of drying operations; labor and capital costs were Canadian $10.65 respectively per wet ton; drying added Canadian $4.78 labor and Canadian $0.47 capital.
Mechanical harvesting increases the productivity per individual but greatly increases capital costs. The Aquamarine harvester harvests 2 to 3 t h-1 but costs Canadian $70/000 per unit. An average yield of 12.5 wet ton per day and 120 operational days per year was used to calculate operational costs (Table 8 and 9). If fresh materials are utilized directly, costs are reduced by Canadian $7.32 per wet ton (Table 9). Operations in southwestern Nova Scotia requiring a tow vessel and 120 t capacity barge require Canadian $250,000 capital costs, amortizable over 15 years with daily operational costs of Canadian $240 resulted in a total daily cost of Canadian $370. If daily production is a multiple of harvesting units (x12-18 t d-1) total costs are Canadian $38.71 per ton. The Norwegian harvester capital cost is approximately Canadian $138,000 but daily production is 50 t per unit. The harvesting operation does not require a barge or large tow vessel assuming the harvest boat can tow bags directly to the plant. Under these circumstances capital and operating costs per ton are 9.78 per ton. Transport overland would involve a 25 t capacity truck and crane at a cost similar to the tow vessel and barge combination.
Persons hand harvesting Ascophyllum use this activity as a 5% to 20% supplement to their annual income. Alternative income sources during the summer months are ground fish, fishing or processing, herring fishing by seine or gillnet and Chondrus crispus (Irish moss) harvesting (Sharp and Roddick 1982).Some individuals combine Ascophyllum and Chondrus crispus, harvesting them on either alternating tides or using the low tide period for Chondrus and rising or falling tides for Ascophyllum . The remainder of the year fishermen either captain or deckhand on a lobster fishing vessel (Sharp and Roddick 1982). Individuals employed directly by the processing company to operate Aquamarine machines are active from March-April to November-December depending on weather conditions in the early fall and spring. Alternate employment is lobster fishing or utilization of the Canadian Unemployment benefit program. Harvesting income comprises 60 to 100% of employment income for these individuals. Family income in southwestern Nova Scotia averages Canadian $20,000 to $22,000; the male contributes 55 to 65% of this income (Raymond 1985). Primary industry, including fishing and marine plant harvesting employs 26 to 34% of the labor force (opp. cit.).
Table 6. Capital and operating costs associated with hand harvesting of rockweed in Year 1, assuming that all equipment except holding nets and the collecting seine are shared with other fisheries and that these nets would be used for at least 50 days per year (or would have proportionately extended amortization rates if used less). A mean daily harvest of 8.4 net metric tons per day was assumed for a three-man harvest crew. (Sharp and Neish 1975).
|
|
Gross |
Amort. Rate |
Deprec. Cost |
Hourly Op. Cost |
Maint. Cost as % of |
Maint. cost |
Days of use in all |
Daily cost for Rockweed |
|
|
a) |
Harvesting boat and barge |
20,000 |
10 |
2,000 |
5.00 |
10 |
2,000 |
200 |
32.50 |
|
b) |
Winch, mast and boom |
4,000 |
10 |
400 |
- |
10 |
400 |
100 |
8.00 |
|
c) |
Dory or skiff |
1,000 |
10 |
100 |
- |
10 |
100 |
100 |
2.00 |
|
d) |
25-HP motor |
1,400 |
5 |
280 |
2.00 |
10 |
140 |
100 |
6.20 |
|
e) |
Large holding nets (20) |
1,000 |
2 |
500 |
- |
20 |
200 |
50 |
14.00 |
|
f) |
Collecting seine(100 x 2m) |
600 |
5 |
120 |
- |
20 |
120 |
50 |
4.80 |
|
g) |
Truck (3 ton) |
14,000 |
5 |
2,800 |
6.00 |
10 |
1,400 |
200 |
24.00 |
|
h) |
Drying field(0.2 ha) |
1,250 |
20 |
63 |
- |
10 |
125 |
100 |
1.88 |
|
|
Daily Cost |
|
|
|
|
|
|
|
93.38 |
|
|
Cost/wet metric ton |
|
|
|
|
|
|
|
11.12 |
|
|
Cost exclusive of drying |
|
|
|
|
|
|
|
10.65 |
Table 7. Labour costs associated with hand harvesting of rockweed assuming a desired return to the fisherman of $4.00 per hour and a daily harvest rate of 8.4 wet metric tons per day (Sharp and Neish 1976).
|
Activity (See Figure 1) |
|
Crew size |
Man-Hours Day-1 |
Labour Cost at-$4.00 Hr-1 |
Labour Cost per wet metric ton ($) | |
|
Travel |
(1) |
3 |
0.75 |
3.00 |
.35 | |
|
Harvest |
(2) |
3 |
15.00 |
60.00 |
7.14 | |
|
Return to port |
(3) |
3 |
0.75 |
3.00 |
.35 | |
|
Unload Harvest |
(4) |
3 |
3.00 |
12.00 |
1.43 | |
|
Ship |
(6) |
3 |
1.50 |
6.00 |
.71 | |
|
Unload Truck |
(9) |
4 |
1.50 |
6.00 |
.71 | |
|
Spread Crop |
(10) |
4 |
4.00 |
16.00 |
1.90 | |
|
Attend Crop |
(11) |
- |
- |
- |
- | |
|
Collect and pack * |
(12) |
4 |
6.00 |
24.00 |
2.85 | |
|
Hill and pack |
(13) |
|
|
|
| |
|
|
Totals |
|
|
32.50 |
130.00 |
15.47 |
|
|
exclusive of drying |
|
10.69 | |||
*Costs associated with drying.
Table 8. Labour costs associated with mechanical harvesting assuming a desired return to the fishermen of $4.00 per hour and a daily harvest of 12.5 metric tons. All figures are estimates based on experience with other crops (Sharp and Neish 1976).
|
Activity (See Figure 1) |
Crew size |
Man-Hours Day-1 |
Labour Cost at $4.00 Hr-1 |
Labour Cost per wet metric ton ($) | |
|
Travel |
2 |
2.00 |
8.00 |
0.64 | |
|
Harvest |
2 |
12.00 |
48.00 |
3.84 | |
|
Return to port |
2 |
2.00 |
8.00 |
0.64 | |
|
Unload boat |
4 |
8.00 |
32.00 |
2.56 | |
|
Ship |
2 |
1.00 |
4.00 |
0.32 | |
|
Unload Truck |
4 |
8.00 |
32.00 |
2.56 | |
|
Spread Crop* |
4 |
12.00 |
48.00 |
3.84 | |
|
Attend Crop* |
- |
- |
- |
- | |
|
Collect and pack* |
4 |
12.00 |
48.00 |
3.84 | |
|
Mill and pack* |
|
|
|
| |
|
|
Totals |
|
57.00 |
228.00 |
18.24 |
|
Total exclusive of drying and packing |
|
|
|
10.56 | |
*Costs associated with drying operations
Table 9. Capital and operating costs associated with mechanical rockweed harvesting in Year 1 using the Aquamarine HN462 harvester. A harvest rate of 12.5 wet metric tons per day and sharing of boat and truck costs with other fisheries are assumed. Harvest rate is based on estimated rates achieved by Scotia Marine in Nova Scotia (Sharp and Neish 1975).
|
|
Gross |
Amort. Rate |
Deprec. Cost |
Hourly Op. Cost |
Maint.Cost as% of |
Maint. cost |
Days of use in all |
Daily cost ($) at 12.5m.t./day |
|
|
a) |
Harvest boat & skiff |
20,000 |
10 |
2,000 |
5.00 |
10 |
2,000 |
200 |
32.50 |
|
b) |
Winch, mast, boom |
4,000 |
10 |
400 |
- |
10 |
400 |
200 |
4.00 |
|
c) |
HN462 Harvester |
70,000 |
7 |
10,000 |
5.00 |
10 |
7,000 |
- |
182.00 |
|
*d) |
Holding nets (50) |
2,500 |
2 |
1,250 |
- |
20 |
500 |
- |
14.58 |
|
*d) |
Tractor |
8,500 |
10 |
850 |
5.00 |
10 |
850 |
- |
54.00 |
|
*f) |
Spreader |
3,500 |
10 |
350 |
- |
10 |
350 |
- |
5.83 |
|
g) |
Baler and handling gear |
10,000 |
10 |
1,000 |
- |
10 |
1,000 |
- |
16.67 |
|
h) |
3-Ton Truck |
14,000 |
5 |
2,800 |
6.00 |
10 |
1,400 |
200 |
24.00 |
|
*i) |
Drying ground(1 ha.) |
6,000 |
20 |
1,200 |
- |
10 |
600 |
- |
15.00 |
|
|
Daily Cost |
|
|
|
|
|
|
|
348.58 |
|
|
Cost/wet metric ton |
27.89 |
|||||||
|
|
Exclusive of drying |
20.57 |
|||||||
*Costs of drying
Until recently the aquaculture of macrophytes, such as Ascophyllum , which dominate the intertidal algal biomass of many North Atlantic shores was not considered economically viable. The desire to control composition of phycolloid end products has justified applied research on culture techniques.
A spray technique first developed by L. Hanic University of Prince Edward Island for grow out of macrophytes has been further developed to grow Ascophyllum on draining surfaces (Rheault and Ryther, 1983). Spraying with unenriched seawater and pulse feeding of nutrients obtained 14.3 g m-2 d-1 Similar experiments with a multiple tier growing system and continuous addition of nutrients produced 40 g m-2 d-1 of dry matter (Moeller et al. 1984). Excluding land and inoculum costs this system produced one ton of Ascophyllum at a cost of U.S. $1620 per dry ton (opp. cit.). Present costs of harvesting and delivering the factory naturally grown Ascophyllum is approximately US $140 per dry ton (See Section 5.3).
Local phycologists recognized the problems of over harvesting Ascophyllum resources, as observed in Europe/were possible in the Maritime Province (C. MacFarlane, pers. comm.). Provincial governments enacted Sea Plants Harvesting Acts in response to the advice of seaweed experts. These regulations included harvesting gear restrictions requiring the use of cutting implements only to prevent holdfast loss by regulating cutting height at 12.7 cm above the substrate to allow regrowth.
The Sea Plants Act of Nova Scotia made provision for exclusive licenses for designated portions of the shoreline for up to 15 years. Licenses were granted to companies providing a monopoly for raw material from that area. It was the responsibility of the licensee to respect the regulations within his region. A company could then have numerous harvesters as contractors in this license. In 1975 the responsibility for management of marine plant resources were transferred to the Federal government of Canada. The Sea Plants Harvesting Act was incorporated under the Atlantic Coast Marine Plants Regulations within the Fisheries Act. In 1985 the Atlantic Coast Marine Plant Regulations were incorporated in the Atlantic Fishery Regulations Part I (Appendix I). The regulations were not altered but provision was made for alteration of the harvesting season and setting of quotas, Part I (Appendix I).
There were two major changes in regulations from the Provincial Act. Marine Plant Harvesting districts No.'s 1-14 were established (Fig. 1) and licenses were personal and annually renewed. No exclusive privileges to the resource were provided and a fisherman's boat registration and general fisherman's license were required. Enforcement of regulations is the responsibility of the Department of Fisheries and Oceans, Operations Branch regional fisheries officers. All purchases of marine plants must be recorded on individual purchase slips for statistical reporting. Due to the large number of small (<1 t) transactions purchasing companies are permitted to report total monthly landings by port to the Statistical Branch of Fisheries and Oceans Canada. In areas where less than 2 companies are active landings are not made public.
As marine plants form a part of the fish habitat Section 31 of the Fisheries Acts of 1977 provides protection against disruption of the habitat. "No person shall carry on any work or undertaking that results in the harmful alteration/description or destruction of fish habitat".
There are no additional restrictions/quota/season or area of operations which require annual assessment of standing crop. The Research Branch of the Federal department of Fisheries has the mandate to obtain that biological information relevant to resource management.
Biologists are provided with species and regional responsibilities within the branch. Information relevant to management advice must pass thru a peer review process known as the Canadian Atlantic Fisheries Scientific Advisory Committee (CAFSAC) and thence as advice to upper level fisheries managers.
To communicate information to the industry/regional Marine Plant Advisory Committees were established including representatives of government, processors/buyers and harvesters. The biologist can present his advice to these committees and the committees can provide recommendations directly to the Minister of Fisheries via an area manager.
8.1 Chemical and nutritional content
8.2 Human food
8.3 Animal fodder
8.4 Fertilizer
8.5 Industrial products and processes
The early industrial utilization of Ascophyllum nodosum required for practical purposes an analysis of its constituents. Baardseth (1970) recorded studies of a wide range of organic and inorganic substances in A. nodosum. Seasonal/geographical, and inter-thallus variations of water content, trace elements, alginates, and pigments were among the topics reviewed (Table 10). Since 1970, more detailed data on some of these constituents were recorded and additional substances identified.
Pharmacological research continues to find potential in seaweeds. Fucoidan, as prepared from Ascophyllum nodosum, shows excellent metal binding abilities and binds lead most efficiently of the divalent metal cations (Paskins-Hurlburt et al. 1976). In vivo experiments a 75% reduction in lead absorbed by rats from their gastrointestinal system was observed.
The antihypercholesterol activity of sterols in algae has led to studies of their content in Ascophyllum nodosum (Knights, 1970; Safe et al. 1974). Fucosterol comprises over 95% of the sterol content of A. nodosum. The presence of other sterols was considered the result of oxidation (Knights, 1970). A more detailed study of sterol content found 0.10% sterols of dry weight, of which fucosterol was 90% with positive identification of five other sterols of which saringosterol was 6% (Safe et al. 1974). Hemagglutinic activity was found with solutions of powdered A. nodosum tested on human blood groups. No differences were found in hemagglutinic activity between thallus regions (Blunden et al. 1975).
Table 10. Average composition of Norwegian seaweed meal (Baardseth, 1970 from Jensen Nebb and Saester 1966).
|
Component |
Content |
Component |
Content | ||
|
Moisture |
12-15 |
% |
Crude fibre |
8 |
% |
|
Ash |
17-20 |
% |
Crude protein |
5-10 |
% |
|
Alginic acid |
20-26 |
% |
Ether extract |
2- 4 |
% |
|
Mannitol |
5- 8 |
% |
Fucoidan |
10 |
% |
|
Laminaran |
2- 5 |
% |
N-free extractives |
45-60 |
% |
|
S |
2.5-3.5 |
% |
Caloric value |
0.56 |
SPU/kg*) |
|
K |
2- 3 |
% |
Ascorbic acid |
500-2000 |
mg/kg |
|
Cl |
3.1-4.4 |
% |
Carotene |
30-60 |
mg/kg |
|
Na |
3- 4 |
Biotin |
0.1-0.4 |
mg/kg | |
|
Mg |
0.5-0.9 |
% |
Folic acid |
0.1-0.5 |
mg/kg |
|
Ca |
1- 3 |
% |
Folinic acid |
0.1-0.5 |
mg/kg |
|
P |
0.1-0.15 |
% |
Niacin |
10-30 |
mg/kg |
|
B |
40-100 |
mg/kg |
Riboflavin |
5-10 |
mg/kg |
|
CO |
1-10 |
mg/kg |
Tocopherols |
150-300 |
mg/kg |
|
Fe |
150-1000 |
mg/kg |
Vit.B12 |
0.004 |
mg/kg |
|
Mn |
10-50 |
mg/kg |
Vit.K |
10 |
mg/kg |
|
I |
700-1200 |
mg/kg |
V |
1.5- 3 |
mg/kg |
|
Zn |
50-200 |
mg/kg |
Ni |
2- 5 |
mg/kg |
|
Mo |
0.3- 1 |
mg/kg |
Ba |
15-50 |
mg/kg |
*) SFU = Scandinavian feed units
Alginic acid 150-1000 mg/kg N-free extractives 500-2000 mg/kg
Vitamin content of Ascophyllum nodosum was extensively catalogued by Baardseth (1970). However, several concurrent papers were not included in the synopsis. Tocopherol (Vitamin E) content and its variation with individual diurnal, and seasonal changes/was investigated after a rapid and accurate analytical technique was developed (Jensen, 1969a). Total tocopherol was maximal in September and February at 500 mg/kg dry matter (Jensen/1969b). Proportions of tocopherols varied with immersion and emersion periods. Optimally growing plant sections contained less total tocopherol than older portions of the plant (Jensen opp. cit.). Choline is one of the B complex vitamins which is included in commercial feeds. The choline content of A. nodosum was 0.9 to 3.2 mg/g dry weight over one year. Highest choline content occurred in winter and spring, dropping in June, July, and August (DaSilva and Jensen, 1973).
Anti-microbial compounds were detected in Ascophyllum nodosum as early as 1954 (Vacca and Walsh/1954). Recent studies have found summer antimicrobial activity but none in spring or winter (Horsney and Hide 1976). This points out the further need for year round sampling in any study of chemical constituents.
Several recent studies of Ascophyllum nodosum chemical composition relate directly to an overall metabolic strategy within its geographical and zonal ranges. Amino acid composition was found to change with decreasing salinity (Munda, 1977). Nitrogen content increased with lower salinities. This change was attributed directly to the increasing presence of glutamic acid which is a key amino acid in the synthesis of other amino acids (opp. cit.).
Exposure and season were found to have the greatest correlation with caloric value of Ascophyllum nodosum tissues (Breton-Provencher/1976). Maximal calorific values occur in July in the period of maximum growth. The highest overall calorific values corresponded to locations where A. nodosum was dominant over other Fucoids.
Growth hormones were extensively investigated in higher plants and have only recently been examined in detail for algae. G.L.C. analysis of water soluble components from Ascophyllum nodosum found compounds similar to abscisic acid. These compounds were found to inhibit growth of lettuce hypocotyl (Hussain and Boney, 1973). Auxin-like/gibberellin-like, and cytokinin-like substances were found in A. nodosum extracts using a bio-assay system (Kingman and Senn, 1977). A seasonal survey of gibberellin activity in three fucoids showed a maximum activity of extracts occurred in October for A. nodosum and a minimum of gibberellin activity in January (Wildgoose et al. 1978).
Descriptions of alginate structure and its composition in a variety of algal species has been ongoing since 1970. The sequences of uronic acid, building blocks of alginate, were described for Ascophyllum nodosum (Smidsrod & Glover 1973). Although earlier studies have described wide difference between the relative amounts of the various uronic acid groups, recent work indicates all young tissue is rich in manuronic blocks (Haug et al. 1974). Therefore, any differences in composition of alginates occurs in older portions of the plants (opp. cit.). Other polysaccharides were isolated from A. nodosum and their structure examined. Fucoidan was isolated and described as a sulphated glucuronoxylogucan, existing as a building block of the complex macromolecule (Larsen et al 1970).
Concern for the effects of hydrocarbon spills in our coastal environment lead to an assessment of natural hydrocarbon background levels in various benthic algae (Youngblood et al. 1971). N-pentadecane predominates in Ascophyllum nodosum and other tested brown algae. Polyunsaturated hydrocarbons occur only in the reproductive tissues of A. nodosum.
Ascophyllum nodosum is not normally used directly as a foodstuff but may appear as a constituent of health food tablets. The polysaccharide algin extracted from Ascophyllum as alginic acid is used in the form of salts as a viscosity control gelating and emulsifying agent. Alginic acid is composed of polymannu-ronic acid segments, polyguluronic acid segments and alternating segments of these acids. Ascophyllum nodosum alginic acid is composed of 65% mannuronic acid, 35% guluronic acid the ratio of these components can range between 1.40 and 1.95 (Cottrell and Kovacs, 1980). Common food uses for alginates include prepared pie fillings, instant mixes and sauces.
Ascophyllum nodosum is used as a stock feed additive. It may make up to 5% of the diet for poultry, sheep, cattle, pigs and horses. The trace element and vitamin components of Ascophyllum meal are the active ingredients for growth of cattle, milk production, color in eggs and improving wool color in sheep (Neeb and Jensen 1965). Canada has imported seaweed meal for use as a fodder supplement. Agricultural committees near the sea have allowed farm animals access to storm tossed seaweed.
The use of fresh Ascophyllum as a soil conditioner, source of trace elements and general growth stimulant began in the earliest settlements of Maritime Canada. European settlers brought this custom from centuries of use in Europe. Liquid seaweed extracts developed since 1950 and wetable powders are used as in foliar sprays. Extracts have proven chelating properties that improve the utilization of minerals (Lynn 1972). Application of extracts to seeds increases germination and early growth (Britton and Nayes, 1964). Auxin and gibberellin like activities are reported for seaweed extracts but the results are conflicting. Soluble powders as foliar sprays increased protein content of soybeans, soluble solids in tomatoes and yield of Southern pea (Annon, 1978).
A liquid extract of Nova Scotian Ascophyllum nodosum was produced in Yarmouth by Argosy Seaweeds Ltd., but commercial production did not follow.
Sodium alginate is used as a part of a sizing solution for liner-board. Alginate enhances penetration of adhesives, acts as a dye carrier in fire reactive dyes. Its gelling properties are used in air fresheners and explosives. As an emulsifier it is used in polishes. The stabilizing properties of alginates make them useful in ceramics, welding rods and cleaners. The industrial processing of Ascophyllum nodosum includes a fixative step in addition to those shown in (Fig. 18) to prevent coloration of the final product.
Archambault, D., and E. Bourget, 1983 Importance du régime de dénudation sur la structure et la succession des communautés intertidales de substrat rocheux en milieu subarctique. Can.J.Fish.Aquat.Sci., 40 (8):1278-92
Asare, S., and M. Harlin, 1983 Seasonal fluctuations in tissue nitrogen for five species of perennial macroalgae in Rhode Island Sound. J.Phycol., 19:254-7
Baardseth, E., 1970 Synopsis of biological data on knobbed wrack, Ascophyllum nodosum (Limnaeus) Le Jolis. FAO Fish. Synop., (38) Rev.1:41 p.
Bell, H.P., and C. MacFarlane, 1933 The marine algae of the Maritime Provinces of Canada. 1. List of species with their distribution and prevalence. Can.J.Res., 9:265-93
Bernard-Theriault, L., and A. Cardinal, 1973 Variations de la teneur en acide alginique des Fucacées de l'estuaire du Saint Laurent (Quebec) en fonction de certains facteurs écologiques. Bot.Mar., 16:96-102
Blunden, G., and D.J. Rogers, 1975 Survey of British seaweeds for hemogglutinins. Lloydia, 38(2):162-8
Breton-Provencher, M., 1976 Aspects écologiques de la production des algues benthiques mediolittorales de la région de bic, estuaire Maritime du Saint-La.urent. M.Sc. Thesis, Université de Laval, Quebec, P.Q.
Britton, E.F., and C.F. Nayes, 1964 Effect of seaweed extract on emergence and survival of seedlings of creeping and red fescue. Agron.J., 56:444-5
Cardinal, A., 1967 Inventaire des algues marines benthiques de la Baie des Chaleurs et de la Baie de Gaspe (Quebec). 1. Pheophycees. Nat.Can., 94:233-71.
Cardinal, A., and M. Villalard, 1971 Inventaire des algues marines benthiques de l'estuaire du Saint-Laurent (Quebec). Nat.Can., 98:887-904
Clemson University, 1978 Department of Horticulture, Seaweed Research in Crop Production, 1958-78. Washington, D.C., Economic Development Administration, 185 p.
Chock, J.S., and A.C. Mathieson, 1983 Variations of New England estuarine seaweed biomass. Bot.Mar., 26:87-97
Cottrell, I.W., and P. Kovacs, Alginates, 1980 pp. 2-1-2-43. In: Handbook of water-soluble gums and resins, edited by R.L. Davidson. New York, McGraw-Hill, pp.2-1-2-43
Cousens, R., 1981 The population biology of Ascophyllum nodosum. Ph.D Thesis. Dalhousie University/Halifax, Nova Scotia
Cousens, R., 1981a Variation in annual production by Ascophyllum nodosum with degree of exposure to wave action. Proc.Int. Seaweed Symp., 10:253-8
Cousens, R., 1982 The effect of exposure to wave action on the morphology and pigmentation of Ascophyllum nodosum (L.) Le Jolis in southeastern Canada. Bot.Mar., 25: 191-5
Cousens, R., 1984 Estimation of annual production by the intertidal brown alga Ascophyllum nodosum (L.) Le Jolis. Bot. Mar., 27;217-27
DaSilva, E., and A. Jensen, 1973 Benthic marine and blue-green algal species as a source of choline. J-Sci.Food Agric., 24:855-61
David, H.M., 1943 Studies in the autoecology of Ascophyllum nodosum Le Jol. J.Ecol., 31:178-98
Dorgelo, J., 1976 Intertidal fucoid zonation and desiccation. Hydrobiol.Bull., 10(2):112-5
Edelstein, T., L. Chen and J. McLachlan, 1970 Investigations of the marine algae of Nova Scotia. 8. The flora of Digby Neck Peninsula, Bay of Fundy. Can.J.Bot., 48(3):621-9
Edelstein, T., and J. McLachlan, 1966 Investigations of the marine algae of Nova Scotia. I. Winter flora of the Atlantic coast. Can.J.Bot., 44:1035-55
Fortes, M.D., and K. Luning, 1980 Growth rates of North Sea macroalgae in relation to temperature, irradiance and photoperiod. Helgol.Meeresuntes., 34:15-29
Gunnarson, K., and K. Porisson, 1976 The effect of sewage on the distribution and cover of littoral algae near Reykjavik. Arch.Bot.Isl., 4:58-66
Hariot, P., 1889 Liste de algues recurillies a l'ile Miquelon por M. le docteur Delamare. J.Bot., 3:154-7
Haug, A., B. Larsen and O. Smidsrod, 1974 Carbohyd.Res., 32:217
Hawkins, S.J., and R.G. Hartnoll, 1985 factors determining the upper limits of intertidal canopy-forming algae. Mar. Ecol., 30(3):265-71
Hornsey, I.S., and D. Hide, 1976 The production of anti-microbial compounds by British marine algae. Part 2. Seasonal variation in production of antibiotics. Brit. Phycol.J., 11(1):63-7
Hussain, A., and A.D. Boney, 1973 Hydrophilic growth inhibitors from Laminaria and Ascophyllum . New Phytol., 72:403-10
Jensen, A., 1969 Tocopherol content of seaweed and seaweed meal. 1. Analytical methods and distribution of tocopherols in benthic algae. J.Sci.Food Aqric., 20:449-53
Jensen, A., 1969a Seasonal variations in the content of individual tocopherols in Ascophyllum nodosum, Pelvetia canaliculata and Fucus serratus. Proc.Int.Seaweed Symp., 6:493-500
Josselyn, M.N., and A.C. Mathieson, 1978 Contribution of receptacles from the fucoid Ascophyllum nodosum to the detrital pool of a north temperate estuary. Estauries,1:(4): 258-61
Keser, M., and B.R. Larson, 1984 Colonization and growth of Ascophyllum nodosum (Phaeophyta) in Maine. J.Phycol., 20: 83-7
Keser, M., R.L. Vadas and B.R. Larson, 1981 Regrowth of Ascophyllum nodosum and Fucus vesiculosus under various harvesting regimes in Maine, U.S.A. Bot.Mar., 24:29-38
Khailov, K.M., 1976 The relationships be tween weight, length, age and intensity of photosynthesis and organotrophy of macrophytes in the Barents Sea. Bot.Mar., 19:335-9
Kingman, A.R., and T.L. Senn, 1977 Bioassay systems to test for plant growth hormones in extracts of Ascophyllum nodosum. J.Phycol., 13(Suppl.): 36
Knights, B.A., 1970 Sterols in Ascophyllum nodosum. Phytochemistry, 9:903-5
Larsen, B., A. Haug and T. Painter, 1970 Sulphated polysaccharides in brown algae. 3. The native state of fucoidan in Ascophyllum nodosum and Fucus vesiculosus. Acta Chem.Scand., 24:3339-52
Lee, R.K.S., 1968 A collection of marine algae from Newfoundland. I. Introduction and Phaeophyta. Nat.Can., 85:957-78
Lee, R.K.S, 1980 A catalogue of the marine algae of the Canadian Arctic. NatlMus.Can.Publ.Bot., (9):82 p.
Lein, T.E., J. Rueness and O. Wiik, Algological observations in 1974 and the adjacent fjord areas SE Norway. Biyttia, 32:155-68
Lubchenco, J., 1978 Plant species diversity in a marine intertidal community: importance of herbivore food preference and algal competitive abilities. Am.Nat., 112: 22-39
Lubchenco, J, 1980 Algal zonation in a New England rocky intertidal community: an experimental analysis. Ecology, 61: 333-44
Lynn, L.B., 1972 The chelating properties of seaweed extract, Ascophyllum nodosum vs. Macrocystis pyrifera, on the mineral nutrition of sweet peppers, Capsicum annum. M.Sc. Thesis. Clemson University, South Carolina
MacDonald, M.A., D.S. Fensom and A.R.A. Taylor, 1974 Electrical impedance in Ascophyllum nodosum and Fucus vesiculosus in relation to coding, freezing and desiccation. J.Phycol., 10(4):462-9
MacFarlane, C., 1932 Observations on the annual growth of Ascophyllum nodosum. Proc.Trans.Nova Scotian Inst. Sci., (1932): 27-32
MacFarlane, C, 1952 A survey of certain seaweeds of commercial importance in southwest Nova Scotia. Can.J.Bot., 30:78-97
Mathieson, A.C., E. Tveter, M. Daly and J. Howard, 1977 Marine algal ecology in a New Hampshire tidal rapid. Bot.Mar., 20:277-90
Mathieson, A.C., et al., 1976 Seasonal growth and reproduction of estuarine fucoid algae in New England. J.Exp.Mar. Biol.Ecol., 25:273-84
Mathieson, A.C., et al, 1982 Effects of ice on Ascophyllum nodosum within the Great Bay Estuary System of New Hampshire. J.Phycol., 18:331-6
McArthur, A.C., 1971 Harvesting and extraction of Ascophyllum nodosum in Nova Scotia. In Proceedings of the Meeting on the Canadian Atlantic seaweeds industry. Charlottetown, P.E.I. Industrial Development Branch, Fisheries Service Canada, pp.67-70
Menge, B.L., 1975 Effects of herbivores on community structure of the New England rocky inter-tidal region: distribution and diversity of algae. Ph.d. Thesis. Harvard University, Cambridge, Mass., 165 p.
Moeller, H.W., S.M. Garber and G. Giffin, 1984 Biology and economics of growing seaweeds on land in a film culture. Hydrobiologia, 116/117:299-302
Munda, I.M., 1967 Der einfluss des saizgehaltes auf die chemische zzusammensetung wachstum und fruktifizierung van einigen Fucaceen. Nova Hedwigia, 13:471-508
Munda, I.M, 1977 Differences in amino acid composition of estuarine and marine fucoids. Aquat.Bot., 3:273-80
Neeb, H., and A. Jensen, 1965 Seaweed meal as a source of minerals and vitamins in rations for dairy cows and bacon pigs. Proc.Int.Seaweed Symp., 5:387-93
Neish, I., 1971 The distribution and abundance of rockweed on the Bay of Fundy shore of New Brunswick. Report to the New Brunswick Department of Fisheries and Environment. Atlantic Mariculture Ltd.
Normandeau Associates, 1977 Seabrook benthic report 7-6. Manchester, New Hampshire, Public Service Co. of New Hampshire, 437
Paskins-Hurlburt, A., et al., 1976 Isolation and metal binding properties of fucoidan. Bot.Mar., 19:327-8
Pielou, E.G., 1981 Rapid estimation of the standing crop of intertidal fucoids on an exposed shore. J.Environ.Manage., 13: 85-98
Pringle, J.D., and R.E. Semple., 1980 Benthic algal biomass, commercial harvesting, and Chondrus growth and colonization off southwestern Nova Scotia. Can.Tech.Rep.Fish.Aquat. Sci., (954):144-69
Printz, H., 1956 Recuperation and recolonization in Ascophyllum . Proc. Int.Seaweed Symp., 2:194-7
Ramus, J., F. Lemons and C. Zimmerman, 1977 Adaptation of light-harvesting pigments to downwelling light and the consequent photosynthetic performance of the eulittoral rockweeds Ascophyllum nodosum and Fucus vesiculosus Mar.Biol., 42:293-303
Raymond, J., 1985 Scotia-Fundy region fishing community profiles. Can. Data Rep.Fish.Aquat.Sci., (540):461 p.
Rheault, R.B., and J.H. Ryther, 1983 Growth, yield and morphology of Ascophyllum nodosum (Phaeophyta) under continuous and intermittent seawater spray culture regimens. J. Phycol., 19:252-4
Rueness, J., 1973 Pollution effects on littoral algal communities in inner Oslofjord with special reference to Ascophyllum nodosum. Helgol.Meeresunters., 24:446-54
Safe, L.M., C.J. Wong and R.F. Chandler, 1974 Sterols in marine algae. J.Pharm.Sci., 63(3):464-66
Seip, K.L., 1980 A computational model for growth and harvesting of the marine alga Ascophyllum nodosum. Ecol.Modelling, 8: 189-200
Schonbeck, M., and T. Norton, 1978 Factors controlling the upper limits of fucoid algae on the shore. J.Exp.Mar. Biol.Ecol., 31:303-13
Schonbeck, M., and T. Norton, 1980 Factors controlling the lower limits of fucoid algae on the shore. J.Exp.Mar.Biol.Ecol., 43:131-50
Sharp, G.J., 1981 An assessment of Ascophyllum nodosum harvesting methods in southwestern Nova Scotia. Can.Tech.Rep. Fish.Aquat.Sci., (1012):28 p.
Sharp, G.J., and I Neish, 1976 Cost estimates for the harvesting, holding and mechanical reduction of various seaweed products from Charlotte Co., New Brunswick. Report of Atlantic Mariculture Ltd. to New Brunswick Department of Fisheries.
Sharp, G.J., and D.L. Roddick, 1982 Catch and effort trends of the Irish moss (Chondrus crispus Stackhouse) fishery in southwestern Nova Scotia, 1978 to 1980. Can.Tech. Rep.Fish.Aquat.Sci., (1118): 4 3 p.
Sheader, A., and B. Moss, 1975 Effects of light and temperature on germination and growth of Ascophyllum nodosum (L.) Le Jol. Estaur.Coast.Mar.Sci., 3:125-32
Smidsrod, O. and R.M. Glover, 1973 The relative extension of alginates having different chemical composition. Carbohydr.Res., 27(1):107-18
Smith, R., and R. Loucks, 1980 A literature and photo-assessment of the marine plant biomass of eastern Canada. A report to the Atlantic Regional Laboratory, National Research Council of Canada
South, G.R., and R.D. Hill, 1970 Studies on marine algae of New-foundland. 1. Occurrence and distribution of free living Ascophyllum nodosum in Newfoundland. Can.J. Bot., 48 (10):1697-701
South, R., and R. Hooper, 1980 A catalogue and atlas of the benthic marine algae of the island of Newfoundland. St. Johns, Memorial University of Newfoundland, Occasional papers in biology, 136 p.
Stromgren, T., 1976 Length changes of growing apices of eulittoral fucales during drying and soaking. Sarsia, 61:47-54
Stromgren, T., 1977 Length growth rates of three species of intertidal Fucales during exposure to air. Oikos, 29:245-9
Stromgren, T., 1983 Temperature-length growth strategies in the littoral alga Ascophyllum nodosum (L.). Limnol.Oceanogr., 28(3):516-21
Sundene, O., 1973 Growth and reproduction in Ascophyllum nodosum (Phaeophyceae). Norw.J. Bot., 20:249-55
Taylor, W.R., 1957 Marine algae of the northeastern coast of North America. Ann Arbor, Mich., University of Michigan Press, 509 p.
Terry, L.A., and B.L. Moss, 1980 The effect of photoperiod on receptacle initiation in Ascophyllum nodosum. Brit. Phycol.J., 15:291-301
Topinka, J., L. Tucker and W. Korjeff, 1981 The distribution of fucoid macroalgal biomass along central coastal Maine. Bot.Mar., 24: 311-9
Vacca, D.D., and R.A. Walsh, 1954 The anti-bacterial activity of an extract obtained from Ascophyllum nodosum. J.Am.Pharm.Assoc. (Sci.Ed.), 43:24-6
Vadas, R.L. and M. Keser, 1972 Field studies on growth rates in Ascophyllum nodosum. J.Phycol., 8:9 p.
Vadas, R.L., M. Keser and P.C. Rusanowski, 1976 Influence of thermal loading on the ecology of intertidal algae, pp. 202-212. In Thermal ecology, 2, edited by G.W. Esch and R.W. MacFarlane. ERDA Symp.Ser., (CONF-750425): 202-12
Youngblood, W.W., et al., 1971 Saturated and unsaturated hydrocarbons in marine benthic algae. Mar.Biol., 8:190-201
Wilce, R.T., 1959 The marine algae of the Labrador Peninsula and northwest Newfoundland (ecology and distribution). Bull.Natl.Mus.Can., (158):105 p.
Wildgoose, P.B., C. Blunden and K. Jewers, 1978 Seasonal variations in gibberellin activity of some species of Fucaceae and Laminariaceae. Bot.Mar., 21:63-5
PART I GENERAL
Variation of Close Times and Quotas
4. A Regional Director-General may, by order, vary any close time or fishing quota fixed by these Regulations.
5. Where, pursuant to Section 4, a Regional Director-General varies a close time or a fishing quota, he shall, by one or more of the following methods, give notice of the variation to the persons affected or likely to be affected by the variation:
SCHEDULE XXVI
(s. 94 and 96)
MARINE PLANT HARVESTING DISTRICTS AND CLOSE TIMES+
|
Item |
Column I: Harvesting Districts |
Column II: Period |
|
1. |
District 1. Waters adjacent to the west shore of Prince Edward Island between West Point, Prince County, and North Point, Prince County. |
November 1 to June 10 |
|
2. |
District 2. Waters adjacent to the north shore of Prince Edward Island between North Point, Prince County, and the Prince, Queens Counties boundary. |
November 1 to July 30 |
|
3. |
District 3. Waters adjacent to the north shore of Queens County, Prince Edward Island. |
November 1 to June 30 |
|
4. |
District 4. Waters adjacent to Kings county, Prince Edward Island. |
November 1 to June 30 |
|
5. |
District 5. Waters adjacent to the south shore of Queens County, Prince Edward Island. |
November 1 to June 6 |
|
6. |
District 6. Waters adjacent to the south shore of Prince County, Prince Edward Island, between the Prince, Queens Counties boundary and West Point. |
November 1 to June 6 |
|
7. |
District 7. Waters adjacent to the north and northeast shores of New Brunswick between the Quebec and Nova Scotia borders. |
November 1 to June 6 |
|
8. |
District 8. Waters adjacent to the north shores of Cumberland, Colchester, and Pictou Counties, Nova Scotia. |
November 1 to June 30 |
|
9. |
District 9. Waters adjacent to the shores of Antigonish and Inverness Counties, Nova Scotia. |
November 1 to June 30 |
|
10. |
District 10. Waters adjacent to the shores of Victoria, Cape Breton, and Richmond Counties, Nova Scotia. |
November 1 to July 15 |
|
11. |
District 11. Waters adjacent to the shores of Guysborough, Halifax, and Lunenburg Counties, Nova Scotia. |
November 1 to June 30 |
|
12. |
District 12. Waters adjacent to the shores of Queens, Shelburne, Yarmouth, Digby, Annapolis, and Kings Counties, Nova Scotia. |
November 1 to June 6 |
|
13. |
District 13. Waters adjacent to the shores of Kings County and the south shores of Colchester and Cumberland Counties, Nova Scotia. |
November 1 to June 6 |
|
14. |
District 14. Waters adjacent to the south shore of Westmoreland County and the shores of Albert, Saint John, and Charlotte Counties, New Brunswick. |
November 1 to June 30 |
+Close times apply to Irish moss (Chondrus crispus) and Gigartina stellata) only.
PART IX
MARINE PLANTS
94. In this part,
"District" means a harvesting district described in Schedule XXVI; (arrondissement)
"rockweed" means a brown marine plant of the species Ascophyllum nodosum; (Ascophyllum n nodosum) "holdfast," in relation to a marine plant, means that portion of the marine plant by which it is attached to the ocean floor, (pied)
Harvesting Methods and Gear
95. (1) Subject to the conditions of a licence, no person shall harvest rockweed unless he
(a) uses an instrument that severs with a cutting action; and
(b) leaves a length of at least 127 mm above the holdfast.
(2) No person shall have in his possession any rockweed plant to which the holdfast is attached.