
Animal health

9 May 2024, 08:30 hours; Rome
Situation: High pathogenicity avian influenza (HPAI) virus (H5, H5N1, H5N2, H5N6, H5N8, H7 and H7N6 subtypes) with pandemic potential in countries of Sub-Saharan Africa since February 2017.
Confirmed countries (H5): South Africa*
Confirmed countries (H5N1): Benin, Botswana, Burkina Faso*, Cameroon, Côte d’Ivoire, Réunion (France), Gambia, Gabon, Ghana, Guinea, Lesotho, Mali, Mauritania, Namibia, Niger*, Nigeria*, Senegal, South Africa, and Togo.
Confirmed countries (H5N2): Nigeria and South Africa.
Confirmed countries (H5N6): Nigeria.
Confirmed countries (H5N8): Cameroon, Democratic Republic of the Congo, Namibia, Niger, Nigeria, South Africa, Uganda, and Zimbabwe.
Confirmed countries (H7): Mozambique*
Confirmed countries (H7N6): South Africa*
Animal findings: 2 new events since the last update on 12 April 2024.
Number of human cases: 0 new events since last update (Last reported case 10 November 2021).
*Countries reporting cases in current wave (since 1 October 2023).
Map. Officially reported HPAI outbreaks (H5, H5N1, H5N2, H7 and H7N6 subtypes) in sub-Saharan Africa, by onset date (1 October 2023 to date)
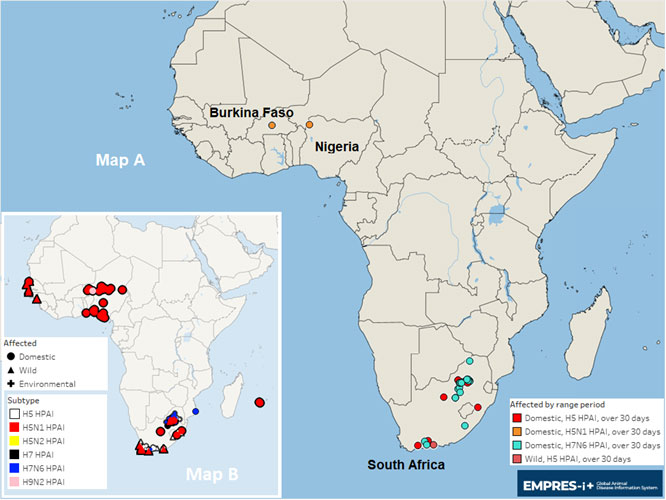
Note: Map A shows confirmed HPAI events observed from 1 October to 9 May 2024 (current wave).
Map B shows HPAI events observed from 1 October 2022 to 30 September 2023 (previous wave).
Situation update
Table. High pathogenicity avian influenza events reported in animals since 1 October 2023 (i.e. current wave)
| Virus | Country (administrative regions affected) |
Last event observed | #Events since last update | Total #events since 1 October 2023 | Species affected since the last update or during the last observed events |
|---|---|---|---|---|---|
|
H5 |
South Africa |
29/02/2024 |
1 |
23 |
Commercial ostrich, poultry, Jackass Penguin, Swift Tern |
|
H5N1
|
Nigeria |
27/02/2024 |
0 |
3 |
Geese, Captive peacocks |
|
Burkina Faso |
01/03/2024 |
0 |
1 |
Poultry |
|
|
Niger |
27/01/2024 |
0 |
1 |
Domestic poultry |
|
|
H7 |
Mozambique |
29/09/2023 |
0 |
1 |
Layer chicken |
|
H7N6 |
South Africa |
18/12/2023 |
1 |
66 |
Domestic poultry and domestic non-poultry birds |
For a summary of H5N1, H5N6, and H5N8 HPAI events reported in sub-Saharan African countries in previous waves (i.e. before 1 October 2023) please contact EMPRES-Animal Health
Peer-reviewed publications
Bedair, N.M., Sakr, M.A., Mourad, A., Eissa, N., Mostafa, A. & Khamiss, O. 2024. Molecular characterization of the whole genome of H9N2 avian influenza virus isolated from Egyptian poultry farms. Archives of Virology, 169(5): 99. reference In this study, we sequenced the genome of an H9N2 avian influenza virus (AIV) isolated from chickens in Egypt in 2021. Phylogenetic analysis placed the strain within the G1 sublineage of the Eurasian lineage. While some segments were related to Egyptian genotype II, others were related to genotype I. Molecular analysis revealed mutations in the HA protein suggesting a preference for human-like receptors and changes in antigenic sites. Mutations in the NA protein were reminiscent of past human pandemic strains. Internal proteins showed virulence markers, indicating intrasubtype reassortment and potential for increased virulence. These findings underscore the evolving nature of AIV H9N2 and raise concerns about its future pathogenicity for both poultry and humans.
Kutkat, O., Gomaa, M., Aboulhoda, B.E., Moatasim, Y., El Taweel, A., Kamel, M.N., El Sayes, M., et al. 2024. Genetic and virological characteristics of a reassortant avian influenza A H6N1 virus isolated from wild birds at a live-bird market in Egypt. Archives of Virology, 169(5): 95. reference The first human infection with avian influenza A/H6N1 in Taiwan in 2013 raised concerns about this virus. During routine surveillance in Egyptian live-bird markets, an H6N1 virus was isolated from a garganey duck and characterized as A/Garganey/Egypt/20869C/2022(H6N1). Phylogenetic analysis revealed a unique genomic makeup, with segments inherited from various subtypes previously found in Egypt and Eurasian countries. Replication kinetics in mammalian cell lines (A549, MDCK, and Vero cells) showed efficient replication in C57BL/6 mice without prior adaptation, with faster growth and higher titers than the ancestral strain A/Quail/HK/421/2002(H6N1). These findings suggest that reassortant H6 AIVs could pose a threat to human health, emphasizing the importance of ongoing surveillance for H6 Avian Influenza virus in nature.
Olawuyi, K., Orole, O., Meseko, C., Monne, I., Shittu, I., Bianca, Z., Fusaro, A., et al. 2024. Detection of clade 2.3.4.4 highly pathogenic avian influenza H5 viruses in healthy wild birds in the Hadeji-Nguru wetland, Nigeria 2022. Influenza Other Respiratory Viruses, 18(2): e13254. reference In Nigeria, multiple avian influenza virus (AIV) subtypes have caused poultry outbreaks, potentially linked to trade and wild birds. A study at the Hadejia-Nguru wetlands collected 452 swabs from wild aquatic birds, identifying highly pathogenic H5N1 AIV in clinically healthy species. Prevalence was 11.1%, with phylogenetic analysis showing clade 2.3.4.4b. These viruses differed from those in Nigerian poultry, suggesting separate introductions. Whole-genome characterization revealed mammalian adaptive marker E627K in Afro-tropical resident aquatic ducks, indicating zoonotic potential. Surveillance in wild birds is crucial for monitoring virus diversity, enhancing epidemiological understanding, and facilitating risk assessment.
Fusaro, A., Zecchin, B., Giussani, E., Palumbo, E., Agüero-García, M., Bachofen, C., Bálint, Á., et al. 2024. High pathogenic avian influenza A(H5) viruses of clade 2.3.4.4b in Europe – why trends of virus evolution are more difficult to predict. Virus Evolution, veae027. [reference]
Li, Y., An, Q., Sun, Z., Gao, X. & Wang, H. 2024. Multifaceted analysis of temporal and spatial distribution and risk factors of global poultry HPAI-H5N1, 2005-2023. Animal, 18(3):101085. [reference]
Miller, L.N., Saadawi, W.K., Hamouda, W.B., Elgari, A.S., Abdulkarim, E.A., Lmrabet, A.M.M., Elbukhmari, A.E., et al. 2024. Assessing One Health capacities for transboundary zoonotic diseases at the Libya-Tunisia border. One Health Outlook, 6(1):3. [reference]
Si, Y., Skidmore, A. K., Wang, T., de Boer, W. F., Debba, P., Toxopeus, A. G., Li, L., & Prins, H. H. 2009. Spatio-temporal dynamics of global H5N1 outbreaks match bird migration patterns. Geospatial Health, 4(1):65–78. [reference]
Olawuyi, K., Orole, O., Meseko, C., Monne, I., Shittu, I., Bianca, Z., Fusaro, A., et al. 2024. The Public Health Importance and Management of Infectious Poultry Diseases in Smallholder Systems in Africa. Influenza Other Respir Viruses, 18(2):e13254. [reference]
Monjane, I. V. A., Djedje, H., Tamele, E., Nhabomba, V., Tivane, A. R., Massicame, Z. E., Arone, D. M., Pastori, A., Bortolami, A., Monne, I., Woma, T., Lamien, C. E. & Dundon, W. G. 2024. H7N6 highly pathogenic avian influenza in Mozambique, 2023. Emerging Microbes & Infections, 13, (1). [reference]
Sanogo, I.N., Guinat, C., Dellicour, S., Diakité, M.A., Niang, M., Koita, O.A., Camus, C. & Ducatez, M. 2024. Genetic insights of H9N2 avian influenza viruses circulating in Mali and phylogeographic patterns in Northern and Western Africa. Virus Evolution, 10(1):veae011. [reference]
Glazunova, A., Krasnova, E., Bespalova, T., Sevskikh, T., Lunina, D., Titov, I., Sindryakova, I. & Blokhin, A. 2024. A highly pathogenic avian influenza virus H5N1 clade 2.3.4.4 detected in Samara Oblast, Russian Federation. Front Vet Sci, 11 – 2024. [reference]
Grace, D., Knight-Jones, T. J., Melaku, A., Alders, R. & Jemberu, W. T. 2024. The Public Health Importance and Management of Infectious Poultry Diseases in Smallholder Systems in Africa. Foods, 13(3), 411. [reference]
Kenmoe, S., Takuissu, G.R., Ebogo-Belobo, J.T., Kengne-Ndé, C., Mbaga, D.S., Bowo-Ngandji, A. & Ondigui Ndzie, J.L. et al. 2024. A systematic review of influenza virus in water environments across human, poultry, and wild bird habitats. Water Res X, 22:100210. [reference]
Roberts, L.C., Abernethy, D., Roberts, D.G., Ludynia, K., O'Kennedy, M.M., Abolnik, C. 2023. Vaccination of African penguins (Spheniscus demersus) against high-pathogenicity avian influenza.Vet Rec, e3616. [reference]
Abolnik, C. 2023. Spillover of an endemic avian Influenza H6N2 chicken lineage to ostriches and reassortment with clade 2.3.4.4b H5N1 high pathogenicity viruses in chickens. Vet Res Commun. [reference]
Fagrach, A., Arbani, O., Karroute, O., El-Ftouhy, F.Z., Kichou, F., Bouslikhane, M., Fellahi, S. 2023. Prevalence of major infectious diseases in backyard chickens from rural markets in Morocco. Vet World, 16(9):1897-1906. [reference]
Meseko, C., Ameji, N.O., Kumar, B. & Culhane, M. 2023. Rational approach to vaccination against highly pathogenic avian influenza in Nigeria: a scientific perspective and global best practice. Arch Virol, 168(10):263. [reference]
Agha, A.S.K., Benlashehr, I., Naffati, K.M., Bshina, S.A. & Khashkhosha, A.A. 2023. Correlation of avian influenzaH9N2 with high mortality in broiler flocks in the southwest of Tripoli, Libya. Open Vet J, 13(6):715-722. [reference].
Bongono, E.F., Kaba, L., Camara, A., Touré, A., Ngoma, M.P., Yanogo, P.K., Kanyala, E. & SOW A. 2023. Évaluation de la biosécurité et facteurs associés à l'influenza aviaire dans les fermes avicoles de Coyah, Guinée, 2019-2020. Med Trop Sante Int. 2023 3(2):25. [reference].
Isibor, P.O., Onwaeze, O.O., Kayode-Edwards, I.I., Agbontaen, D.O., Ifebem-Ezima, A.M., Bilewu, O., Onuselogu, C., Akinniyi, A.P., Obafemi, Y.D. & Oniha, M.I. 2023. Investigating and combatting the key drivers of viral zoonoses in Africa: an analysis of eight epidemics. Braz J Biol, 84:e270857. [reference]
Lebarbenchon, C., Boucher, S., Feare, C., Dietrich, M., Larose, C., Humeau, L., Le Corre, M. & Jaeger, A. 2023. Migratory patterns of two major influenza virus host species on tropical islands. R Soc Open Sci, 10(10):230600. [reference]
Jbenyeni, A., Croville, G., Cazaban, C. & Guérin, J.L. 2023. Predominance of low pathogenic avian influenza virus H9N2 in the respiratory co-infections in broilers in Tunisia: a longitudinal field study, 2018-2020. Vet Res, 54(1):88. [reference]
Alhaji, N.B., Adeiza, A.M., Godwin, E.A., Haruna, A.E., Aliyu, M.B. & Odetokun, I.A. 2023. An assessment of the highly pathogenic avian influenza resurgence at human-poultry-environment interface in North-central Nigeria: Sociocultural determinants and One Health implications. One health, 16:100574 [reference]
Miller, LmNm, Elmselati, H., Fogarty, A.S., Farhat, M.E., Standley, C.J., Abuabaid, H.M. & Zorgani, A. 2023. Using One Health assessments to leverage endemic disease frameworks for emerging zoonotic disease threats in Libya. PLOS Glob Public Health, 3(7):e0002005 [reference]
Abolnik, C., Phiri, T., Peyrot, B., de Beer, R., Snyman, A., Roberts, D., Ludynia, K. et al. 2023. The Molecular Epidemiology of Clade 2.3.4.4B H5N1 High Pathogenicity Avian Influenza in Southern Africa, 2021–2022. Viruses, 15(6):1383. [reference]
Meseko, C., Milani, A., Inuwa, B., Chinyere, C., Shittu, I., Ahmed, J., Giussani, E. et al. 2023. The Evolution of Highly Pathogenic Avian Influenza A (H5) in Poultry in Nigeria, 2021–2022. Viruses,15:1387. [reference]
Nma Bida Alhaji, Abdulrahman Musa Adeiza, Enid Abutu Godwin, Aliyu Evuti Haruna, Mohammed Baba Aliyu and Ismail Ayoade Odetokun. 2023. An assessment of the highly pathogenic avian influenza resurgence at human-poultry-environment interface in North-central Nigeria: Sociocultural determinants and One Health implications. One Health, 16:100574. [reference]
Lo, F.T., Zecchin, B., Diallo, A.A., Racky, O., Tassoni, L., Diop, A., Diouf, M., Diouf, M., Samb, Y.N., Pastori, A., Gobbo, F., Ellero, F., Diop, M., Lo, M.M., Diouf, M.N., Fall, M., Ndiaye, A.A., Gaye, A.M., Badiane, M., Lo, M., Youm, B.N., Ndao, I., Niaga, M., Terregino, C., Diop, B., Ndiaye, Y., Angot, A., Seck, I., Niang, M., Soumare, B., Fusaro, A. & Monne, I. 2022. Intercontinental Spread of Eurasian Highly Pathogenic Avian Influenza A(H5N1) to Senegal. Emerg Infect Dis. 28(1):234-237. [reference]
Panzarin, V., Marciano, S., Fortin, A., Brian, I., D'Amico, V., Gobbo, F., Bonfante, F., Palumbo, E., Sakoda, Y., Le, K.T., Chum D.H., Shittu, I., Meseko, C., Haido, A.M., Odoom, T., Diouf, M.N., Djegui, F., Steensels, M., Terregino, C. & Monne, I. 2022. Redesign and Validation of a Real-Time RT-PCR to Improve Surveillance for Avian Influenza Viruses of the H9 Subtype. Viruses. 14(6):1263. [reference]
FAO's support to countries or by country
Global level
- The FAO Virtual Learning Centers have launched a new self-paced virtual learning course on avian influenza (AI). The course aims to raise awareness of AI and to develop capacity on its detection and prevention. It is available here. This is an introductory course, designed for a range of stakeholders with an interest in AI. It is intended to be useful for veterinarians, veterinary paraprofessionals and others working in the poultry industries. There are six short modules, each of approximately fifteen to twenty minutes duration. You can study these at a time and place to suit you and use them as a ready-reference resource as needed. The course supports multiple platforms and can be studied on a smartphone, as well as on a computer or a tablet device.
- OFFLU (WOAH/FAO network of expertise on animal influenza) has launched a webpage for collection of relevant links on link.
- On 21 December 2023 OFFLU released its statement "Continued expansion of high pathogenicity avian influenza H5 in wildlife in South America and incursion into the Antarctic region”. [link]
Regional level
- FAO Emergency Center for Transboundary Animal Disease Control (ECTAD) for West and Central Africa (WCA) monitors suspected HPAI outbreaks in several countries in the region and provides various assistance, including laboratory reagents and consumables, as well as transport of specimens to the reference laboratory Istituto Zooprofilattico Sperimentale delle Venezie (IZSVe-Italy) for confirmation and sequencing.
- In West Africa, FAO ECTAD WCA has supported poultry value chain studies and the development of biosecurity manuals for poultry farms and live bird markets in Togo and Benin under the USAID funded project “Emergency assistance for prevention and control of Highly Pathogenic Avian Influenza (HPAI) in selected Countries in Africa”.
- FAO ECTAD WCA held a Regional Training of Surveillance Evaluation Tool (SET) Evaluators in West and Central Africa in Abidjan (Cote D’Ivoire) from 25 to 29 March 2024.
- FAO ECTAD regional offices in Eastern and Southern Africa (ESA) supports procurement of reagents (primers, probes, polymerase chain reaction (PCR) kits, extraction kits) and consumables in support of timely diagnostic testing and to enhance preparedness in Ethiopia, Kenya, the United Republic of Tanzania and Uganda through USAID funded GHSA programme.
- ECTAD ESA is supporting Kenya, Ethiopia, the United Republic of Tanzania, Zambia, Zimbabwe enhance biosecurity in poultry farms which contributes to minimising HPAI incursion. Poultry value chain risk assessment along the entire poultry value chain is planned in 10 countries namely Ethiopia, Kenya, Madagascar, Malawi, Mozambique, Rwanda, South Sudan, the United Republic of Tanzania, Uganda and Zambia as a part of ECTAD's ongoing support for HPAI risk mitigation/management.
- FAO ECTAD ESA and WCA have been coordinating annual Proficiency testing schemes supported by USAID for AIV diagnosis at national and sub-national level since 2008, through Letter of Agreement signed between FAO and WOAH reference laboratory (IZSVe- Italy) prepared by ECTAD HQ.
National level
Benin:
- A poultry value chain study was conducted, resulting in the development of a biosafety manual aimed at enhancing biosafety measures in poultry farms and live bird markets. Reports titled "Study of Poultry Value Chains and Risk Factors for Highly Pathogenic Avian Influenza (HPAI) in Benin" have been finalized.
- A training of trainers on biosecurity measures and best practices for managing poultry farms was conducted, using both the Biosecurity Manual for Commercial Poultry Farms and the Biosecurity Manual for Live Poultry Markets in Benin.
- FAO ECTAD supported Benin by providing reagents, disinfectants, and personal protective equipment (PPE).
Burkina Faso:
- Burkina Faso released an alert for poultry farmers regarding HPAI outbreak in Ouagadougou on 6 March 2024. FAO is providing supports with laboratory reagents, consumables, PPE, as well as transport of samples to the reference laboratory (IZSVe) for confirmation and sequencing.
- As part of the Technical Cooperation Programme (TCP) project TCP/BKF/3901, FAO is supporting the strengthening of capacities to combat HPAI as well as the preparation for the implementation of recovery mechanisms for the poultry industry. The project has trained 175 people in epidemiological surveillance of HPAI; and to develop training modules on good poultry farming practices, hygiene and biosecurity measures on farms with the training of 46 trainers; the trainers raised awareness among 300 model poultry breeders from 10 regions of Burkina. The project also made it possible to train 30 communicators and journalists from the written and audiovisual press on HPAI.
- Under the FAO TCP project on HPAI, the General Directorate of Veterinary Services conducted active surveillance around the outbreak and developed eight tools through a Letter of Agreement. These include: Case Definition of HPAI, HPAI Investigation Form, HPAI Notification Form, HPAI Outbreak Information/Management Form, Standard Operating Procedure (SOP) for sampling and diagnosis in case of suspected HPAI, SOP on communication during an HPAI crisis, SOP on the donning/doffing PPE, SOP on biosecurity measures in poultry farms in case of suspected HPAI.
Côte d’Ivoire:
- FAO ECTAD Côte d'Ivoire helped the government to carry out surveys through the acquisition of field equipment and to strengthen the capacities of the national laboratory for diagnosis of diseases such as HPAI. Standard operating procedures for investigating events in poultry have been revised, including outbreak investigation sheets.
Democratic Republic of the Congo:
- Following the discovery of avian influenza in South Africa and taking into account trade between South Africa and the Democratic Republic of the Congo, the veterinary services have been put on alert. FAO, through ECTAD, had already pre-positioned Personal Protective Equipment (PPE) and other sampling equipment in high-risk provinces. Apart from strengthening national laboratories, other measures have also been taken to help the government send samples to international reference laboratories for confirmatory testing and molecular characterization if necessary.
Ethiopia:
- As part of the government's strategic initiative known as "Yelemat Turufat," the Ministry of Agriculture has developed a comprehensive National Biosecurity Management Guideline for Poultry Farms in January 2024. This guideline aims to establish a standardized framework of biosecurity practices that are applicable to all poultry producers and stakeholders involved in the industry. The finalization of the guideline is underway, and it will serve as a valuable resource to provide guidance and support to poultry farm owners in implementing effective biosecurity measures.
- Voluntary producers (host farms) have been identified from four districts, namely Sebeta, Bishoftu, Bahir Dar, and Debre Birhan, to enroll in the Broiler Farmer Field School programme. This programme aims to mitigate AMR and generate data for action in agrifood systems using a One Health Approach. FAO ECTAD, in collaboration with the Animal Health Institute and local animal health practitioners, is conducting a baseline assessment on the broiler farms. This assessment seeks to evaluate the farm premises' biosecurity, antimicrobial resistance, and antimicrobial usage statuses, as well as gather essential farm economic data.
Gambia:
- A train-the-trainers workshop on improving biosecurity on poultry farms was organized, training 37 poultry value chain stakeholders and decision-makers. A Good Emergency Management Practices (GEMP) training mission trained 52 participants from different sectors in animal health emergency management, based on the FAO GEMP manual.
Ghana:
- Community event-based surveillance is reinforced with support of the FAO Event Mobile Application (EMA-i).
Guinea:
- Over the coming months, FAO plans to support the training of actors in the value chain of the poultry sector on biosecurity, the training of veterinary service agents from very high- and high-risk prefectures on good management practices, emergencies, carrying out local investigative surveys and active surveillance in the field, and supervision missions of field investigations; because the disease is still far from being controlled.
- FAO ECTAD supports Guinea by providing reagents, disinfectants and PPE.
Kenya:
- FAO is supporting Kenya Animal Bio-Surveillance (KABS) disease reporting platform roll-out for syndromic surveillance and the refresher trainings.
Liberia:
- The Central Veterinary Laboratory has participated in EQA (Proficiency testing) for HPAI organized by the WOAH/FAO International Reference Laboratory for Animal influenza and Newcastle Disease (APHA) with the support of FAO.
- The Ministry of Agriculture in collaboration with FAO plans to conduct active surveillance for Low Pathogenicity Avian Influenza (LPAI)/HPAI in five high risk counties (Montserrado, Nimba, Grand Cape Mount, Bomi, Grand Bassa) in May 2024.
Mali:
- FAO ECTAD Mali conducted a study on biosecurity in poultry, dairy and pig farms to assess best practices and recommend actions to improve biosecurity measures on farms. Following the recommendations from the study, training is planned for stakeholders (women and young people) working in poultry, dairy and pig farms, which will take place in 2023 and 2024.
Niger:
- The outbreak on a family farm in Niamey (domestic poultry) in January 2024 has been the only confirmed case to date. In February 2024, six suspicions were reported in the regions of Maradi (1), Tahoua (1), Zinder (1), and Niamey (3). Samples analyzed by Laboratoire Central de l'Elevage (LABOCEL) tested negative. However, a case of low-pathogenic avian influenza was confirmed in a backyard guinea fowl farm in Niamey.
- FAO ECTAD supported with rapid test kits as well as PCR reagents (probes and primers) from the stockpile project, and ongoing support for the dispatch of samples from the February outbreak to the reference laboratory.
Nigeria:
- On 1 February 2024 peacock mortality was reported on a farm in Kebbi State, northwest Nigeria. The peacocks became ill a few days after local birds from several sources were introduced into the farm. H5N1 HPAI was confirmed by the National Veterinary Research Institute (NVRI) on 8 February and between 20 and 27 February, two outbreaks of H5N1 HPAI were detected in Plateau State [link]. FAO ECTAD supported the Federal Ministry of Agriculture and Food Security and other One Health stakeholders in conducting follow-up investigations into the outbreak and ensuring disease containment. FAO ECTAD provided PPE, disinfectants, sampling equipment and biohazard bags for the field survey. Additional samples were collected and sent to the laboratory. In addition, FAO ECTAD visited live poultry markets in neighbouring towns to raise awareness of biosecurity measures, in particular, the risk of mixing different species in the same space/location was emphasised, and the alternate to put each animal species in its own space was stressed [link].
- FAO ECTAD is also purchasing motorized sprayers and PPE to help veterinary services improve implementation of certain biosecurity measures and response to outbreaks. FAO will participate in the meeting of the Ministerial Advisory Committee of Experts on control measures for HPAI to be held in March/April 2024.
Senegal:
- An HPAI active surveillance mission at sentinel ornithological sites was conducted from 20 to 27 January 2024 in Saint Louis, Fatick and the Ziguinchor regions, to detect HPAI virus during this high-risk period, as migratory birds arrive in Senegal. A total of 350 swab samples of fresh droppings were collected in six parks and reserves in the regions of Saint-Louis, Fatick, and Ziguinchor. These include the Djoudj National Bird Sanctuary, the Langue de Barbarie National Park, the Gueumbeul Wildlife Reserve, the Saloum Delta National Park, the Abéné Marine Protected Area, and the Palmarin Community Reserve. Laboratory analysis results of the samples collected all tested negative for avian influenza.
- In addition to active surveillance, passive surveillance and vigilance in the main risk area continue to be implemented.
Sierra Leone:
- FAO ECTAD supported the Ministry of Agriculture and Food Security to develop guidelines for Infection Prevention and Appropriate Antimicrobial Use in the poultry sector. The guidelines will provide all poultry farmers - from small scale to large commercial - with routine biosecurity procedures to reduce occurrence and spread of infections on the farms and promote effective and safe use of drugs.
Somalia:
- FAO is implementing a pilot poultry value chain development project.
Togo:
- FAO supports active surveillance missions in live poultry markets and organized training for 18 trainers on biosecurity measures and good breeding practices. Also, FAO trained 43 Village Livestock Assistants, including two women, on community surveillance to improve early warning for the detection and rapid response to diseases.
Uganda:
- FAO ECTAD and Ministry of Agriculture, Animal Industry and Fisheries are finalizing the HPAI surveillance plan. At the time of this report, no recent HPAI surveillance reports have been received from the Animal Health Sector.
Zambia:
- Active surveillance activities were conducted in the past 3 months (January 2024-March 2024). A total of 600 samples were collected from layer chickens (22-75 weeks) in 5 breeder farms from Copperbelt, Central, Lusaka and Southern provinces. All samples tested negative for M, H5 and H7 genes by PCR at the Central Veterinary Research Institute (CVRI) in Lusaka.
ECTAD ESA
- Despite no HPAI events reported in the region FAO-ECTAD continues to support annual Proficiency testing schemes for AIV since 2018 through USAID funded GHSA program at national and sub-national level in Eastern Africa (Ethiopia Kenya, Tanzania and Uganda) as well as supporting procurement of reagents (molecular supplies-primers, probes, PCR kits, extraction kits) and consumables in support of diagnostic testing and to enhance preparedness.
ECTAD WCA
- FAO ECTAD WCA organized a regional training on Shipment of Infectious Substances from 26 to 28 July 2023 with a trainer from WHO. A total of 11 participants from 7 francophone countries successfully completed the training and were certified to ship infectious substances by air.
- FAO ECTAD WCA supported Benin, Burkina Faso, Côte d’Ivoire, Gabon, Ghana, Guinea, Mali, Mauritania, Niger, Nigeria, Senegal, Togo in managing domestic and wild bird mortality events in 2022 due to H5 HPAI including the provisions of laboratory reagents, consumables and PPE for field activities, as well as at-risk countries to address the situation as a regional coordination effort.
- In 2023, the regional project OSRO/GLO/501/USA “Emergency assistance for prevention and control of Highly Pathogenic Avian Influenza (HPAI) in selected Countries in West Africa” funded by USAID continues to enhance strengthening the capabilities of veterinary services to prevent and sustainably control HPAI within the region with a focus on Benin, the Gambia and Togo.
- FAO ECTAD WCA conducted backstopping missions: to Benin and Togo in from 9 to 13 May 2023 to provide follow up support also for the implementation of the HPAI project, assessed the readiness, reviewed planned activities, visited the veterinary laboratory of Lomé and provided recommendations to improve biosafety/biosecurity measures and working environment; and to the Gambia from 22 to 26 May 2023 to provide technical support in the HPAI project implementation, reviewed additional response needs for the recent HPAI outbreak and handed over emergency supplies (rapid test kits, disinfectants, personal protective equipment [PPE]) from the USAID funded Stockpile Project to the Department of Livestock Services of the Ministry of Agriculture.
Burkina Faso:
- From 25 to 27 June 2023, a meeting was held in Koudougou to raise awareness among small-scale poultry farmers on disease recognition and their responsibility on early detection of mortality. Forty-five poultry producers attended the session. Before the meeting, a ToT was organized and intended to field veterinary agents who will cascade the training in their respective regions. The objective is to get 650 poultry producers sensitized on HPAI and biosecurity.
- The TCP/BKF/3901 project has so far: trained 175 staff on HPAI epidemiological surveillance; developed training modules on good poultry farming practices, hygiene and biosecurity measures on farms and trained 46 trainers; the trainers conducted sensitization of 300 model poultry farmers from 10 regions of Burkina. The project also provided training for 30 communicators and journalists of the press (print and audiovisual) on HPAI.
Ethiopia:
- Diseases due to the absence or poor biosecurity account for more than 56% of annual mortality in the poultry sector in Ethiopia, discouraging farmers from expanding their production. FAO in collaboration with the Ethiopian Agricultural Research Institute has prepared a biosecurity brochure covering three key areas - conceptual, structural, and operational biosecurity measures. The brochure provides guidance to small and medium commercial poultry farms on implementing effective biosecurity measures for increased productivity, and a more sustainable and profitable industry, and is aimed to be used by Farmers Field School (FFS).
- FAO ECTAD Ethiopia, in collaboration with the Ethiopian agricultural research institute, implemented a pilot study in four municipalities to assess their current biosecurity practices and identify gaps, recommend best practices, assess the adoption by farms, and evaluate their effects on disease incidence/prevalence and farm productivity/profitability. The study was conducted on 40 poultry farms between July 2022 and March 2023, and the findings have been validated to suggest best practices to some poultry farms included in this study. The study will generate the evidence for poultry farm biosecurity which will inform commercial poultry farms the value of best practices, raise awareness and to scale up good practices along the poultry value chain, and promote infection prevention through biosecurity practices.
Gabon:
- An FAO Emergency Management Centre (EMC) emergency field mission was conducted in Gabon from 4 to 8 July 2022 in response to the recent H5N1 HPAI outbreaks reported in Estuaire Province. The FAO-EMC team collaborated with the General Directorate of Livestock to investigate affected farms, assess the emergency response capacity, identify the current gaps in the animal health surveillance system, including the capacity of the Central Veterinary Laboratory of Libreville for the diagnosis of priority diseases, and develop tailored recommendations. The outbreak has been managed and no additional outbreaks have been reported since mid-May 2022.
Ghana:
- FAO with support from USAID organized a one-day sensitization workshop in three regions in Ghana (Ashanti, Bono and Bono East) in January 2023 to prevent zoonoses resulting from handling, trade and consumption of bushmeat [link]
Liberia
- In December 2022, FAO-ECTAD Liberia supported the Ministry of agriculture to investigate suspected Outbreaks of HPAI in Nimba; all samples tested negative for HPAI.
Mali:
- In 2022, FAO ECTAD Mali supported Strengthen biosecurity practices at farm levels targeting 35 farmers including 8 women.
Niger:
- FAO ECTAD Niger supported the development of a Biosafety/Biosecurity guide (2022) for actors in the dairy, meat and poultry value chains in Niger.
Nigeria:
- FAO ECTAD Nigeria, in collaboration with ECOWAS and AU-IBAR, supported risk assessment for HPAI spread in the country in 2022. A debriefing session and an After-Action Review (AAR) on the SOP used for the mission were held virtually on 26 May and 24 July respectively, to the latter the national coordinator and epi-expert participated from ECTAD Senegal.
Senegal:
- FAO ECTAD Senegal assisted FAO Emergency Management Center (EMC-AH) expert mission visiting Saint-Louis as a part of support to control HPAI outbreaks in May 2023.
- FAO ECTAD Senegal trained 32 personnels from the Ministries of Environment and Livestock on sampling techniques for wild birds in January and February 2023.
Sierra Leone:
- In 2022, when neighbouring Guinea reported HPAI outbreaks, FAO ECTAD Sierra Leone supported the Ministry of Agriculture and Food security to undertake active search for HPAI in Koinadugu, Karene, Pujehun and Kambia districts adjacent to infected /high risk areas in Guinea collected 161 samples (serum and swabs) and tested at the Central Veterinary Laboratory using ELISA and PCR. All samples tested negative for H5.
- In collaboration with FAO ECTAD Guinea, FAO ECTAD Sierra Leone provided timely updates on HPAI status in Guinea during weekly One Health Emergency Preparedness and Response Group meetings which helped the Government to follow on the evolution of the outbreak and to assess potential threats of introduction of HPAI in Sierra Leone. This support complemented Government efforts to prevent HPAI incursions into the country.
Important links
- FAO-WHO-WOAH Joint FAO/WHO/WOAH preliminary assessment of recent influenza A(H5N1) viruses
- USDA Highly Pathogenic Avian Influenza (HPAI) Detections in Livestock
- WHO Avian Influenza A(H5N1) - United States of America
- WOAH High Pathogenicity Avian Influenza in Cattle
- FAO Stay vigilant with Highly Pathogenic Avian Influenza A(H5N1)
- CDC Technical Update: Summary Analysis of Genetic Sequences of Highly Pathogenic Avian Influenza A(H5N1) Viruses in Texas
- CDC How CDC is monitoring influenza data among people to better understand the current avian influenza A (H5N1) situation
- The list of wild bir and mammalian species which have been infected with H5Nx HPAI [link]
- Scientific Task Force on Avian Influenza and Wild Birds H5N1 High pathogenicity avian influenza in wild birds - Unprecedented conservation impacts and urgent needs
- EFSA Avian Influenza Overview December 2023 – March 2024
- EFSA Avian Influenza Overview reports [link]
- WHO Recommended composition of influenza virus vaccines for use in the 2024 southern hemisphere influenza season
- WHO Rapid risk assessment of risk associated with recent influenza A(H5N1) clade 2.3.4.4b viruses
- WHO Consultation on the Composition of Influenza Virus Vaccines related documents (from 2011 to date)
- WOAH Strategic challenges in the global control of high pathogenicity avian influenza [90SG/8]
- OFFLU The OFFLU website
- OFFLU HPAI Detections in Livestock page
- OFFLU Avian influenza matching (OFFLU-AIM) report
- OFFLU Avian data package for the VCM - February 2023 to September 2023
- OFFLU Summary reports of OFFLU contribution to WHO VCM (2013-2023)
- ECOWAS, FAO, USAID Report on the Regional Consultative Meeting on the Prevention and Control of Highly Pathogenic Avian Influenza (HPAI) in West Africa
- FAO-WHO-WOAH Tripartite Zoonotic Guide
- FAO-WHO-WOAH Tripartite Joint Risk Assessment (JRA) Operational Tool
- FAO-WHO-WOAH Ongoing avian influenza outbreaks in animals pose risk to humans - Situation analysis and advice to countries from FAO, WHO, WOAH
- FAO Report: Global consultation on highly pathogenic avian influenza (HPAI) 2-4 May 2023, Rome
- FAO Good emergency management practice - The essentials: A guide to preparing for animal health emergencies, the third edition
- FAO Manual for the management of operations during an animal health emergency
- FAO Effective disposal of animal carcasses and contaminated materials on small to medium-sized farms
- FAO Veterinary laboratory testing protocols for priority zoonotic diseases in Africa
- FAO Guiding principles for the design of avian influenza active surveillance in Asia: Designing active, comprehensive, risk-based avian influenza surveillance
- FAO Webinar: Managing large-scale highly pathogenic avian influenza outbreaks in wild birds: part 1, part 2
- FAO Addressing H5N1 Highly Pathogenic Avian Influenza Qualitative risk assessment on spread in the Central African region Issue no. 4, including H5N8 HPAI in Uganda and the risk to neighbouring countries.
- Focus On: 2016–2018 Spread of H5N8 highly pathogenic avian influenza (HPAI) in sub-Saharan Africa
- Focus On: Highly Pathogenic H5 Avian Influenza in 2016 and 2017–Observations and future perspectives
Disclaimer
Information provided herein is current as of the date of issue. Information added or changed since the last Sub-Saharan HPAI situation update appears in orange. For poultry cases with unknown onset dates, reporting dates were used instead. FAO compiles information communicated by field officers on the ground in affected countries, from regional offices, and from the World Organisation for Animal Health [WOAH], as well as peer-reviewed scientific articles. FAO makes every effort to ensure, but does not guarantee, accuracy, completeness or authenticity of the information. The boundaries and names shown and the designations used on these map(s) do not imply the expression of any opinion whatsoever on the part of FAO concerning the legal status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers and boundaries. Dashed lines on maps represent approximate border lines for which there may not yet be full agreement.
Contact
If interested in a previous issue please send an email to EMPRES-Animal Health specifying the intended use of the document.
