Abstract
Résumé
Introduction
Materials and methods
Results
Discussion
Conclusion
References
M.O. Akusu, E. Nduka and B.A. Soyebo
University of Ibadan
Ibadan, Nigeria
Oestrus was synchronised with PGF2-alpha in four pluriparous West African Dwarf (WAD) goats and blood collected through jugular venupuncture. The length of the oestrous cycle and oestrus duration were observed in three successive cycles while peripheral plasma progesterone and oestradiol-17B levels were determined by the radioimmunoassay technique. The mean oestrous cycle lengths and oestrus duration were 19.4 ± 1.7 d and 33.3 ± 6.6 h, respectively. Progesterone levels were very low during the first four days of the cycle (day 1 = day of oestrous) with the lowest level of 0.3 ± 0.02 ng/ml being recorded on day 2 of the cycle. There was a steady rise in the levels during the cycle until day 15 when the highest level of 2.2 ± 0.05 ng/ml was recorded which was followed by a sharp decline on day 20 (P <0.05). The profile of oestradiol-17B was erratic during the cycle. Mean maximum and minimum concentrations were recorded on day 1 (152.6 ± 31.6 ng/ml) and day 2 (58.8 ± 22.6 ng/ml), respectively. These results showed that while progesterone and oestradiol profiles in the WAD goat followed the luteal and follicular phases of the oestrous cycle, progesterone profile is more reliable in assessing the phases of the oestrous cycle.
L'oestrus de quatre chèvres naines d'Afrique occidentale multipares a été synchronisé avec du PGF2-alpha; des échantillons de sang ont été collectés à travers la veine jugulaire. Les durées du cycle oestral et de l'oestrus de trois cycles successifs ont été mesurées, et les taux de progestérone et d'oestradiol-17B déterminés par radio-immunologie. La durée moyenne du cycle oestral était de 19,4 ± 1,7 jours et celle de l'oestrus de 33,3 ± 6,6 h. Très faible au cours des quatre premiers jours du cycle, la teneur en progestérone du sang périphérique atteignait son niveau minimum au deuxième jour du cycle, avec une valeur de 0,3 ± 0,02 ng/ml. Elle augmentait ensuite régulièrement jusqu'au 15e jour du cycle où elle atteignait le pic de 2,2 ± 0,05 ng/ml, avant d'enregistrer une chute brutale au 20e jour (P<0,05). Le taux d'oestradiol-17B a varié de manière erratique; les teneurs maximum (152,6 ± 31,6 ng/ml) et minimum (58,8 ± 22,6 ng/ml) ayant été enregistrées respectivement au premier et au deuxième jours du cycle oestral. Ces résultats montrent que, bien que les niveaux d'oestradiol et de progestérone suivent les phases lutéique et folliculaire, la progestérone est plus indiquée pour la détermination de la phase du cycle oestral à laquelle se trouve la chèvre naine d'Afrique de l'Ouest.
Progesterone and oestradiol-17B are ovarian hormones that have several practical biological applications in the management of reproduction in animals They are involved in oestrus manifestations, the ovulatory process, cyclic regression of the corpus luteum, establishment of pregnancy and parturition. Progesterone levers have also been used in the determination of the onset of puberty in cattle (Diaz et al, 1986), pigs (Shille et al, 1979) and sheep (Oyedipe et al, 1986) and verification of the accuracy of oestrus detection (Sawyer et al, 1986). A knowledge of the levels of these hormones can be of benefit for diagnostic and therapeutic purposes in cases of endocrine dysfunction involving normal sexual activity of the female.
The levels of these hormones during the oestrous cycle and pregnancy have been determined in several species of domestic animals, especially in sheep and cattle (Scaramuzzi, 1976; Adeyemo and Health, 1980; Voh et al, 1987). Compared to the other species, fewer reports are available for the goat (Mori and Kano, 1984; Akusu et al, 1989).
The objective of this study was to generate basal data on the levels and profiles of progesterone and oestradiol-17B during oestrous cycle of the West African Dwarf goat. This breed of goat is the most numerous of the ruminant species in the humid rainforest zone of southern Nigeria (FAO, 1966; Carew, 1982) and therefore represents the quickest means of increasing meat production for the populace through oestrous synchronisation and artificial insemination.
Four pluriparous WAD goats, aged 3-4 years and weighing between 15-20 kg were used for this study. They were managed intensively on concrete-floored pens in the small ruminant unit of the Department of Veterinary Surgery and Reproduction, University of Ibadan. Animals were fed on a corn-based ration (0.5 kg/d). Clean water and Giant Stargrass were constantly available.
Oestrus was synchronised with 10 mg PGF2 a as described previously (Akusu and Egbunike, 1984; Akusu et al, 1989). Animals were teased for oestrus five times daily at intervals of 4 hours between 08.00 and 24.00 h with an apronned sexually mature buck.
Animals were bled through jugular venupuncture and plasma stored at-20°C until assayed for progesterone and oestradiol-17B by radioimmunoassay techniques (Dada et al, 1984; Akusu et al, 1989). Hormone concentration was calculated from linearised logit-log plots of standard curves. Inter- and intra-assay variations for progesterone were 11.5% and 10.9%, the corresponding values for oestradiol-17B were 10.3% and 11.5%, respectively.
Blood was collected during the first five days of the oestrous cycle (day 1 = day of oestrus) and then on days 10, 15 and 20. The study was carried out during three successive oestrus cycles.
Oestrus was synchronised in all goats treated with PGF2-a The mean oestrus duration was 33.3 ± 6.6 h while oestrous cycle occurred at 19.7 ± 1.7 d.
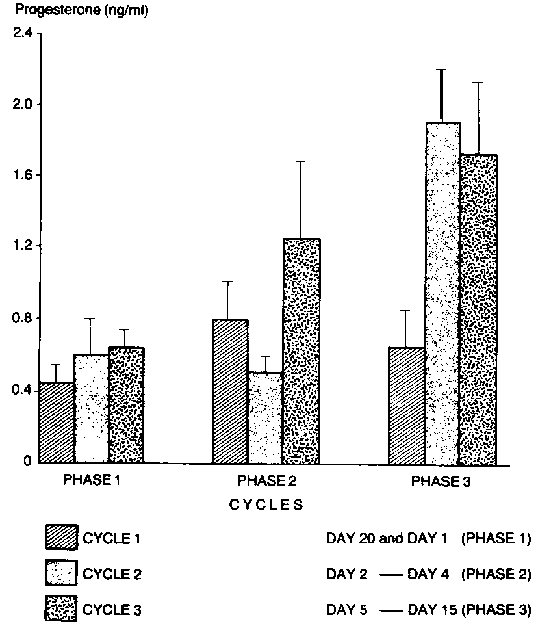
Peripheral plasma progesterone and oestradiol-17B profiles are summarised in Figure 1. The levels of progesterone were generally lower than 1 ng/ml during oestrus and metoestrus (d 1-d 4). The lowest level of 0.3 ± 0.02 ng/ml was determined on d 2 of the cycle. -This was followed by a gradual rise in d 3 and d 4 (0.5 ± 0.03 ng/ml and 0.87 ± 0.02 ng/ml, respectively). D4 value was significantly higher than d 2 (P<0.05). There was a sharp rise on d 5 and remained stable up to d 10 and a further rise in progesterone level to a peak value of 2.2 ± 0.05 on d 15 followed by a sharp decline (P<0.05) by d 20. There were differences in the levels between the three cycles but the profiles were not affected (Figure 2).
Figure 1. Peripheral plasma levels of progesterone and oestradiol- 17ß of West African Dwarf goats during the oestrous cycle.
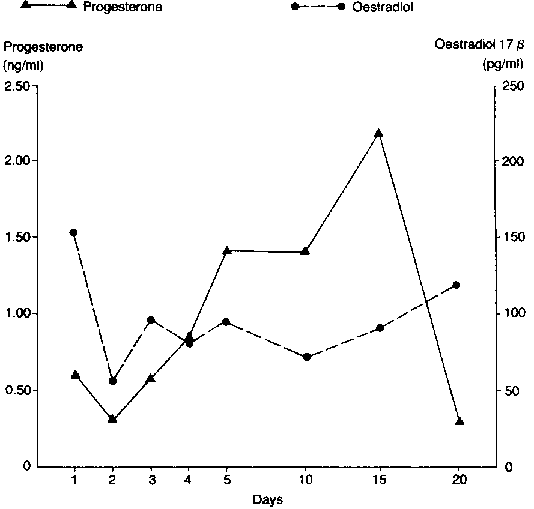
Figure 2. Peripheral plasma progesterone profiles during three successive oestrous cycles in WAD goats (X ± Sem).
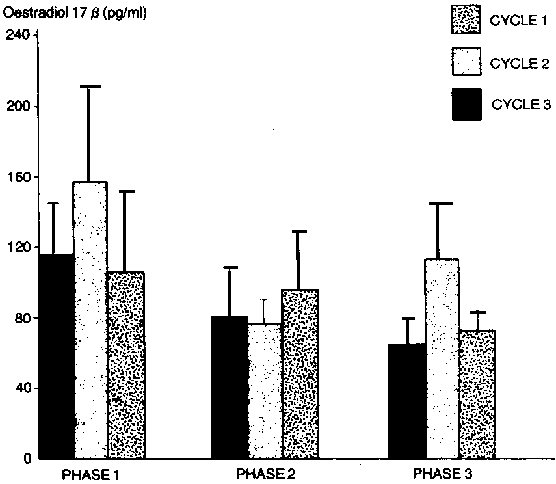
Peripheral plasma levels of oestradiol-17B were highest on d 1 (152.6 ± 31.6). There was a rapid decline (P<0.05) on d 2. Thereafter, there were fluctuations in the levels but a gradual rise was observed Up to d 20 (131.7 ± 44.3 ng/ml). The profile of oestradiol was similar irrespective of the cycle (Figure 3).
Figure 3. Peripheral plasma oestradiol 17B profiles in WAD goats during three successive oestrous cycles.
The general profile of progesterone and oestradiol-17B of the West African Dwarf goat was similar to reports in various species of domestic animals (Shearer et al, 1972; Sarda et al, 1973; Perera et al, 1978; Arora and Pandey, 1982; Oyedipe et al, 1986; Danell, 1987). The mean level of progesterone of the WAD goat was lower than the values reported for other breeds of goats (Heap and Linzell, 1966; Thornburn and Schneider, 1972). This may be a breed characteristic which has been reported in the ewe (Bindon et al, 1979).
The individual variations in the peak values of progesterone and oestradiol-17B in the goats may be due to variations in individual traits that may be correlated with expression of overt heat signs and may serve as an index of selection (Perera, 1979). However, it was observed that all does had a common pattern with relatively levels Of progesterone during the luteal phase, a decline between the 15th and 20th d of the cycle and values approximating zero at and around oestrus. Similarly, the peak values Of oestradiol-17B generally corresponded with the follicular and luteal phases of the oestrous cycle. It showed a fluctuating level of between 58 and 92 pg/ml until the day preceding the onset of behavioural oestrus when an increase was demonstrated. The peak value was for a day and there was no subsequent rise. This was consistent with the report of Shearer et al (1972) and therefore suggested that the psychic manifestation of behavioural oestrus in the goat is the result of oestradiol acting on the central nervous system (McDonald, 1977; Hansel and Convey, 1983).
These results showed that while progesterone and oestradiol profiles in the WAD goat followed the luteal and follicular phases Of the oestrous cycle, progesterone profile is more reliable in assessing the phases of the oestrous cycle.
Adeyemo O and Health E.1980. Plasma progesterone concentration in Bos taurus heifers. Theriogenology 14:411-420.
Akusu M O and Egbunike G N.1984. Fertility of the West African dwarf goat in its native environment following PGF2 induced oestrus. Veterinary Quarterly 6:173-176.
Akusu M O, Nduka E and Egbunike G N.1989. Peripheral plasma levels of progesterone and oestradiol-17B during the reproductive cycle of West African Dwarf goats. In: Wilson R T and Azeb Melaku (eds), African Small Ruminant Research and Development. Proceedings of a Conference held at Bamenda, Cameroon, 18-25 January 1989. ILCA (International Livestock Centre for Africa), Addis Ababa, Ethiopia. pp. 316-328.
Arora R C and Pandey R S. 1982. Pattern of plasma progesterone, oestradiol-17B, luteinizing hormone and androgen in non pregnant buffalo (Bubalus bubalis). Acta endocrinologica 100:279-284.
Bindon B M, Blanc M R. Pelletier J. Terqui M and Thimonier J. 1979. Peri-ovulatory gonadotropin and ovarian steroid patterns in sheep of breeds with differing fecundity. Journal of Reproduction and Fertility 55: 15-25.
Carew B A R.1982. Production potential and nutritional studies of goats and sheep in south western Nigeria. PhD thesis, University of Ibadan, Nigeria.
Dada O A, Osinusi B O, Nduka E U. Osotimehin B O and Ladipo O A.1984. 17B oestradiol, progesterone and testosterone in the normal menstrual cycle of Nigerians. International Journal of Gynaecology and Obstetrics 22:151 -154.
Danell B. 1987. Oestrous behaviour, ovarian morphology and cyclical variation in follicular system and endocrine pattern in water buffalo heifers. Ph D thesis, Swedish University of Agricultural Sciences, Uppsala, Sweden.
Diaz T. Manco M, Troconiz J. Benacchio N and Verde O.1986. Plasma progesterone levels during the oestrous cycle of Holstein and Brahman cows. Carora type and cross-breed heifers. Theriogenology 26:419-432.
FAO (Food and Agriculture Organization of the United Nations). 1966. Agricultural development in Nigeria 1965-1980. FAO, Rome, Italy. 512 pp.
Hansel W and Convey E M. 1983. Physiology of the oestrous cycle. Journal of Animal Science 57: Supplement 2:404-423.
Heap R B and Linzell J L. 1966. Arterial concentration, ovarian secretion and mammary uptake of progesterone in goats during the reproductive cycle. Journal of Endocrinology 36:389-399.
McDonald L E. 1977. Veterinary endocrinology and reproduction. 2nd edition. Lea and Febiger, Philadelphia, USA. 560 pp.
Mori Y and Kano Y. 1984. Changes in plasma concentrations of luteinizing hormone, progesterone and oestradiol in relation to the occurrence of luteolysis, oestrus and time of ovulation in the Shiba goat. Journal of Reproduction and Fertility 72:223-230.
Oyedipe E O. Pathiraja N. Edqvist L E and Buvanendran V. 1986. Onset of puberty and oestrous cycle phenomena in Yankasa ewes as monitored by plasma progesterone concentrations. Animal Reproduction Science 12:195-199.
Perera B M A O. 1979. Radioimmunoassay of progesterone in the blood of buffaloes during normal and prostaglandin synchronised oestrous cycles. In: Sastradipradja D (ed), Proceedings of the Second Research Coordination Meeting of the Joint FAO/IAEA Division of Atomic Energy in Food and Agriculture held in Bogor, Indonesia. pp.65-72.
Perera B M A O. Pathiraja N. Buvanendran V, Abeywardena S A and Piyasena R D.1978. Plasma progesterone levels during natural and prostaglandin-synchronised oestrous cycles in buffaloes. Ceylon Veterinary Journal 26:29-34.
Sarda I R. Robertson H A and Smeaton T C. 1973. Sequencial changes in plasma progesterone levels in the ewe during the oestrous cycle and during pregnancy in intact and ovariectomized sheep. Canadian Journal of Animal Science 53:25-34.
Sawyer G J. Russel-Brown I D and Silcock J K. 1986. A comparison of three methods of oestrus detection in commercial dairy herds verified by serum progesterone analyses. Animal Production Science 10:1-10.
Scaramuzzi R J. 1976. Inhibition of oestrous behaviour in ewes by passive immunization against oestradiol 17-Beta. Journal of Reproduction and Fertility 42:145-148.
Shearer I J. Purvis K, Jenkins G and Haunes N B. 1972. Peripheral plasma progesterone and oestradiol-17B levels before and after puberty in gills. Journal of Reproduction and Fertility 30:347- 360.
Shille V M, Karlbom I, Einarsson S. Larsson K, Kindahl H and Edqvist L E. 1979. Concentrations of progesterone and 15-keto-13, 14-dihydroprostaglandin F2 in peripheral plasma during the oestrous cycle and early pregnancy in gilts. Zoobiology and Veterinary Medicine. A26:169-181.
Thornburn G D, and Schneider W. 1972. The progesterone concentration in the plasma of the goat during the oestrous cycle and pregnancy. Journal of Endocrinology 52:23-36.
Voh Jr A A, Oyedipe E O. Pathiraja N. Buvanendran V, and Kumi-Diaka J.1987. Peripheral plasma levels of progesterone in Nigerian zebu cows following synchronization of oestrus with prostaglandin F2-Alpha analogue (Dinoprost Tromethamine). British Veterinary Journal 143 (3):254-263.